Succinylacetone
 | |
| Names | |
|---|---|
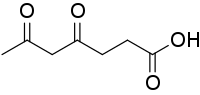
| IUPAC name
4,6-Dioxoheptanoic acid | |
| Other names
Succinyl acetone | |
| Identifiers | |
3D model (JSmol) |
|
PubChem CID |
|
| |
| |
| Properties | |
| C7H10O4 | |
| Molar mass | 158.15 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Succinylacetone is a chemical compound that is formed by the oxidation of glycine and is a precursor of methylglyoxal.[1]
References
- ↑ "Interplay Between Oxidative and Carbonyl Stresses: Molecular Mechanisms, Biological Effects and Therapeutic Strategies of Protection". Intechopen.com. Retrieved January 28, 2016.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.