Oral cancer
| Oral cancer | |
|---|---|
 | |
| Oral cancer on the side of the tongue, a common site along with the floor of the mouth | |
| Specialty | Oncology, ENT, Oral and maxillofacial surgery, |
| Deaths | 146,000 (2015)[1] |
Oral cancer, also known as mouth cancer,[2] is a type of head and neck cancer and is any cancerous tissue growth located in the oral cavity.[3]
It may arise as a primary lesion originating in any of the tissues in the mouth, by metastasis from a distant site of origin, or by extension from a neighboring anatomic structure, such as the nasal cavity. Alternatively, the oral cancers may originate in any of the tissues of the mouth, and may be of varied histologic types: teratoma, adenocarcinoma derived from a major or minor salivary gland, lymphoma from tonsillar or other lymphoid tissue, or melanoma from the pigment-producing cells of the oral mucosa. There are several types of oral cancers, but around 90% are squamous cell carcinomas,[4] originating in the tissues that line the mouth and lips. Oral or mouth cancer most commonly involves the tongue. It may also occur on the floor of the mouth, cheek lining, gingiva (gums), lips, or palate (roof of the mouth). Most oral cancers look very similar under the microscope and are called squamous cell carcinoma, but less commonly other types of oral cancer occur, such as Kaposi's sarcoma.
In 2015 oral cancer resulted in 146,000 deaths up from 84,000 deaths in 1990.[1][5] Five-year survival rates in the United States are 63%.[6]
Signs and symptoms

In its early stages, it can go unnoticed. It can be painless with slight physical changes. But the precursor tissue changes can be noticed by the doctors.
Early stage symptoms can include persistent red or white patches, a non-healing ulcer, progressive swelling or enlargement, unusual surface changes, sudden tooth mobility without apparent cause, unusual oral bleeding or epitaxis and prolonged hoarseness.[7]
Late stage symptoms can include an indurated area, paresthesia or dysesthesia of the tongue or lips, airway obstruction, chronic serous otitis media, otalgia, trismus, dysphagia, cervical lymphadenopathy, persistent pain or referred pain and altered vision.[7]
Causes
Oncogenes are activated as a result of mutation of the DNA. Risk factors that predispose a person to oral cancer have been identified in epidemiological (epidemiology) studies.
Around 75 percent of oral cancers are linked to modifiable behaviors such as tobacco use and excessive alcohol consumption. Other factors include poor oral hygiene, irritation caused by ill-fitting dentures and other rough surfaces on the teeth, poor nutrition, and some chronic infections caused by fungi, bacteria or viruses.[8] If oral cancer is diagnosed in its earliest stages, treatment is generally very effective.
Chewing betel, paan and Areca is known to be a strong risk factor for developing oral cancer. In India where such practices are common, oral cancer represents up to 40% of all cancers, compared to just 4% in the UK.
Oral cancer often presents as a non-healing ulcer (shows no sign of healing after 2 weeks). In the US oral cancer accounts for about 8 percent of all malignant growths. Men are affected twice as often as women, particularly men older than 40/60.
Premalignant lesions
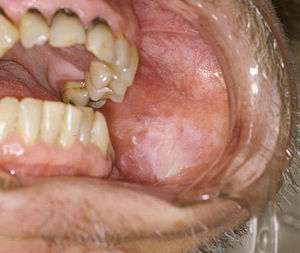
A premalignant (or precancerous) lesion is defined as "a benign, morphologically altered tissue that has a greater than normal risk of malignant transformation." There are several different types of premalignant lesion that occur in the mouth. Some oral cancers begin as white patches (leukoplakia), red patches (erythroplakia) or mixed red and white patches (erythroleukoplakia or "speckled leukoplakia"). Other common premalignant lesions include oral lichen planus (particularly the erosive type), oral submucous fibrosis and actinic cheilitis.[9] In the Indian subcontinent oral submucous fibrosis is very common. This condition is characterized by limited opening of mouth and burning sensation on eating of spicy food. This is a progressive lesion in which the opening of the mouth becomes progressively limited, and later on even normal eating becomes difficult. It occurs almost exclusively in India and Indian communities living abroad. The overall prevalence of oral potentially malignant disorders in the Middle East was 2.8%. Lichen planus/lichenoid lesions were the most common lesions (1.8%) followed by leukoplakias (0.48%), chronic hyperplastic candidiosis (0.38%), and erythroplakia (0.096%). Smoking, alcohol, and age (>40 years) were the main identifiable risk factors.[10]
Tobacco
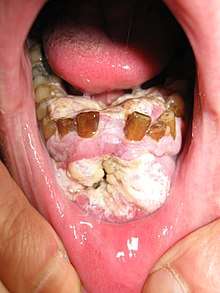
In a study of Europeans, smoking and other tobacco use was associated with about 75 percent of oral cancer cases,[11] caused by irritation of the mucous membranes of the mouth from smoke and heat of cigarettes, cigars, and pipes. Tobacco contains over 60 known carcinogens, and the combustion of it, and by-products from this process, is the primary mode of involvement. Use of chewing tobacco or snuff causes irritation from direct contact with the mucous membranes.
