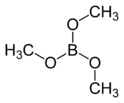
Trimethyl borate
| |||
| Names | |||
|---|---|---|---|
| Other names
trimethoxyborane, boron trimethoxide | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.004.063 | ||
| EC Number | 204-468-9 | ||
PubChem CID |
|||
| |||
| |||
| Properties | |||
| C3H9BO3 | |||
| Molar mass | 103.91 g·mol−1 | ||
| Appearance | colourless liquid | ||
| Density | 0.932 g/ml | ||
| Melting point | −34 °C (−29 °F; 239 K) | ||
| Boiling point | 68 to 69 °C (154 to 156 °F; 341 to 342 K) | ||
| decomposition | |||
| Hazards | |||
| Main hazards | flammable | ||
| Related compounds | |||
Other cations |
Trimethyl phosphite Tetramethyl orthosilicate | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Trimethyl borate is the organoboron compound with the formula B(OCH3)3. It is a colourless liquid that burns with a green flame.[1] It is an intermediate in the preparation of sodium borohydride and is a popular reagent in organic chemistry. It is a weak Lewis acid (AN = 23, Gutmann-Beckett method). [2]

Borate esters are prepared by heating boric acid or related boron oxides with alcohols under conditions where water is removed.[1]
Applications
Trimethyl borate is the main precursor to sodium borohydride by its reaction with sodium hydride:
- 4 NaH + B(OCH3)3 → NaBH4 + 3 NaOCH3
It is a gaseous anti-oxidant in brazing and solder flux. Otherwise, trimethyl borate has no announced commercial applications. It has been explored as a fire retardant, as well as being examined as an additive to some polymers.[1] It is a useful reagent in organic synthesis, as a precursor to boronic acids, which are used in Suzuki couplings. These boronic acids are prepared via reaction of the trimethyl borate with Grignard reagents followed by hydrolysis:.[3][4]
- ArMgBr + B(OCH3)3 → MgBrOCH3 + ArB(OCH3)2
- ArB(OCH3)2 + 2 H2O → ArB(OH)2 + 2 HOCH3
References
- 1 2 3 Robert J. Brotherton, C. Joseph Weber, Clarence R. Guibert, John L. Little "Boron Compounds" in Ullmann's Encyclopedia of Industrial Chemistry 2000, Wiley-VCH. doi:10.1002/14356007.a04_309
- ↑ M.A. Beckett, G.C. Strickland, J.R. Holland, and K.S. Varma, "A convenient NMR method for the measurement of Lewis acidity at boron centres: correlation of reaction rates of Lewis acid initiated epoxide polymerizations with Lewis acidity", Polymer, 1996, 37, 4629–4631. doi: 10.1016/0032-3861(96)00323-0
- ↑ Kazuaki Ishihara, Suguru Ohara, Hisashi Yamamoto (2002). "3,4,5-Trifluorophenylboronic Acid". Organic Syntheses. 79: 176. ; Collective Volume, 10, p. 80
- ↑ R. L. Kidwell, M. Murphy, and S. D. Darling (1969). "Phenols: 6-Methoxy-2-naphthol". Organic Syntheses. 49: 90. ; Collective Volume, 10, p. 80