Rocaglamide
 | |
| Names | |
|---|---|
| IUPAC name
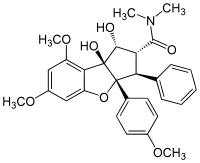
(1R,2R,3S,3aR,8bS)-1,8b-Dihydroxy-6,8-dimethoxy-3a-(4-methoxyphenyl)-N,N-dimethyl-3-phenyl-2,3-dihydro-1H-cyclopenta[b][1]benzofuran-2-carboxamide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C29H31NO7 | |
| Molar mass | 505.57 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Rocaglamide is a natural product which belongs to a class of molecules called flavaglines.[1][2] This compound was isolated in 1982 by King and colleagues based on its antileukemic activity.[3] Like other flavaglines, rocaglamide displays potent insecticidal, antifungal, anti-inflammatory and anticancer activities. Rocaglamide A (RocA) inhibits eukaryotic translation initiation by binding to the translation initiation factor eIF4A and converting it into a translational repressor.[4]
Rocaglamide was first synthesized by Barry Trost in 1990.[5] Although other syntheses have been described since, Trost’s remains the only one to afford rocaglamide in an enantio-specific manner.
See also
References
- ↑ Ebada SS, Lajkiewicz N, Porco JA, Li-Weber M, Proksch P (2011). "Chemistry and biology of rocaglamides (= flavaglines) and related derivatives from aglaia species (meliaceae)". Progress in the Chemistry of Organic Natural Products. 94: 1–58. doi:10.1007/978-3-7091-0748-5_1. ISBN 978-3-7091-0747-8. PMC 4157394. PMID 21833837.
- ↑ Ribeiro N, Thuaud F, Nebigil C, Désaubry L (March 2012). "Recent advances in the biology and chemistry of the flavaglines". Bioorganic & Medicinal Chemistry. 20 (6): 1857–64. doi:10.1016/j.bmc.2011.10.048. PMID 22071525.
- ↑ King ML, Chiang CC, Ling HC, Fujita E, Ochiai M, McPhail AT (1992). "X-Ray crystal structure of rocaglamide, a novel antileukemic 1H-cyclopenta[b]benzofuran from Aglaia elliptifolia". Chem. Commun. (20): 1150–1. doi:10.1039/c39820001150.
- ↑ Iwasaki S, Floor SN, Ingolia NT (June 2016). "Rocaglates convert DEAD-box protein eIF4A into a sequence-selective translational repressor". Nature. 534 (7608): 558–61. doi:10.1038/nature17978. PMC 4946961. PMID 27309803.
- ↑ Trost BM, Greenspan PD, Yang BV, Saulnier MG (November 1990). "An unusual oxidative cyclization. A synthesis and absolute stereochemical assignment of (−)-rocaglamide". Journal of the American Chemical Society. 112 (24): 9022–4. doi:10.1021/ja00180a081.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.