Tobacco use in any form by itself, and even more so in combination with heavy alcohol consumption, continues to be an important risk factor for oral cancer. However, due to the current trends in the spread of HPV16, as of early 2011 the virus is now considered the primary causative factor in 63% of newly diagnosed patients.
Alcohol
Some studies in Australia, Brazil and Germany pointed to alcohol-containing mouthwashes as also being potential causes. The claim was that constant exposure to these alcohol-containing rinses, even in the absence of smoking and drinking, leads to significant increases in the development of oral cancer. However, studies conducted in 1985,[12] 1995,[13] and 2003[14] summarize that alcohol-containing mouth rinses are not associated with oral cancer. In a March 2009 brief, the American Dental Association said "the available evidence does not support a connection between oral cancer and alcohol-containing mouthrinse".[15] A 2008 study suggests that acetaldehyde (a breakdown product of alcohol) is implicated in oral cancer,[16][17] but this study specifically focused on abusers of alcohol and made no reference to mouthwash. Any connection between oral cancer and mouthwash is tenuous without further investigation.
Human papillomavirus
Infection with human papillomavirus (HPV), particularly type 16 (there are over 180 types), is a known risk factor and independent causative factor for oral cancer.[18] A fast-growing segment of those diagnosed does not present with the historic stereotypical demographics. Historically that has been people over 50, blacks over whites 2 to 1, males over females 3 to 1, and 75% of the time people who have used tobacco products or are heavy users of alcohol. This new and rapidly growing sub population between 30 and 50 years old,[19] is predominantly nonsmoking, white, and males slightly outnumber females. Recent research from multiple peer-reviewed journal articles indicates that HPV16 is the primary risk factor in this new population of oral cancer victims. HPV16 (along with HPV18) is the same virus responsible for the vast majority of all cervical cancers and is the most common sexually transmitted infection in the US. Oral cancer in this group tends to favor the tonsil and tonsillar pillars, base of the tongue, and the oropharynx. Recent data suggest that individuals that come to the disease from this particular cause have a significant survival advantage,[20] as the disease responds better to radiation treatments than tobacco caused disease.
Stem cell transplantation
Patients after hematopoietic stem cell transplantation (HSCT) are at a higher risk for oral squamous cell carcinoma. Post-HSCT oral cancer may have more aggressive behavior with poorer prognosis, when compared to oral cancer in non-HSCT patients.[21] This effect is supposed to be owing to the continuous lifelong immune suppression and chronic oral graft-versus-host disease.[21]
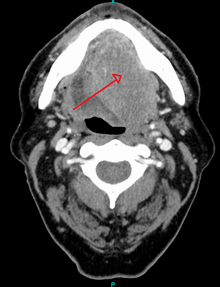
Diagnosis
_squamous_cell_carcinoma_histopathology.jpg)
Early diagnosis of oral cancer patients would decrease mortality and help to improve treatment. Oral surgeons and dentists can diagnose these patients in the early stages. Health providers, dentists, and oral surgeons shall have high knowledge and awareness that would help them to provide better diagnosis for oral cancer patients. An examination of the mouth by the health care provider, dentist, oral surgeons shows a visible and/or palpable (can be felt) lesion of the lip, tongue, or other mouth area. The lateral/ventral sides of the tongue are the most common sites for intraoral SCC. As the tumor enlarges, it may become an ulcer and bleed. Speech/talking difficulties, chewing problems, or swallowing difficulties may develop. A feeding tube is often necessary to maintain adequate nutrition. This can sometimes become permanent as eating difficulties can include the inability to swallow even a sip of water. The doctor can order some special investigations which may include a chest x-ray, CT or MRI scans, and tissue biopsy.
