Tezacaftor
 | |
| Clinical data | |
|---|---|
| Trade names | Symdeko (with ivacaftor) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
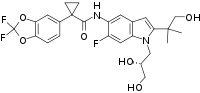
| Formula | C26H27F3N2O6 |
| Molar mass | 520.51 g·mol−1 |
Tezacaftor, also known as VX-661, is a drug approved by the FDA to treat some cases of cystic fibrosis.
In 2018, the FDA approved a combination of ivacaftor and tezacaftor; the manufacturer announced a list price of $292,000 per year.[1] The combination of ivacaftor and tezacaftor is being sold as Symdeko.[2]
Mechanism of action
Tezacaftor helps move the CFTR protein to the correct position on the cell surface, and is designed to treat people with the F508del mutation.[3]
Properties
Its chemical formula is C26H27F3N2O6, with a molecular weight of 520.505 g/mol.[4]
Clinical trials
The EVOLVE and EXPAND study findings were published in 2017.[2]
References
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.