Iminoborane
Iminoboranes comprise a group of inorganic compounds with the generic formula XB≡NY, where X, Y can be H, F, RO, R2N, R3C, etc, among which the simplest form is HB≡NH. They are electronically related acetylenes but are usually more reactive due to the polarity.[1][2][3]
Structure and Bonding
Parent compound HB≡NH
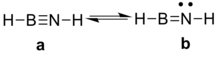
For iminoboranes, two resonance structures mainly contribute to the final structure (Figure 1).[4] For parent compound HB≡NH, the NBO(Natural Bond Orbital) analysis shows the weight of form a is 99.56% which apparently is dominant, and this means that the compound exhibits a typical triple bond character involving a dative bond from N lone pair to boron’s low lying p orbital. This is further confirmed by ELF (Electron localized function) analysis,[5] showing a torus associated with the B-N bond, which is typical for triplet bond (Figure 2).[6]
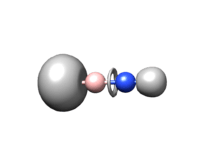
From NBO analysis, the composition of triple bonds is:
ψ π(BN),1 = 0.4694 pB + 0.8830 pN
ψ π(BN),2 = 0.4694 pB + 0.8830 pN
ψ σ(BN) = 0.5135 sp1.24(B) + 0.8581 sp0.66(N)
The integrated basin population of ELF basin shows that the population of the BN bond basin is 5.49, with the N having a contribution of 5.025 and B having a contribution of 0.489. Both NBO and ELF analysis reveal that B-N has relative stronger polarity, and for this reason, it tends to be more reactive than acetylene.[7]
Substituent effect
Because the substituents can donate their lone pairs into one of the B-N anti-bonding orbitals, the bonding scheme can be dramatically altered when substitution occurs, and the new bonding scheme highly depends on the nature of substituents and the site of substitution, which can be investigated theoretically.[8]
Substitution will change B-N bonding scheme.
Geometry Analysis
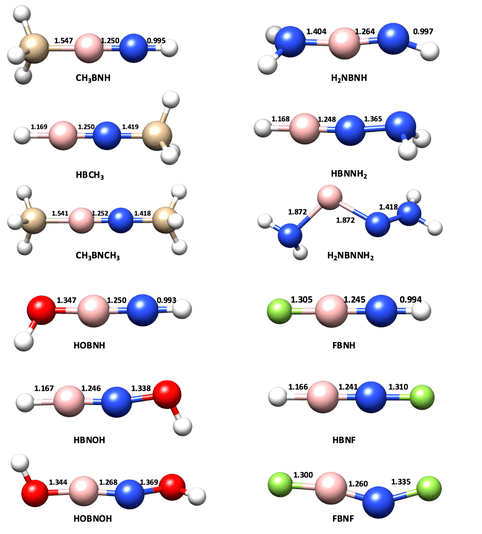
The optimized geometries of B-substituted, N-substituted and di-substituted iminoboranes are shown in Figure 3. Substitution always results in longer B-N bond, which means the weakening of B-N bond, and boron substitution can alter the bond length to greater extent than nitrogen substitution.
NBO Analysis and NRT (Natural Resonance Theory) Resonance Weightings Analysis
The contributions of three main lewis structures are listed in the following table from which we could see the effect that substitution has on iminoboranes' bonding scheme. For example, for H2NB≡NNH2, there is a strong contribution coming from resonance structure b (Figure 4), and this is in consistent with the short B-NH2 bond length, and the destroy of torus ring in ELF isosurface picture.
Table 1: Contributions from different lewis structures in iminoboranes. a, b, c correspond to three resonance structures respectively (figure 4).
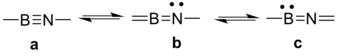
| X | XBNH | HBNX | XBNX | ||||||
| a | b | c | a | b | c | a | b | c | |
| H | 99.56 | 0.18 | 99.56 | 0.18 | 99.56 | 0.18 | |||
| CH3 | 95.85 | 3.36 | - | 94.26 | - | 4.90 | 90.59 | 3.14 | 2.86 |
| NH2 | 64.37 | 33.93 | - | 93.35 | - | 4.52 | 58.53 | 32.18 | 2.45 |
| OH | 89.16 | 10.06 | - | 93.46 | - | 5.38 | 83.93 | 10.00 | 3.79 |
| F | 90.31 | 9.20 | - | 95.00 | - | 4.46 | 85.81 | 9.85 | 3.15 |
ELF analysis
The three-dimensional representations of ELF isosurfaces for iminoboranes HBNX, XBNH, XBNX are shown in Figure 5. The integration of the basins associated with the BN group tells us the populations of each valence.