While a dentist, physician or other health professional may suspect a particular lesion is malignant, there is no way to tell by looking alone - since benign and malignant lesions may look identical to the eye. A non-invasive brush biopsy (BrushTest) can be performed to rule out the presence of dysplasia (pre-cancer) and cancer on areas of the mouth that exhibit an unexplained color variation or lesion. The only definitive method for determining if cancerous or precancerous cells are present is through biopsy and microscopic evaluation of the cells in the removed sample. A tissue biopsy, whether of the tongue or other oral tissues and microscopic examination of the lesion confirm the diagnosis of oral cancer or precancer.
Screening
The US Preventive Services Task Force (USPSTF) in 2013 stated evidence was insufficient to determine the balance of benefits and harms of screening for oral cancer in adults without symptoms by primary care providers.[22] The American Academy of Family Physicians comes to similar conclusions while the American Cancer Society recommends that adults over 20 years who have periodic health examinations should have the oral cavity examined for cancer.[22] The American Dental Association recommends that providers remain alert for signs of cancer during routine examinations.[22]
There are a variety of screening devices, however, there is no evidence that routine use of these devices in general dental practice is helpful.[23] However, there are compelling reasons to be concerned about the risk of harm this device may cause if routinely used in general practice. Such harms include false positives, unnecessary surgical biopsies and a financial burden on the patient.
Management

Surgical excision (removal) of the tumor is usually recommended if the tumor is small enough, and if surgery is likely to result in a functionally satisfactory result. Radiation therapy with or without chemotherapy is often used in conjunction with surgery, or as the definitive radical treatment, especially if the tumour is inoperable. Surgeries for oral cancers include:
- Maxillectomy (can be done with or without orbital exenteration)
- Mandibulectomy (removal of the mandible or lower jaw or part of it)
- Glossectomy (tongue removal, can be total, hemi or partial). When glossectomy is performed for smaller tumors (< 4 cm), the adequacy of resection (margin status) is best assessed from the resected specimen itself. The status of the margin (positive/tumor cut through versus negative/clear margin) obtained from the glossectomy specimen appears to be of prognostic value, while the status of the margin sampled from the post-glossectomy defect is not. The method of margin sampling appears to correlate with local recurrence: preference for tumor bed/defect margins may be associated with worse local control.[24][25]
- Radical neck dissection
- Mohs surgery or CCPDMA
- Combinational, e.g. glossectomy and laryngectomy done together
- Feeding tube to sustain nutrition
Owing to the vital nature of the structures in the head and neck area, surgery for larger cancers is technically demanding. Reconstructive surgery may be required to give an acceptable cosmetic and functional result. Bone grafts and surgical flaps such as the radial forearm flap are used to help rebuild the structures removed during excision of the cancer. An oral prosthesis may also be required. Most oral cancer patients depend on a feeding tube for their hydration and nutrition. Some will also get a port for the chemo to be delivered. Many oral cancer patients are disfigured and suffer from many long term after effects. The after effects often include fatigue, speech problems, trouble maintaining weight, thyroid issues, swallowing difficulties, inability to swallow, memory loss, weakness, dizziness, high frequency hearing loss and sinus damage.
Survival rates for oral cancer depend on the precise site and the stage of the cancer at diagnosis. Overall, 2011 data from the SEER database shows that survival is around 57% at five years when all stages of initial diagnosis, all genders, all ethnicities, all age groups, and all treatment modalities are considered. Survival rates for stage 1 cancers are approximately 90%, hence the emphasis on early detection to increase survival outcome for patients. Similar survival rates are reported from other countries such as Germany.[26]
Following treatment, rehabilitation may be necessary to improve movement, chewing, swallowing, and speech. Speech and language pathologists may be involved at this stage.
Chemotherapy is useful in oral cancers when used in combination with other treatment modalities such as radiation therapy. It is not used alone as a monotherapy. When a cure is unlikely, it can also be used to extend life and can be considered palliative but not curative care. Biological agents such as Cetuximab have recently been shown to be effective in the treatment of squamous cell head and neck cancers, and are likely to have an increasing role in the future management of this condition when used in conjunction with other established treatment modalities. Likewise, molecularly targeted therapies and immunotherapies maybe be effective for the treatment of oral and oropharyngeal cancers.[27] Adding epidermal growth factor receptor monoclonal antibody (EGFR mAb) to standard treatment may increase survival, keeping the cancer limited to that area of the body and may decrease reappearance of the cancer.[27]
Treatment of oral cancer will usually be by a multidisciplinary team, with treatment professionals from the realms of radiation, surgery, chemotherapy, nutrition, dentistry, and even psychology all possibly involved with diagnosis, treatment, rehabilitation, and patient care.
