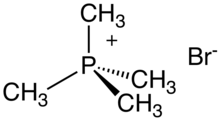
Tetramethylphosphonium bromide
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C4H12BrP | |
| Molar mass | 171.02 g·mol−1 |
| Appearance | white solid |
| Hazards | |
| GHS pictograms |  |
| GHS signal word | Warning |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+352, P304+340, P305+351+338, P312, P321, P332+313, P337+313, P362, P403+233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tetramethylphosphonium bromide is an organophosphorus compound with the formula (CH3)4PBr. It is a white, water-soluble solid, the salt of the cation tetramethylphosphonium and the bromide anion. It is prepared by treating trimethylphosphine with methyl bromide. Deprotonation gives methylenetrimethylphosphorane.[1]
References
- ↑ H. F. Klein (1978). "Trimethylphosphonium Methylide (Trimethyl Methylenephosphorane)". Inorganic Syntheses. XVIII: 138–140. doi:10.1002/9780470132494.ch23.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.