CED9 (gene)
| CED-9 (gene) | |
|---|---|
| Identifiers | |
| Organism | |
| Symbol | CED-9 |
| Entrez | 3565776 |
| HomoloGene | 80344 |
| PDB | 1OHU |
| RefSeq (mRNA) | NM_066883 |
| RefSeq (Prot) | NP_499284 |
| UniProt | P41958 |
| Other data | |
| Chromosome | III: 10.34 - 10.34 Mb |
Cell death abnormality gene 9 (CED-9), also known as apoptosis regulator CED-9, is a gene found in Caenorhabditis elegans that inhibits/represses programmed cell death (apoptosis).[1] The gene was discovered while searching for mutations in the apoptotic pathway after the discovery of the apoptosis promoting genes CED-3 and CED-4.[2] The gene gives rise to the apoptosis regulator CED-9 protein found as a Integral membrane protein in the mitochondrial membrane.[3] The protein is homologous to the human apoptotic regulator Bcl-2 as well as all other proteins in the Bcl-2 protein family.[4] CED-9 is involved in the inhibition of CED-4 which is the activator of the CED-3 caspase.[5] Because of the pathway homology with humans as well as the specific protein homology, CED-9 has been used to represent the human cell apoptosis interactions of Bcl-2 in research.[6]
Discovery
The CED-9 gene was discovered in 1992 while searching the genome of C. elegans for mutations affecting cell death.[2] The first mutation identified was a dominant gain of function mutation referred to as n1950 that allowed cells to survive when they were fated to die.[2] The observed phenotype was similar to that observed in CED-3 and CED-4 loss of function mutants (known proteins from the apoptotic pathway).[2] It was also observed that loss of function mutations in CED-3 and CED-4 were able to rescue cells with a CED-9 loss of function mutation.[7] These observations suggested that CED-9 functioned upstream of the CED-3 and CED-4 proteins in the same pathway.[7]
Structure
Gene
The CED-9 gene is located on chromosome 3 of the C. elegans genome.[1] CED-9 is transcribed from a polycistronic locus that also contains genes required for the mitochondrial Oxidative phosphorylation.[4] The CED-9 gene has been identified in two distinct transcripts, both transcribed from this locus.[4] The first was identified as a 1.3 kb transcript encoding only the CED-9 sequence. The second being a rare 2.1 kb bicistronic transcript containing the 1.3 kb transcript and an additional 0.75 kb transcript from an upstream gene found in the gene locus.[4] this 0.75 kb transcript corresponds to the cytochrome protein cyt-1 that functions in the electron transport chain within the mitochondria.[4] The bicistronic transcript is then spliced giving rise to the two distinct mature messenger RNA (mRNA) for both genes.[4] The most prevalent transcript however, is the 1299-nucleotide (~1.3kb) long transcript that encodes an 843-nucleotide mRNA containing 4 exons.[8]
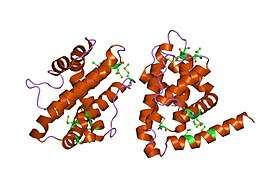
Protein
CED-9 encodes the apoptosis regulator CED-9 protein which is an important negative regulator protein in the apoptosis pathway of C. elegans.[9] The protein consists of 280 amino acids and has a molecular weight of 31824.42 Da.[10] The structure of this protein has been solved using X-ray diffraction revealing 9 Helices, 2 Beta strands, and 2 turn motifs.[3] The CED-9 protein belongs to the Bcl-2-like protein family. This refers to the homology between the ced-9 protein and the B-cell lymphoma proteins (Bcl) found in humans, specifically the Bcl-2 protein.[4] CED-9 contains a BCL domain homologous to Bcl-2 domains BH1, BH2, and part of BH3 as well as a separate domain homologous to BH4 located near the N-terminus.[11] CED-9 also includes a transmembrane domain on the C-terminal end of the structure that anchors the protein to the mitochondrial membrane.[12] However, research shown that the C-terminal domain is not necessary for the protein's main function as an inhibitor of the CED-4 protein found in the same apoptosis signalling pathway.[12]
Function
