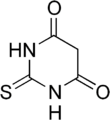
Thiobarbituric acid
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-Sulfanylidene-1,3-diazinane-4,6-dione | |||
| Other names
2-Thioxodihydropyrimidine-4,6(1H,5H)-dione 2-Thiobarbituric acid | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.007.260 | ||
| EC Number | 207-985-8 | ||
PubChem CID |
|||
| |||
| |||
| Properties | |||
| C4H4N2O2S | |||
| Molar mass | 144.15 g/mol | ||
| Melting point | 245 °C (473 °F; 518 K) | ||
| -72.9·10−6 cm3/mol | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Thiobarbituric acid is an organic compound and a heterocycle. It is used as a reagent in assaying malondialdehyde (the TBARS assay of lipid peroxidation).[1]
References
- ↑ Thiobarbituric acid reactive substances (TBARS) Assay Archived 2006-09-14 at the Wayback Machine., AMDCC Protocols, Animal Models of Diabetic Complications Consortium
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.