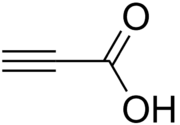
Propiolic acid
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Prop-2-ynoic acid | |
| Other names
Propiolic acid (no longer recommended[1]) Acetylene carboxylic acid Propargylic acid Acetylene mono-carboxylic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.006.763 |
| EC Number | 207-437-8 |
| KEGG | |
| MeSH | C011537 |
PubChem CID |
|
| |
| |
| Properties | |
| C3H2O2 | |
| Molar mass | 70.05 g/mol |
| Density | 1.1325 g/cm3 |
| Melting point | 9 °C (48 °F; 282 K) |
| Boiling point | 144 °C (291 °F; 417 K) (decomposes) |
| Hazards | |
| Safety data sheet | External MSDS |
EU classification (DSD) (outdated) |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Propiolic acid is the organic compound with the formula HC2CO2H. It is the simplest acetylenic carboxylic acid. It is a colourless liquid that crystallises to give silky crystals. Near its boiling point, it decomposes. It is soluble in water and possesses an odor like that of acetic acid.[2][3]
Preparation
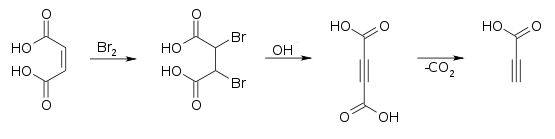
It is prepared commercially by oxidizing propargyl alcohol at a lead electrode.[4] It can also be prepared by decarboxylation of acetylenedicarboxylic acid.
Reactions and applications
Exposure to sunlight converts it into trimesic acid (benzene-1,3,5-tricarboxylic acid).[3] It undergoes bromination to give dibromoacrylic acid. With hydrogen chloride it forms chloroacrylic acid. Its ethyl ester condenses with hydrazine to form pyrazolone.[3]
It forms a characteristic explosive solid upon treatment to its aqueous solution with ammoniacal silver nitrate.[3] An amorphous explosive precipitate forms with ammoniacal cuprous chloride.
Propiolates
Propiolates are esters or salts of propiolic acid. Common examples include methyl propiolate and ethyl propiolate.
See also
References
- ↑ Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 748. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ↑ ed, Susan Budavari, (1990). The Merck index an encyclopedia of chemicals, drugs, and biologicals (11. ed., 2. print. ed.). Rahway, NJ: Merck. pp. 7833, 1911. ISBN 9780911910285.
- 1 2 3 4

- ↑ Wilhelm Riemenschneider (2002). "Carboxylic Acids, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a05_235.