Table 2: Integrated basin populations (in e) of the relevant ELF basins associated with the BN bond in XBNY iminoboranes. “-” means there is not an individual basin here.
| X | XBNH | HBNX | XBNX | |||
| V(B,N) | V(N) | V(B,N) | V(N) | V(B,N) | V(N) | |
| H | 5.49 | - | 5.49 | - | 5.49 | - |
| CH3 | 5.58 | - | 5.70 | - | 5.73 | - |
| NH2 | 3.96 | 1.78 | 5.95 | - | 4.06 | 2.04 |
| OH | 5.69 | - | 6.08 | - | 3.84 | 2.48 |
| F | 5.67 | - | 6.12 | - | 3.58 | 2.73 |
Substitution will change the electron density distribution, atomic charge and bond polarity.
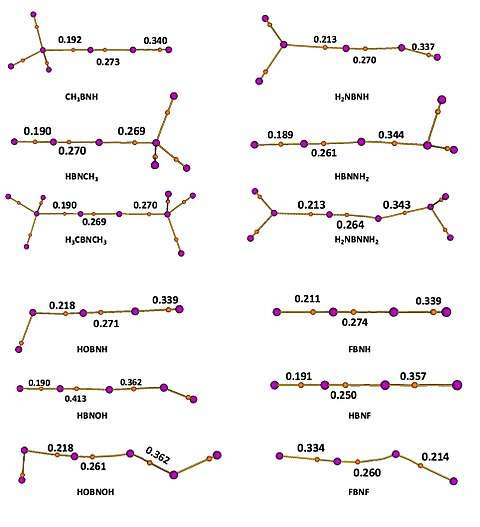
Parent compound HBNH has linear geometry and the electron density at B-N bond critical point is 0.281 (in a.u.). The molecular graphs and electron density at some relevant bond critical points are shown in Figure 6. Note that the lengthening the bond does not lead to decrease of electron density at the bond critical point. Since there is a torus area around the B-N axis, it is possible to shortening the bond length by increasing the localization of the electrons at the exterior of the torus, but depleting the electron density along the bond axis after the substitution. Under this circumstance, the electron density within the bonding region actually increases more in the periphery than decreases along the bond axis.
The bond polarity can be analyzed by atomic charge distribution. NBO charges and AIM(Atom In Molecules) charges are shown in table 3 and table 4. We could see that substitution on B further depletes the B electron density, thereby increases the bond polarity of B-N bond, while substitution on N decreases the B-N bond polarity.
Table 3: B and N net atomic charge in iminoborane derivates obtained by means of the NBO analysis
| X | XBNH | HBNX | XBNX | |||
| B | N | B | N | B | N | |
| H | +0.68 | -1.00 | +0.68 | -1.00 | +0.68 | -1.00 |
| CH3 | +0.89 | -1.04 | +0.64 | -0.79 | +0.87 | -0.81 |
| NH2 | +0.97 | -1.07 | +0.55 | -0.59 | +0.87 | -0.63 |
| OH | +1.06 | -1.10 | +0.52 | -0.45 | +0.98 | -0.57 |
| F | +1.13 | -1.11 | +0.52 | -0.31 | +1.05 | -0.44 |
Table 4: B and N net atomic charge in iminoborane derivates obtained by means of the AIM analysis
| X | XBNH | HBNX | XBNX | |||
| B | N | B | N | B | N | |
| H | +1.85 | -1.77 | +1.85 | -1.77 | +1.85 | -1.77 |
| CH3 | +1.89 | -1.79 | +1.76 | -1.39 | +1.86 | -1.81 |
| NH2 | +2.07 | -1.78 | +1.77 | -1.38 | +2.02 | -1.40 |
| OH | +2.12 | -1.81 | +1.77 | -1.17 | +2.05 | -1.20 |
| F | +2.13 | -1.81 | +1.79 | -1.01 | +2.08 | -1.03 |
Synthesis
HBNH
HBNH is an intermediate from the photolysis of solid H3BNH3[9][10][11] Strong shielding effect of bulky substitutions can make the iminoboranes more stable and more resistant to oligomerization, which allows for the storage at room temperature.[12]
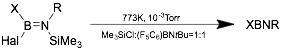
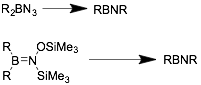
Thermal decomposition of azidoboranes and trimethyl-silyl(trimethylsilyloxy)aminoboranes can be used to access symmetric iminoboranes, especially those with bulky R groups.[13]
Reactivity
Oligomerization
Iminoboranes tend to oligomerize. Five types of oligomerization product are mainly produced: cyclodimers (1,3,2,4-diazadiboretidines, Di[14][15]), cyclotrimers (borazines, Tr), bicyclotrimers (Dewar borazines, Tr’[16]), cyclotetramers (octahydro-1,3,5,7-tetraza-2,4,6,8-trteaborocines, Te[17]), and polymers (polyiminoboranes, Po); which are shown below.[18] Which product is dominant depends on the structures of reactants and the reaction conditions. Some of the products can be interconverted.[19]