Prognosis
- Postoperative disfigurement of the face, head and neck
- Complications of radiation therapy, including dry mouth and difficulty swallowing
- Other metastasis (spread) of the cancer
- Significant weight loss
Prognosis depends on stage and overall health. Grading of the invasive front of the tumor is a very important prognostic parameter.[28]
Epidemiology

In 2013 oral cancer resulted in 135,000 deaths, up from 84,000 deaths in 1990.[5] Oral cancer occurs more often in people from the lower end of the socioeconomic scale.[29]
Europe
Europe places second highest after Southeast Asia among all continents for age-standardised rate (ASR) specific to oral and oropharyngeal cancer. It is estimated that there were 61,400 cases of oral and lip cancer within Europe in 2012. Hungary recorded the highest number of mortality and morbidity due to oral and pharyngeal cancer among all European countries while Cyprus reported the lowest numbers [30]
United Kingdom
British Cancer Research found 2,386 deaths due to oral cancer in 2014; other studies show this is particularly within the elder population category; only 6% of people affected by oral cancer are under 45.[31] The UK is 16th-lowest for males and 11th-highest for females for oral cancer incidence among Europe. Additionally, there is a regional variability within the UK, with Scotland and northern England having higher rates than southern England. The same analysis applies to lifetime risk of developing oral cancer, as in Scotland it is 1.84% in males and 0.74% in females, higher than the rest of the UK, being 1.06% and 0.48%, respectively.
Oral cancer is the sixteenth most common cancer in the UK (around 6,800 people were diagnosed with oral cancer in the UK in 2011), and it is the nineteenth most common cause of cancer death (around 2,100 people died from the disease in 2012).[31]
Northern Europe
The highest incidence of oral and pharyngeal cancer was recorded in Denmark, with age-standardised rates per 100,000 of 13.0, followed by Lithuania (9.9) and the United Kingdom (9.8).[30] Lithuania reported the highest incidence in men while Denmark reported the highest in women. The highest rates for mortality in 2012 were reported in Lithuania (7.5), Estonia (6.0) and Latvia (5.4).[30] The high incidence rate of oral and pharyngeal cancer in Denmark could be attributed to their higher alcohol intake than citizens of other Scandinavian countries and low intake of fruits and vegetables in general.
Eastern Europe
Hungary (23.3), Slovakia (16.4) and Romania (15.5) reported the highest incidences of oral and pharyngeal cancer.[30] Hungary also recorded the highest incidence in both genders as well as the highest mortality rates in Europe.[30] It is ranked third globally for cancer mortality rates.[32] Cigarette smoking, excessive alcohol consumption, inequalities in the care received by cancer patients and gender-specific systemic risk factors have been determined as the leading causes for the high morbidity and mortality rates in Hungary.[33][34][35][36]
Southern Europe
The incidence rates of oral cancer in western Europe found France, Germany and Belgium to be highest. The ASRs (per 100,000) were 15.0, 14.6 and 14.1, respectively. When filtered by gender category, the same countries rank top 3 for male, however, in different order of Belgium (21.9), Germany (23.1), France (23.1). France, Belgium and Netherlands ranks highest for females, with ASRs 7.6, 7.0, 7.0, respectively.[30]
Western Europe
Incidence of oral and oropharyngeal cancers were recorded, finding Portugal, Croatia and Serbia to have highest rates (ASR per 100,000). These values are 15.4, 12 and 11.7, respectively.
