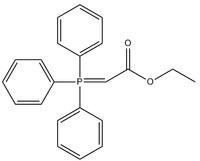
Triphenylcarbethoxymethylenephosphorane
 | |
| Names | |
|---|---|
| Other names
(2-Ethoxy-2-oxoethylidene)triphenylphosphorane (Carbethoxymethylene)triphenylphosphorane | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.012.865 |
PubChem CID |
|
| |
| |
| Properties | |
| C22H21O2P | |
| Molar mass | 348.38 g·mol−1 |
| Melting point | 124 to 129 °C (255 to 264 °F; 397 to 402 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Triphenylcarbethoxymethylenephosphorane is an organophosphorus compound with the chemical formula Ph3PCHCO2Et (Ph = phenyl, Et = ethyl). It is a white solid that is soluble in organic solvents.
The compound is a Wittig reagent. It is used to replace oxygen centres in ketones and aldehydes with CHCO2Et.[1]
References
- ↑ Lang, R. W.; Hansen, H.-J. (1984). "α-Allenic Esters from α-Phosphoranylidene Esters and Acid Chlorides: Ethyl 2,3-Pentadienoate" (PDF). Organic Syntheses. 62: 202. ; Collective Volume, 7, p. 232
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.