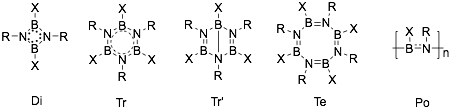
Polar Addition
Acid
If there is a π-electron donating group such as -NR2 connected to B, the neutral Lewis acids can be bonded to the N atom of the B≡N. The product shown in the following scheme is a famous diaminoboron cation [C9H18N=B=NR2]+, where the positive charge is balanced intramolecularly, not by a external anion.[20]
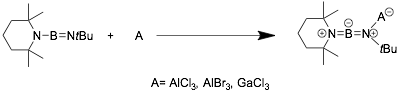
Base
The 11BNMR signals of iminoboranes can be altered when the iminoboranes are dissolved in liquid with Lewis base activity (tertiary amines, tetrahydrofuran). There may be interactions between the positive-charged B and the Lewis base, but the chemistry behind this still needs to be further investigated.[21]
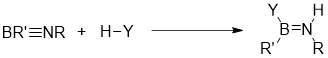
Addition of protic agents
The addition of protic agents is a quantitive reaction, fast even far below 0 ℃, while the addition of protic agents of acetylenes requires higher temperature, which also indicates that iminoboranes are more reactive towards polar addtition than acetylenes.[22]
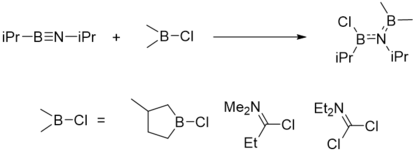
Boration
Boration reaction of iminoboranes is the addition of B-X single bond to B≡N, where -X can be -Cl (chloro-boration), -N3 (azido-boration), -SR (thio-boration), -NR2 (amino-boration) and R (alkyl-boration). One of these reactions are illustrated here.
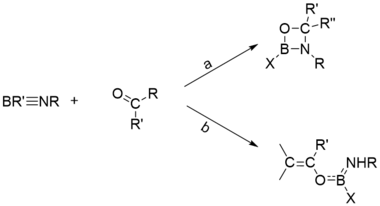
Cycloadditions
Cycloaddition is the addition of π bond to B≡N, and can be categorized into [2+2]-cycloadditions, [2+3]-cycloadditions, [2+4]-cycloadditions etc.
One of the widely investigated [2+2]-cycloadditions is the reaction of aldehydes and ketones with iminoboranes, which has two possible pathways. Usually, the relatively stable iminoboranes which are less likely to undergo the total opening of B≡N, and the ketones containing enolic protons, prefer to undergo path b.[23]
The typical [2+3]-cycloaddition is the addition of B≡N and RN3,and the typical [2+4]-cycloaddition is the addition of B≡N and cyclopentadiene.
Coordination to transition metals
Like acetylenes, iminoboranes can also coordinate with transition metals.
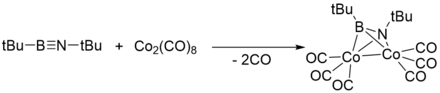
References
- ↑ Inorganic ring systems : 7th International symposium : Papers. Chivers, Tristram,. Gordon and Breach Science Publishers. 1994. ISBN 2884491686. OCLC 81135356.
- ↑ Advances in inorganic chemistry. Volume 31. Emeléus, H. J. (Harry Julius), Sharpe, A. G. New York: Academic Press. 1987. ISBN 0120236311. OCLC 277086713.
- ↑ Paetzold, Peter. Iminoboranes. pp. 123–170. doi:10.1016/s0898-8838(08)60223-8.
- ↑ Mó, Otilia; Yáñez, Manuel; Pendás, Angel Martín; Bene, Janet E. Del; Alkorta, Ibon; Elguero, José (2007-07-23). "Unusual substituent effects on the bonding of iminoboranes". Phys. Chem. Chem. Phys. 9 (30): 3970–3977. doi:10.1039/b702480k. ISSN 1463-9084.
- ↑ "Chemical Bonding Analysis in Direct and Momentum Space - Electron Localizability". www2.cpfs.mpg.de. Retrieved 2017-12-17.
- ↑ "Chemical Bonding Analysis in Direct and Momentum Space - Electron Localizability". www2.cpfs.mpg.de. Retrieved 2017-12-17.
- ↑ Mó, Otilia; Yáñez, Manuel; Pendás, Angel Martín; Bene, Janet E. Del; Alkorta, Ibon; Elguero, José (2007-07-23). "Unusual substituent effects on the bonding of iminoboranes". Phys. Chem. Chem. Phys. 9 (30): 3970–3977. doi:10.1039/b702480k. ISSN 1463-9084.