Cell death, or apoptosis during early development is crucial for the correct morphology and refractivity of adult C. elegans.[13] This process involves a signal and interaction cascade of proteins leading to the engulfment and death of the targeted cell. Proteins in this cascade can be categorized into two groups; pro-apoptotic and anti-apoptotic.[13] Pro-apoptotic proteins activate the apoptosis pathway while anti-apoptotic proteins suppress the pathway.[14] CED-9 is classified as an anti-apoptotic protein.[15]
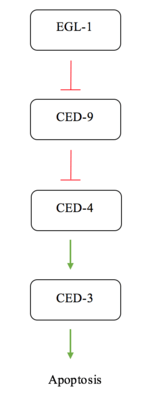
Apoptosis in C. elegans is often simplified to interactions between four major proteins in the pathway; EGL-1, CED-9, CED-4, and CED-3. CED-3 is the final protease in the interaction network and is responsible for activating the proteins involved in cell disassembly.[13] CED-9 is said to protect cells from the apoptosis pathway.[7] Under normal conditions, in a cell not experiencing apoptotic signalling, CED-9 forms a complex with CED-4 at the membrane of the mitochondria.[14] This interaction sequesters the pro-apoptotic signalling of CED-4. CED-4 consists of an asymmetric dimer of CED-4a and CED-4b proteins in which CED-9 can specifically recognize and bind CED-4a.[14] This interaction is a highly specific recognition and binding interaction between the N-terminal tails of both proteins.[14] When the cell receives an apoptotic signal via a receptor commonly referred to as a "death receptor", the protein EGL-1 is activated.[5] The active EGL-1 binds CED-9 causing a Conformational change that interrupts and inhibits the CED-9 - CED-4 interaction.[14] CED-4 is free to dissociate and activate the CED-3 protease effectively triggering the final stages for apoptosis.[5]
Mutations
The cells developed during embryogenesis and early life in C. elegans have one of two fates, to live and differentiate or apoptose.[16] Apoptosis during development is highly regulated and only occurs in specific cells at specific times.[16] Every cell division and cell death in the development of C. elegans from embryo to adult has been studied and documented to reveal a fixed pattern between individual organisms.[16] Apoptosis during development is important for the proper morphology and refractivity of C. elegans, but it is not always essential for survival.[16] Thus, over 100 mutations have been observed and documented as affecting the apoptotic pathway of C. elegans.[7] Many proteins involved in the interaction cascade were discovered because of these mutations and their resulting phenotype. CED-9 mutants are among the mutations that affect this pathway. CED-9 gain of function mutations are unresponsive to apoptosis signalling and allow cells fated to die, to survive.[7] A notable example of a CED-9 dominant gain of function mutation would be the n1950 mutation which was the first mutation documented for CED-9 and responsible for the gene's discovery.[2] Loss of function mutations cause inappropriate cell death in the absence of apoptosis stimuli.[7] Mutations in CED-9 also reveal its maternal effect; where the genotype of the mother determines the phenotype of the progeny.[7] Homozygous, loss of function mutants from a heterozygous mother experience some unpredictable cell death, however, give rise to unviable progeny themselves.[7]
Significance
The apoptotic pathway has been conserved in evolutionary history and is vital for the maintenance of multicellular organisms such as humans.[17] A parallel pathway to the one found in C. elegans is also observed in mammals involving a number of homologous proteins. Disruptions to this pathway often lead to diseases that, in humans, include various cancers, autoimmune diseases, and neurodegenerative disease.[17] Bcl-2 in particular is often found mutated in many human cancers.[6] Due to the conserved nature of the apoptotic pathway and the extensive knowledge and understanding available for C. elegans, the organisms apoptotic pathway can be used as a proxy for the human equivalent.[17] CED-9 is the homologue of Bcl-2 which can provide researchers with information including the pathways the protein is involved in and the consequences of mutation that may parallel pathways or abnormalities in humans.[6]
Interactions
References
- 1 2 "ced-9 Apoptosis regulator ced-9 [Caenorhabditis elegans] - Gene - NCBI". www.ncbi.nlm.nih.gov. Retrieved 2017-11-16.