United States
In 2011, close to 37,000 Americans are projected to be diagnosed with oral or pharyngeal cancer. 66% of the time, these will be found as late stage three and four disease. It will cause over 8,000 deaths. Of those newly diagnosed, only slightly more than half will be alive in five years. Similar survival estimates are reported from other countries. For example, five-year relative survival for oral cavity cancer in Germany is about 55%.[26]
Oral cancers overall risk higher in black males opposed to white males, however specific oral cancers-such as of the lip, have a higher risk in white males opposed to black males. Overall, rates of oral cancer between gender groups (male and female) seem to be decreasing, according to data from 3 studies [37]
Of all the cancers, oral cancer attributes to 3% in males, opposed to 2% in women. New cases of oral cancer in US as of 2013, approximated almost 66,000 with almost 14000 attributed from tongue cancer, and nearly 12000 from the mouth, and the remainder from the oral cavity and pharynx. In the previous year, 1.6% of lip and oral cavity cancers were diagnosed, where the age-standardised incidence rate (ASIR) across all geographic regions of United States of America estimates at 5.2 per 100,000 population.[38] It is the 11th most common cancer in USA among males while in Canada and Mexico it is the 12th and 13th most common cancer respectively. The ASIR for lip and oral cavity cancer among men in Canada and Mexico is 4.2 and 3.1 respectively.[38]
South America
The ASIR across all geographic regions of South America as of 2012 sits at 3.8 per 100,000 population where approximately 6,046 deaths have occurred due to lip and oral cavity cancer, where the age-standardised mortality rate remains at 1.4.[39]
In Brazil, however, lip and oral cavity cancer is the 7th most common cancer, with an estimated 6,930 new cases diagnosed in the year 2012. This number is rising and has an overall higher ASIR at 7.2 per 100,000 population whereby an approx 3000 deaths have occurred [39]
Rates are increasing across both males and females. As of 2017, almost 50000 new cases of oropharyngeal cancers will be diagnosed, with incidence rates being more than twice as high in men than women.[39]
Asia
Oral cancer is one of the most common types of cancer in Asia due to its association with smoking (tobacco, bidi), betel quid and alcohol consumption. Regionally incidence varies with highest rates in South Asia, particularly India, Bangladesh, Sri Lanka, Pakistan and Afghanistan.[40] In South East Asia and Arab countries, although the prevalence is not as high, estimated incidences of oral cancer ranged from 1.6 to 8.6/ 100,000 and 1.8 to 2.13/ 100,000 respectively.[41] [42] According to GLOBOCAN 2012, the estimated age-standardised rates of cancer incidence and mortality was higher in males than females. However, in some area, specifically South East Asia, similar rates were recorded for both genders.[41] The average age of those diagnosed with oral sarcoma cell carcinoma is approximately 51-55.[40] In 2012, there were 97,400 deaths recorded due to oral cancer [43]
India
Oral cancer is the third most common form of cancer in India with over 77 000 new cases diagnosed in 2012 (2.3:1 male to female ratio)[44]. One of the reasons behind such high incidence might be popularity of betel and areca nuts, which are considered to be risk factors for development of oral cavity cancers[45].
Africa
There is limited data for the prevalence of oral cancer in Africa. The following rates describe the number of new cases (for incidence rates) or deaths (for mortality rates) per 100 000 individuals per year.[43]
The incidence rate of oral cancer is 2.6 for both sexes. The rate is higher in males at 3.3 and lower in females at 2.0.[43]
The mortality rate is lower than the incidence rate at 1.6 for both sexes. The rate is again higher for males at 2.1 and lower for females at 1.3.[43]
Australia
The following rates describe the number of new cases or deaths per 100 000 individuals per year. The incidence rate of oral cancer is 6.3 for both sexes; this is higher in males at 6.8-8.8 and lower in females at 3.7-3.9.[46] The mortality rate is significantly lower than the incidence rate at 1.0 for both sexes. The rate is higher in males at 1.4 and lower in females at 0.6.[47] Table 1 provides age-standardised incidence and mortality rates for oral cancer based on the location in the mouth. The location ‘other mouth’ refers to the buccal mucosa, the vestibule and other unspecified parts of the mouth. The data suggests lip cancer has the highest incidence rate while gingival cancer has the lowest rate overall. In terms of mortality rates, oropharyngeal cancer has the highest rate in males and tongue cancer has the highest rate in females. Lip, palatal and gingival cancer have the lowest mortality rates overall.[47]
| Location | Incidence per 100 000 individuals per year | Mortality per 100 000 individuals per year | ||||
|---|---|---|---|---|---|---|
| Both sexes | Males | Females | Both sexes | Males | Females | |
| Lip | 5.3 | 8.4 | 2.4 | 0.1 | 0.1 | 0.0 |
| Tongue | 2.4 | 3.3 | 1.4 | 0.7 | 1.1 | 0.4 |
| Gingivae | 0.3 | 0.4 | 0.3 | 0.1 | 0.1 | 0.0 |
| Floor of mouth | 0.9 | 1.4 | 0.5 | 0.2 | 0.3 | 0.1 |
| Palate | 0.6 | 0.7 | 0.4 | 0.1 | 0.2 | 0.1 |
| Other Mouth | 0.7 | 0.8 | 0.6 | 0.2 | 0.2 | 0.1 |
| Major salivary glands | 1.2 | 1.6 | 0.9 | 0.3 | 0.4 | 0.2 |
| Oropharynx | 1.9 | 3.0 | 0.8 | 0.7 | 1.2 | 0.3 |
See also
References
- 1 2 GBD 2015 Disease and Injury Incidence and Prevalence, Collaborators. (8 October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015". The Lancet. 388 (10053): 1545–1602. PMID 27733282.
