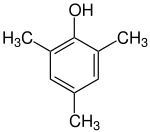
Mesitol
 | |
| Names | |
|---|---|
| IUPAC name
2,4,6-Trimethylphenol | |
| Other names
Hydroxymesitylene; Mesityl alcohol | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.007.655 |
| EC Number | 208-419-2 |
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C9H12O | |
| Molar mass | 136.19 g·mol−1 |
| Melting point | 70–72 °C (158–162 °F; 343–345 K)[1] |
| Boiling point | 220 °C (428 °F; 493 K)[1] |
| 1.01 g/l | |
| Hazards | |
| GHS pictograms | 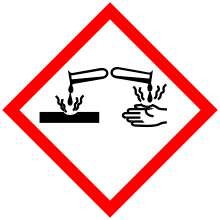  |
| GHS signal word | Danger |
| H314, H318, H411 | |
| P260, P264, P273, P280, P301+330+331, P303+361+353, P304+340, P305+351+338, P310, P321, P363, P391, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Mesitol (2,4,6-trimethylphenol) is an aromatic chemical compound having three methyl groups and one hydroxy group. The name and structure of mesitol derives from the combination of mesitylene and phenol.
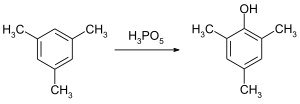
Mesitol can be obtained by reaction of mesitylene with peroxomonophosphoric acid:[2]
References
- 1 2 "2,4,6-Trimethylphenol". Sigma-Aldrich.
- ↑ Ogata, Yoshiro; Sawaki, Yasuhiko; Tomizawa, Kohtaro; Ohno, Takashi (1981). "Aromatic hydroxylation with peroxymonophosphoric acid". Tetrahedron. 37 (8): 1485. doi:10.1016/S0040-4020(01)92087-3.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.