1,2-Butanediol
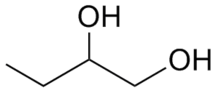
1,2-Butanediol is the organic compound with the formula HOCH2(HO)CHCH2CH3. It is classified as a vic-diol (glycol. It is chiral, although typically it is encountered as the racemic mixture. It is a colorless liquid.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Butane-1,2-diol | |
| Other names
1,2-Dihydroxybutane α-Butylene glycol | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.008.663 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties[1] | |
| C4H10O2 | |
| Molar mass | 90.121 g/mol |
| Density | 1.0023 g/cm3 (20 °C) |
| Melting point | −50 °C (−58 °F; 223 K)[note 1] |
| Boiling point | 195 to 196.9 °C (383.0 to 386.4 °F; 468.1 to 470.0 K) (96.5 °C at 10 mmHg) |
| miscible | |
| Solubility | soluble in ethanol, acetone; sparingly soluble in esters and ethers; insoluble in hydrocarbons |
Refractive index (nD) |
1.4378 (20 °C) |
| Viscosity | 7.3 mPa s (20 °C) |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
−532.8 kJ/mol [2] |
Std enthalpy of combustion (ΔcH⦵298) |
−2479 kJ/mol |
| Hazards[3] | |
| Safety data sheet | ICSC 0395 |
| Flash point | 90 °C (194 °F; 363 K) |
| Related compounds | |
Related butanediols |
1,3-Butanediol 1,4-Butanediol 2,3-Butanediol |
Related compounds |
Ethylene glycol Propylene glycol 2-Hydroxybutyraldehyde 2-Hydroxybutyric acid α-Ketobutyric acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation
It is produced industrially by hydration of 1,2-epoxybutane.[4][5]
1,2-Butanediol is a byproduct of the production of 1,4-butanediol from butadiene,[6] It is also a byproduct of the catalytic hydrocracking of starches and sugars such as sorbitol to ethylene glycol and propylene glycol.[7]
This diol was first described by Charles-Adolphe Wurtz in 1859.[8]
Applications
It has been patented for the production of polyester resins and plasticizers.[5][6] It is a potential feedstock for the industrial production of α-ketobutyric acid, a precursor to some amino acids.[9]
Notes
- The value of −50 °C for the melting point is taken from Ullmann's Encyclopedia of Industrial Chemistry and used by the Hazardous Substances Data Bank and the OECD Screening Information Dataset. Other reported values of the melting point range from −114 °C to −30 °C.
References
- Weast, Robert C., ed. (1981). CRC Handbook of Chemistry and Physics (62nd ed.). Boca Raton, FL: CRC Press. p. C-190. ISBN 0-8493-0462-8..
- Moureu, H.; Dode, M. (1937), "Chaleurs de formation de l'oxyde d'ethylene, de l'ethanediol et de quelques homologues", Bull. Soc. Chim. Fr., 4: 637–47.
- 1,2-Butanediol, International Chemical Safety Card 0395, Geneva: International Programme on Chemical Safety, March 1996.
- "Butanediols, Butenediol, and Butynediol". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. 2005. doi:10.1002/14356007.a04_455.
- 1,2-Butanediol (PDF), SIDS Initial Assessment Report, Geneva: United Nations Environment Programme, February 1995.
- US 4596886, Hasegawa, Ryuichi & Kohji Hayashi, "Polyester containing impure 1,2-butanediol", assigned to Mitsubishi Monsanto Chemical Company.
- US 4966658, Berg, Lloyd, "Recovery of ethylene glycol from butanediol isomers by azeotropic distillation". US 5423955, Berg, Lloyd, "Separation of propylene glycol from 1,2-butanediol by azeotropic distillation".
- Wurtz, A. (1859), Ann. Chim. Phys., 55: 400CS1 maint: untitled periodical (link).
- US 5155263, Imanari, Makoto; Hiroshi Iwane & Masashi Suzuki et al., "Process for preparing α-ketobutyric acid", assigned to Mitsubishi Petrochemical Co., Ltd..
External links
- International Chemical Safety Card 03
- SIDS Initial Assessment Report for 1,2-Butanediol from the Organisation for Economic Co-operation and Development (OECD)