- ↑ Lozano, Rafael; Naghavi, Mohsen; et al. (2012). "Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010". The Lancet. 380 (9859): 2095–128. doi:10.1016/S0140-6736(12)61728-0. PMID 23245604.
- ↑ Werning, John W (May 16, 2007). Oral cancer: diagnosis, management, and rehabilitation. p. 1. ISBN 978-1-58890-309-9.
- ↑ "Oral Cancer Facts - The Oral Cancer Foundation". www.oralcancerfoundation.org.
- 1 2 GBD 2013 Mortality Causes of Death Collaborators (2015). "Global, regional, and national age–sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013". The Lancet. 385 (9963): 117–71. doi:10.1016/S0140-6736(14)61682-2. PMC 4340604. PMID 25530442.
- ↑ "SEER Stat Fact Sheets: Oral Cavity and Pharynx Cancer". NCI. Retrieved 18 June 2014.
- 1 2 Ravikiran Ongole, Praveen B N, ed. (2014). Textbook of Oral Medicine, Oral Diagnosis and Oral Radiology. Elsevier India. p. 387. ISBN 978-8131230916.
- ↑ Srinivasprasad, Vijayan; Dineshshankar, Janardhanam; Sathiyajeeva, J; Karthikeyan, M; Sunitha, J; Ragunathan, Ramachandran (2015). "Liaison between micro-organisms and oral cancer". Journal of Pharmacy and Bioallied Sciences. 7 (Suppl 2): S354–60. doi:10.4103/0975-7406.163451. PMC 4606619. PMID 26538877.
- ↑ Neville BW, Damm DD, Allen CM, Bouquot JE (2002). Oral & maxillofacial pathology (2nd ed.). Philadelphia: W.B. Saunders. pp. 337, 345, 349, 353. ISBN 978-0721690032.
- ↑ Hassona, Y; Scully, C; Almangush, A; Baqain, Z; Sawair, F (2014). "Oral potentially malignant disorders among dental patients: a pilot study in Jordan". Asian Pacific Journal of Cancer Prevention : APJCP. 15 (23): 10427–31. doi:10.7314/apjcp.2014.15.23.10427. PMID 25556487.
- ↑ Rodriguez, Teresa; Altieri, Andrea; Chatenoud, Liliane; Gallus, Silvano; Bosetti, Cristina; Negri, Eva; Franceschi, Silvia; Levi, Fabio; Talamini, Renato; La Vecchia, Carlo (2004). "Risk factors for oral and pharyngeal cancer in young adults". Oral Oncology. 40 (2): 207–13. doi:10.1016/j.oraloncology.2003.08.014. PMID 14693246.
- ↑ Mashberg, Arthur; Barsa, Patrice; Grossman, Martin L. (1985). "A study of the relationship between mouthwash use and oral and pharyngeal cancer". The Journal of the American Dental Association. 110 (5): 731–4. doi:10.14219/jada.archive.1985.0422. PMID 3859544.
- ↑ Elmore, J; Horwitz, R (1995). "Oral cancer and mouthwash use: Evaluation of the epidemiologic evidence". Otolaryngology–Head and Neck Surgery. 113 (3): 253–61. doi:10.1016/S0194-5998(95)70114-1. PMID 7675486.
- ↑ Cole, Philip; Rodu, Brad; Mathisen, Annette (2003). "Alcohol-containing mouthwash and oropharyngeal cancer". The Journal of the American Dental Association. 134 (8): 1079–87. doi:10.14219/jada.archive.2003.0322. PMID 12956348.
- ↑ Science brief on alcohol-containing mouthrinses and oral cancer, Archived March 19, 2012, at the Wayback Machine. American Dental Association, March 2009
- ↑ Warnakulasuriya, Saman; Parkkila, Seppo; Nagao, Toru; Preedy, Victor R.; Pasanen, Markku; Koivisto, Heidi; Niemelä, Onni (2007). "Demonstration of ethanol-induced protein adducts in oral leukoplakia (pre-cancer) and cancer". Journal of Oral Pathology & Medicine. 37 (3): 157–65. doi:10.1111/j.1600-0714.2007.00605.x. PMID 18251940.