- ↑ Mó, Otilia; Yáñez, Manuel; Pendás, Angel Martín; Bene, Janet E. Del; Alkorta, Ibon; Elguero, José (2007-07-23). "Unusual substituent effects on the bonding of iminoboranes". Phys. Chem. Chem. Phys. 9 (30): 3970–3977. doi:10.1039/b702480k. ISSN 1463-9084.
- ↑ Lory, Earl R.; Porter, Richard F. (1973-03-01). "Infrared studies of matrix isolated species in the hydrogen-boron-nitrogen system". Journal of the American Chemical Society. 95 (6): 1766–1770. doi:10.1021/ja00787a012. ISSN 0002-7863.
- ↑ Paetzold, Peter; Richter, Anette; Thijssen, Theo; Würtenberg, Stefan (1979-12-01). "Bildung, Struktur und Reaktivität von (Pentafluorphenyl)bor-tert-butylimid und seinem Cyclodimeren". Chemische Berichte. 112 (12): 3811–3827. doi:10.1002/cber.19791121207. ISSN 1099-0682.
- ↑ Paetzold, Peter; von Plotho, Christoph (1982-08-01). "Über weitere monomere Borimide und ihre Reaktionen". Chemische Berichte. 115 (8): 2819–2825. doi:10.1002/cber.19821150813. ISSN 1099-0682.
- ↑ Haase, Martin; Klingebiel, Uwe (1985-04-01). "Simple Synthesis of Stable Iminoboranes". Angewandte Chemie International Edition in English. 24 (4): 324–324. doi:10.1002/anie.198503241. ISSN 1521-3773.
- ↑ Weiß, Robert; Wagner, Klaus-Georg (1984-05-01). "Notizen. Die Erzeugung von Nitrosylsalzen in wasserfreien organischen Medien". Chemische Berichte. 117 (5): 1973–1976. doi:10.1002/cber.19841170526. ISSN 1099-0682.
- ↑ Paetzold, Peter; Eleftheriadis, Eleftherios; Minkwitz, Rolf; Wölfel, Volker; Gleiter, Rolf; Bischof, Peter; Friedrich, Gert (1988-01-01). "Bildung, Struktur und Reaktionen von Methyl(methylimino)boran". Chemische Berichte. 121 (1): 61–66. doi:10.1002/cber.19881210110. ISSN 1099-0682.
- ↑ Hess, H. (1969-11-15). "Strukturbestimmungen an Bor–Stickstoff-Verbindungen. IV. Die Kristall- und Molekularstruktur von Hexakis(trimethylsilyl)-2,4-diamino-1,3,2,4-diazadiboretidin". Acta Crystallographica Section B (in German). 25 (11): 2342–2349. doi:10.1107/s056774086900567x. ISSN 0567-7408.
- ↑ Steuer, Holger-A.; Meller, Anton; Elter, Gernot. "B-t-butyl-borazine und -diazadiboretidine". Journal of Organometallic Chemistry. 295 (1): 1–6. doi:10.1016/0022-328x(85)88065-7.
- ↑ Turner H. S. and Warne R. J. 1962 Proc. Chem. Soc. 69.
- ↑ Advances in inorganic chemistry. Volume 31. Emeléus, H. J. (Harry Julius), Sharpe, A. G. New York: Academic Press. 1987. ISBN 0120236311. OCLC 277086713.
- ↑ Maier.G. (1978). "Tetra-tert-butyltetrahedrane". Angew. Chem. Int. Ed. Engl. 17: 520.
- ↑ Nöth, Heinrich; Weber, Siegfried (1985-06-01). "Beiträge zur Chemie des Bors, 158. Addukte von Aluminium- und Galliumhalogeniden an ein Aminoiminoboran". Chemische Berichte. 118 (6): 2554–2556. doi:10.1002/cber.19851180631. ISSN 1099-0682.
- ↑ Advances in inorganic chemistry. Volume 31. Emeléus, H. J. (Harry Julius), Sharpe, A. G. New York: Academic Press. 1987. ISBN 0120236311. OCLC 277086713.
- ↑ Nöth, Heinrich; Weber, Siegfried (1985-05-01). "Beiträge zur Chemie des Bors, 154. Addition von Trimethylsily-Verbindungen und von anderen Elektrophilen an (tert-Butylimino) (tetramethylpiperidino)boran". Chemische Berichte. 118 (5): 2144–2146. doi:10.1002/cber.19851180536. ISSN 1099-0682.
- ↑ von Kutepow, Nikolaus (1972-04-01). "Chemistry of acetylenes. Von H. G. Viehe. Marcel Dekker Inc., New York 1969. 1. Aufl., XV, 1298 S., zahlr. Tab. u. Formeln, geb. $ 59.50". Angewandte Chemie. 84 (8): 367–367. doi:10.1002/ange.19720840843. ISSN 1521-3757.