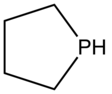
Phospholane
 | |
| Identifiers | |
|---|---|
| Properties | |
| C4H9P | |
| Molar mass | 88.09 g·mol−1 |
| Appearance | colorless liquid |
| Boiling point | 100–103 °C (212–217 °F; 373–376 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
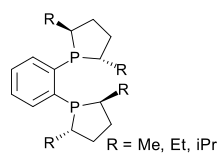
Phospholane is the organophosphorus compound with the formula (CH2)4PH. This colorless liquid is the parent member of a family of five-membered, saturated rings containing phosphorus. Although phospholane itself is only of minor academic interest, the class of C- and P-substituted phospholanes are valued ligands in asymmetric hydrogenation and related areas of homogeneous catalysis.[1] Phospholane is prepared by reduction of 1-chlorophospholane, which in turn is obtained by the reaction of 1-phenylphospholane and phosphorus trichloride.[2]
References
- ↑ Clark, Thomas; Landis, Clark (2004). "Recent developments in chiral phospholane chemistry". Tetrahedron: Asymmetry. 15: 2123–2137. doi:10.1016/j.tetasy.2004.06.025.
- ↑ K. Sommer (1970). "Zur Spaltung tertiärer Phosphine. II". Zeitschrift fuer Anorganische und Allgemeine Chemie. 379: 56–62. doi:10.1002/zaac.19703790110.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.