Isotopes of magnesium
| ||||||||||||||||||||||||||||||
| Standard atomic weight (Ar, standard) |
| |||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Magnesium (12Mg) naturally occurs in three stable isotopes, 24Mg, 25Mg, and 26Mg. There are 19 radioisotopes that have been discovered, ranging from 19Mg to 40Mg. The longest-lived radioisotope is 28Mg with a half-life of 20.915 hours. The lighter isotopes mostly decay to isotopes of sodium while the heavier isotopes decay to isotopes of aluminium. The shortest-lived is 19Mg with a half-life of 4 picoseconds.
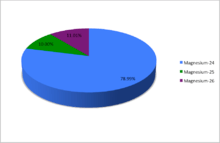
A chart showing the abundances of the naturally occurring isotopes of magnesium.
List of isotopes
| nuclide symbol |
Z(p) | N(n) | isotopic mass (u) |
half-life | decay mode(s)[2] | daughter isotope(s)[n 1] |
nuclear spin and parity |
representative isotopic composition (mole fraction) |
range of natural variation (mole fraction) |
|---|---|---|---|---|---|---|---|---|---|
| 19Mg | 12 | 7 | 19.03547(27) | 4×10−12 s | 2p | 17Ne | 1/2−# | ||
| 20Mg | 12 | 8 | 20.018863(29) | 90.8(24) ms | β+ (97%) | 20Na | 0+ | ||
| β+, p (3%) | 19Ne | ||||||||
| 21Mg | 12 | 9 | 21.011713(18) | 122(2) ms | β+ (66.9%) | 21Na | (5/2,3/2)+ | ||
| β+, p (32.6%) | 20Ne | ||||||||
| β+, α (0.5%) | 17F | ||||||||
| 22Mg | 12 | 10 | 21.9995738(14) | 3.8755(12) s | β+ | 22Na | 0+ | ||
| 23Mg | 12 | 11 | 22.9941237(14) | 11.317(11) s | β+ | 23Na | 3/2+ | ||
| 24Mg | 12 | 12 | 23.985041700(14) | Stable | 0+ | 0.7899(4) | 0.78958–0.79017 | ||
| 25Mg | 12 | 13 | 24.98583692(3) | Stable | 5/2+ | 0.1000(1) | 0.09996–0.10012 | ||
| 26Mg[n 2] | 12 | 14 | 25.982592929(30) | Stable | 0+ | 0.1101(3) | 0.10987–0.11030 | ||
| 27Mg | 12 | 15 | 26.98434059(5) | 9.458(12) min | β− | 27Al | 1/2+ | ||
| 28Mg | 12 | 16 | 27.9838768(22) | 20.915(9) h | β− | 28Al | 0+ | ||
| 29Mg | 12 | 17 | 28.988600(15) | 1.30(12) s | β− | 29Al | 3/2+ | ||
| 30Mg | 12 | 18 | 29.990434(9) | 335(17) ms | β− (99.94%) | 30Al | 0+ | ||
| β−, n (.06%) | 29Al | ||||||||
| 31Mg | 12 | 19 | 30.996546(13) | 230(20) ms | β− (98.3%) | 31Al | 1/2+ | ||
| β−, n (1.7%) | 30Al | ||||||||
| 32Mg | 12 | 20 | 31.998975(19) | 86(5) ms | β− (97.6%) | 32Al | 0+ | ||
| β−, n (2.4%) | 31Al | ||||||||
| 33Mg | 12 | 21 | 33.005254(21) | 90.5(16) ms | β− (83%) | 33Al | 3/2− | ||
| β−, n (17%) | 32Al | ||||||||
| 34Mg | 12 | 22 | 34.00946(25) | 20(10) ms | β− (>99.9%) | 34Al | 0+ | ||
| β−, n (<.1%) | 33Al | ||||||||
| 35Mg | 12 | 23 | 35.01734(43)# | 70(40) ms | β−, n (52%) | 34Al | (7/2−)# | ||
| β− (48%) | 35Al | ||||||||
| 36Mg | 12 | 24 | 36.02300(54)# | 3.9(13) ms | β− | 36Al | 0+ | ||
| 37Mg | 12 | 25 | 37.03140(97)# | 40# ms [>260 ns] | β− | 37Al | 7/2−# | ||
| β−, n | 36Al | ||||||||
| 38Mg | 12 | 26 | 38.03757(54)# | 1# ms [>260 ns] | 0+ | ||||
| 39Mg | 12 | 27 | 39.04677(55)# | <260 ns | 7/2−# | ||||
| 40Mg | 12 | 28 | 40.05393(97)# | 1# ms | 0+ | ||||
- ↑ Bold for stable isotopes
- ↑ Used in radiodating events early in the Solar System's history
Notes
- Values marked # are not purely derived from experimental data, but at least partly from systematic trends. Spins with weak assignment arguments are enclosed in parentheses.
- Uncertainties are given in concise form in parentheses after the corresponding last digits. Uncertainty values denote one standard deviation, except isotopic composition and standard atomic mass from IUPAC, which use expanded uncertainties.
References
- Isotope masses from:
- G. Audi; A. H. Wapstra; C. Thibault; J. Blachot; O. Bersillon (2003). "The NUBASE evaluation of nuclear and decay properties" (PDF). Nuclear Physics A. 729: 3–128. Bibcode:2003NuPhA.729....3A. doi:10.1016/j.nuclphysa.2003.11.001. Archived from the original (PDF) on 2008-09-23.
- Isotopic compositions and standard atomic masses from:
- J. R. de Laeter; J. K. Böhlke; P. De Bièvre; H. Hidaka; H. S. Peiser; K. J. R. Rosman; P. D. P. Taylor (2003). "Atomic weights of the elements. Review 2000 (IUPAC Technical Report)". Pure and Applied Chemistry. 75 (6): 683–800. doi:10.1351/pac200375060683.
- M. E. Wieser (2006). "Atomic weights of the elements 2005 (IUPAC Technical Report)". Pure and Applied Chemistry. 78 (11): 2051–2066. doi:10.1351/pac200678112051. Lay summary.
- Half-life, spin, and isomer data selected from the following sources. See editing notes on this article's talk page.
- G. Audi; A. H. Wapstra; C. Thibault; J. Blachot; O. Bersillon (2003). "The NUBASE evaluation of nuclear and decay properties" (PDF). Nuclear Physics A. 729: 3–128. Bibcode:2003NuPhA.729....3A. doi:10.1016/j.nuclphysa.2003.11.001. Archived from the original (PDF) on 2008-09-23.
- National Nuclear Data Center. "NuDat 2.1 database". Brookhaven National Laboratory. Retrieved September 2005. Check date values in:
|accessdate=(help) - N. E. Holden (2004). "Table of the Isotopes". In D. R. Lide. CRC Handbook of Chemistry and Physics (85th ed.). CRC Press. Section 11. ISBN 978-0-8493-0485-9.
External links
- ↑ Meija, J.; et al. (2016). "Atomic weights of the elements 2013 (IUPAC Technical Report)". Pure and Applied Chemistry. 88 (3): 265–91. doi:10.1515/pac-2015-0305.
- ↑ "Universal Nuclide Chart". nucleonica. (Registration required (help)).
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.