- 1 2 3 4 5 Hengartner MO, Ellis RE, Horvitz HR (April 1992). "Caenorhabditis elegans gene ced-9 protects cells from programmed cell death". Nature. 356 (6369): 494–9. doi:10.1038/356494a0. PMID 1560823.
- 1 2 "ced-9 - Apoptosis regulator ced-9 - Caenorhabditis elegans - ced-9 gene & protein". www.uniprot.org. Retrieved 2017-11-16.
- 1 2 3 4 5 6 7 Hengartner, Michael O.; Horvitz, H. Robert (1994-02-25). "C. elegans cell survival gene ced-9 encodes a functional homolog of the mammalian proto-oncogene bcl-2". Cell. 76 (4): 665–676. doi:10.1016/0092-8674(94)90506-1.
- 1 2 3 del Peso L, González VM, Núñez G (December 1998). "Caenorhabditis elegans EGL-1 disrupts the interaction of CED-9 with CED-4 and promotes CED-3 activation". The Journal of Biological Chemistry. 273 (50): 33495–500. doi:10.1074/jbc.273.50.33495. PMID 9837929.
- 1 2 3 Arvanitis M, Li DD, Lee K, Mylonakis E (2013-10-18). "Apoptosis in C. elegans: lessons for cancer and immunity". Frontiers in Cellular and Infection Microbiology. 3: 67. doi:10.3389/fcimb.2013.00067. PMC 3798828. PMID 24151577.
- 1 2 3 4 5 6 7 8 Riddle, Donald L.; Blumenthal, Thomas; Meyer, Barbara J.; Priess, James R. (1997). Genetics of Programmed Cell Death. Cold Spring Harbor Laboratory Press.
- 1 2 3 "ced-9". Wormbase.
- ↑ "Family: BH4 (PF02180)". pfam.xfam.org. Retrieved 2017-11-16.
- ↑ "ExPASy". web.expasy.org. Retrieved 2017-11-16.
- ↑ "SMART: Sequence analysis results for CED9_CAEEL". smart.embl.de. Retrieved 2017-11-16.
- 1 2 Tan FJ, Fire AZ, Hill RB (November 2007). "Regulation of apoptosis by C. elegans CED-9 in the absence of the C-terminal transmembrane domain". Cell Death and Differentiation. 14 (11): 1925–35. doi:10.1038/sj.cdd.4402215. PMC 3047747. PMID 17703231.
- 1 2 3 Conradt B, Wu YC, Xue D (August 2016). "Programmed Cell Death During Caenorhabditis elegans Development". Genetics. 203 (4): 1533–62. doi:10.1534/genetics.115.186247. PMC 4981262. PMID 27516615.
- 1 2 3 4 5 Yan N, Chai J, Lee ES, Gu L, Liu Q, He J, Wu JW, Kokel D, Li H, Hao Q, Xue D, Shi Y (October 2005). "Structure of the CED-4-CED-9 complex provides insights into programmed cell death in Caenorhabditis elegans". Nature. 437 (7060): 831–7. doi:10.1038/nature04002. PMID 16208361.
- ↑ Jarpe MB, Widmann C, Knall C, Schlesinger TK, Gibson S, Yujiri T, Fanger GR, Gelfand EW, Johnson GL (September 1998). "Anti-apoptotic versus pro-apoptotic signal transduction: checkpoints and stop signs along the road to death". Oncogene. 17 (11 Reviews): 1475–82. doi:10.1038/sj.onc.1202183. PMID 9779994.
- 1 2 3 4 Hedgecock EM, Sulston JE, Thomson JN (June 1983). "Mutations affecting programmed cell deaths in the nematode Caenorhabditis elegans". Science. 220 (4603): 1277–9. doi:10.1126/science.6857247. PMID 6857247.
- 1 2 3 Rudin CM, Thompson CB (1997). "Apoptosis and disease: regulation and clinical relevance of programmed cell death". Annual Review of Medicine. 48: 267–81. doi:10.1146/annurev.med.48.1.267. PMID 9046961.
- 1 2 3 Lab, Mike Tyers. "ced-9 (T07C4.8) Result Summary | BioGRID". thebiogrid.org. Retrieved 2017-11-29.