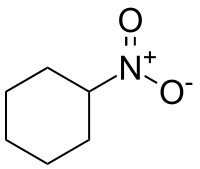
Nitrocyclohexane
 | |
| Names | |
|---|---|
| IUPAC name
Nitrocyclohexane | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.013.050 |
PubChem CID |
|
| |
| |
| Properties | |
| C6H11NO2 | |
| Molar mass | 129.16 g·mol−1 |
| Density | 1.061 g/cm3 |
| Melting point | −34 °C (−29 °F; 239 K) |
| Boiling point | 205.8 °C (402.4 °F; 478.9 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Nitrocyclohexane is an organic compound with the molecular formula C6H11NO2. It is listed as an extremely hazardous substance by the Emergency Planning and Community Right-to-Know Act, and the NOAA warns that it can be explosive.[1] It is a colorless liquid, or a pale yellow liquid.[2]
Properties
At standard temperature and pressure, it is a liquid, and has a melting point of -34 °C, and a boiling point of 205.8 °C. Its density is 1.061 g/cm3, and it is slightly soluble in water.[1][3]
Hazards
Nitrocyclohexane is highly flammable and a strong oxidizing agent.[1] It is listed as an extremely hazardous substance by the Emergency Planning and Community Right-to-Know Act, and the NOAA warns that it can be explosive.[1] It is very toxic.[4]
According to the National Institute of Health, "No reports of human poisoning cases found; Causes seizures and liver injury in animal studies; At lethal concentrations, inhalation causes excitement, somnolence, muscle contractions, and fatty liver degeneration in mice; Causes convulsions in rats after feeding lethal doses;", and it is "highly toxic by inhalation; Effects in high-dose animal studies include convulsions."[2]
References
- 1 2 3 4 "Cameo Chemicals - NITROCYCLOHEXANE". Retrieved July 9, 2012.
- 1 2 "Haz-Map - Nitrocyclohexane". Retrieved July 9, 2012.
- ↑ "Wolfram Alpha". Archived from the original on February 9, 2013. Retrieved July 9, 2012.
- ↑ "Chemical Book". Retrieved July 9, 2012.
Further reading
- "Journal of the American Chemical Society" (75). 1953: 4047. doi:10.1021/ja01112a049.
- "Synthesis of 1-Benzylnaphthalenes" (25 (6)). Journal of Organic Chemistry. 1960: 1040–1041. doi:10.1021/jo01076a605. Retrieved July 9, 2012.