- ↑ Alcohol and oral cancer research breakthrough Archived May 2, 2009, at the Wayback Machine.
- ↑ Gillison ML, Chaturvedi AK, Anderson WF, Fakhry C (2015). "Epidemiology of Human Papillomavirus-Positive Head and Neck Squamous Cell Carcinoma". J Clin Oncol. 33 (29): 3235–3242. doi:10.1200/JCO.2015.61.6995.
- ↑ Martin-Hernan, F.; Sanchez-Hernandez, JG.; Cano, J.; Campo, J.; del Romero, J. (2013). "Oral cancer, HPV infection and evidence of sexual transmission". Medicina Oral Patología Oral y Cirugia Bucal. 18 (3): e439–44. doi:10.4317/medoral.18419. PMC 3668870. PMID 23524417.
- ↑ "HPV-Positive Tumor Status Indicates Better Survival in Patients with Oropharyngeal Cancer - MD Anderson Cancer Center". www.mdanderson.org.
- 1 2 Elad, Sharon; Zadik, Yehuda; Zeevi, Itai; Miyazaki, Akihiro; De Figueiredo, Maria A. Z.; Or, Reuven (2010). "Oral Cancer in Patients After Hematopoietic Stem-Cell Transplantation: Long-Term Follow-Up Suggests an Increased Risk for Recurrence". Transplantation. 90 (11): 1243–4. doi:10.1097/TP.0b013e3181f9caaa. PMID 21119507.
- 1 2 3 "Final Recommendation Statement: Oral Cancer: Screening - US Preventive Services Task Force". www.uspreventiveservicestaskforce.org. November 2013. Retrieved 23 November 2017.
- ↑ Brocklehurst, Paul; Kujan, Omar; O'Malley, Lucy A; Ogden, Graham; Shepherd, Simon; Glenny, Anne-Marie (2013). "Screening programmes for the early detection and prevention of oral cancer". Cochrane Database of Systematic Reviews (11): CD004150. doi:10.1002/14651858.CD004150.pub4. PMID 24254989.
- ↑ Varvares, MA; Poti, S; Kenyon, B; Christopher, K; Walker, RJ (October 2015). "Surgical margins and primary site resection in achieving local control in oral cancer resections". The Laryngoscope. 125 (10): 2298–307. doi:10.1002/lary.25397. PMID 26011037.
- ↑ Maxwell, JH; Thompson, LD; Brandwein-Gensler, MS; Weiss, BG; Canis, M; Purgina, B; Prabhu, AV; Lai, C; Shuai, Y; Carroll, WR; Morlandt, A; Duvvuri, U; Kim, S; Johnson, JT; Ferris, RL; Seethala, R; Chiosea, SI (1 December 2015). "Early Oral Tongue Squamous Cell Carcinoma: Sampling of Margins From Tumor Bed and Worse Local Control". JAMA Otolaryngology–Head & Neck Surgery. 141 (12): 1104–10. doi:10.1001/jamaoto.2015.1351. PMC 5242089. PMID 26225798.
- 1 2 Listl, S.; Jansen, L.; Stenzinger, A.; Freier, K.; Emrich, K.; Holleczek, B.; Katalinic, A.; Gondos, A.; Brenner, H.; GEKID Cancer Survival Working Group (2013). Scheurer, Michael, ed. "Survival of Patients with Oral Cavity Cancer in Germany". PLoS ONE. 8 (1): e53415. doi:10.1371/journal.pone.0053415. PMC 3548847. PMID 23349710.
- 1 2 Chan, Kelvin K. W.; Glenny, Anne-Marie; Weldon, Jo C.; Furness, Susan; Worthington, Helen V.; Wakeford, Helen (2015-12-01). "Interventions for the treatment of oral and oropharyngeal cancers: targeted therapy and immunotherapy". The Cochrane Database of Systematic Reviews (12): CD010341. doi:10.1002/14651858.CD010341.pub2. ISSN 1469-493X. PMID 26625332.
- ↑ Sawair, Faleh A.; Irwin, Christopher R.; Gordon, Derek J.; Leonard, Alan G.; Stephenson, Mike; Napier, Seamus S. (2003). "Invasive front grading: reliability and usefulness in the management of oral squamous cell carcinoma". Journal of Oral Pathology and Medicine. 32 (1): 1–9. doi:10.1034/j.1600-0714.2003.00060.x. PMID 12558952.
- ↑ Social inequalities in oral health: from evidence to action (PDF). 2015. p. 9. ISBN 9780952737766.
