Neber rearrangement
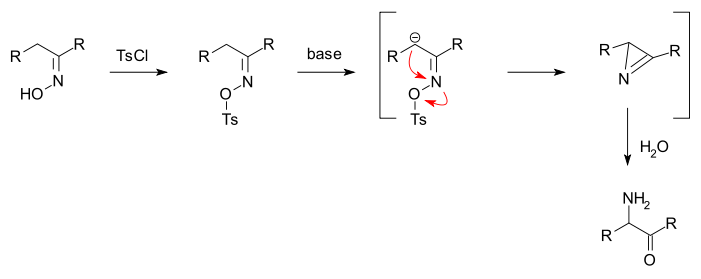
The Neber rearrangement is an organic reaction in which an ketoxime is converted into an alpha-aminoketone via a rearrangement reaction.[1][2][3]
The oxime is first converted to a O-sulfonate, for example a tosylate by reaction with tosyl chloride. Added base forms a carbanion which displaces the tosylate group in a nucleophilic displacement to an azirine and added water subsequently hydrolyses it to the aminoketone.
The Beckmann rearrangement is a side reaction.
References
- ↑ March, Jerry (1985), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (3rd ed.), New York: Wiley, ISBN 0-471-85472-7
- ↑ P. W. Neber and A. v. Friedolsheim (1926). "Über eine neue Art der Umlagerung von Oximen". Justus Liebig's Annalen der Chemie. 449 (1): 109–134. doi:10.1002/jlac.19264490108.
- ↑ O'Brien, Connor (1 April 1964). "The Rearrangement of Ketoxime O-Sulfonates to Amino Ketones (The Neber Rearrangement)". Chemical Reviews. 64 (2): 81–89. doi:10.1021/cr60228a001.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.