- 1 2 3 4 5 6 "Cancer of lip, oral cavity and pharynx. [online]". EUCAN. Retrieved 14 November 2017.
- 1 2 "oral cancer statistics". CancerresearchUK. Retrieved 28 October 2014.
- ↑ "World Life Expectancy". 2014.
- ↑ Diz P, Meleti M, Diniz-Freitas M, Vescovi P, Warnakulasuriya S, Johnson N, Kerr A (2017). "Oral and pharyngeal cancer in Europe". Translational Research in Oral Oncology. 2.
- ↑ Nemes JA, Redl P, Boda R, et al. (2008). "Oral cancer report from Northeastern Hungary". Pathol Oncol Res. 14 (1): 85–92. doi:10.1007/s12253-008-9021-4. PMID 18351444.
- ↑ Suba Z, Mihályi S, Takács D, Gyulai-Gaál S (2009). "Oral cancer: Morbus hungaricus in the 21st century". Fogorv Sz. 102: 63–68.
- ↑ Endre A (2006). "Hungarian national cancer control programme".
- ↑ Moore SR, Johnson NW, Pierce AM, Wilson DF (March 2000). "The epidemiology of mouth cancer: a review of global incidence". Oral Dis. 6 (2): 65–74. PMID 10702782.
- 1 2 Gupta N, Gupta R, Acharya AK, Patthi B, Goud V, Reddy S, Garg A, Singla A (December 2016). "Changing Trends in oral cancer - a global scenario". Nepal J Epidemiol. 6 (4): 613–619. doi:10.3126/nje.v6i4.17255. PMC 5506386. PMID 28804673.
- 1 2 3 "Cancer Facts & Figures 2017". America cancer society.
- 1 2 Krishna Rao SV, Mejia G, Roberts-Thomson K, Logan R (2013). "Epidemiology of oral cancer in Asia in the past decade--an update". Asian Pacific Journal of Cancer Prevention. 14 (10): 5567–77. doi:10.7314/apjcp.2013.14.10.5567.
- 1 2 Cheong, Sok Ching; Vatanasapt, Patravoot; Yi-Hsin, Yang; Zain, Rosnah B; Kerr, Alexander Ross; Johnson, Newell W (2017). "Oral cancer in South East Asia". Translational Research in Oral Oncology. 2: 2057178X1770292. doi:10.1177/2057178X17702921. ISSN 2057-178X.
- ↑ Al-Jaber A, Al-Nasser L, El-Metwally A (2016). "Epidemiology of oral cancer in Arab countries". Saudi Medical Journal. 37 (3): 249–55. doi:10.15537/smj.2016.3.11388. PMC 4800887. PMID 26905345.
- 1 2 3 4 "All Cancers (excluding non-melanoma skin cancer) Estimated Incidence, Mortality and Prevalence Worldwide in 2012". International Agency for Research on Cancer. 2012.
- ↑ Mallath, Mohandas K; Taylor, David G; Badwe, Rajendra A; Rath, Goura K; Shanta, V; Pramesh, C S; Digumarti, Raghunadharao; Sebastian, Paul; Borthakur, Bibhuti B (May 2014). "The growing burden of cancer in India: epidemiology and social context". The Lancet Oncology. 15 (6): e205–e212. doi:10.1016/s1470-2045(14)70115-9. ISSN 1470-2045.
- ↑ Chapman, Christopher H.; Garsa, Adam (2017-10-13), "Cancer of the Lip and Oral Cavity", Handbook of Evidence-Based Radiation Oncology, Springer International Publishing, pp. 193–207, doi:10.1007/978-3-319-62642-0_8, ISBN 9783319626413, retrieved 2018-09-29
- ↑ Chaturvedi AK, Anderson WF, Lortet-Tieulent J, Curado MP, Ferlay J, Franceschi S, Rosenberg PS, Bray F, Gillison ML (2013). "Worldwide Trends in Incidence Rates for Oral Cavity and Oropharyngeal Cancers". Journal of Clinical Oncology. 31 (36): 4550–4559. doi:10.1200/jco.2013.50.3870. PMC 3865341. PMID 24248688.
- 1 2 3 Farah CS, Simanovic B, Dost F (September 2014). "Oral cancer in Australia 1982-2008: a growing need for opportunistic screening and prevention". Aust Dent J. 59 (3): 349–59. doi:10.1111/adj.12198. PMID 24889757.
External links
| Classification | |
|---|---|
| External resources |
| Wikimedia Commons has media related to Oral cancer. |