Isotopes of boron
10B content may be as low as 19.1% and as high as 20.3% in natural samples. 11B is the remainder in such cases.[2] | ||||||||||||||||||||||||
| Standard atomic weight (Ar, standard) |
| |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Boron (5B) naturally occurs as isotopes 10B and 11B, the latter of which makes up about 80% of natural boron. There are 14 radioisotopes that have been discovered, with mass numbers from 6 to 21, all with short half-lives, the longest being that of 8B, with a half-life of only 770 milliseconds (ms) and 12B with a half-life of 20.2 ms. All other isotopes have half-lives shorter than 17.35 ms. Those isotopes with mass below 10 decay into helium (via short-lived isotopes of beryllium for 7B and 9B) while those with mass above 11 mostly become carbon.
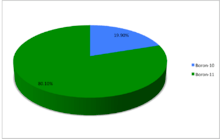
A chart showing the abundances of the naturally occurring isotopes of boron.
List of isotopes
| nuclide symbol |
Z(p) | N(n) | isotopic mass (u) |
half-life [resonance width] |
decay mode(s)[4] | daughter isotope(s) |
nuclear spin and parity |
representative isotopic composition (mole fraction) |
range of natural variation (mole percent) |
|---|---|---|---|---|---|---|---|---|---|
| 6B | 5 | 1 | 6.04681(75)# | ||||||
| 7B | 5 | 2 | 7.02992(8) | 350(50) × 10−24 s [1.4(2) MeV] |
p | 6 Be [n 1] |
(3/2−) | ||
| 8B[n 2] | 5 | 3 | 8.0246072(11) | 770(3) ms | β+, α | 2 4 He |
2+ | ||
| 9B | 5 | 4 | 9.0133288(11) | 80(30) × 10−21 s [0.54(21) keV] |
p, α | 8 Be [n 3] |
3⁄2− | ||
| 10B | 5 | 5 | 10.0129370(4) | Stable | 3+ | 0.199(7) | 18.929–20.386 | ||
| 11B | 5 | 6 | 11.0093054(4) | Stable | 3/2− | 0.801(7) | 79.614–81.071 | ||
| 12B | 5 | 7 | 12.0143521(15) | 20.20(2) ms | β− (98.4%) | 12 C |
1+ | ||
| β−, α (1.6%) | 8 Be [n 4] | ||||||||
| 13B | 5 | 8 | 13.0177802(12) | 17.33(17) ms | β− (99.72%) | 13 C |
3/2− | ||
| β−, n (0.279%) | 12 C | ||||||||
| 14B | 5 | 9 | 14.025404(23) | 12.5(5) ms | β− (93.96%) | 14 C |
2− | ||
| β−, n (6.04%) | 13 C | ||||||||
| 15B | 5 | 10 | 15.031103(24) | 9.87(7) ms | β−, n (93.6%) | 14 C |
3/2− | ||
| β− (6.0%) | 15 C | ||||||||
| β−, 2n (0.40%) | 13 C | ||||||||
| 16B | 5 | 11 | 16.03981(6) | < 190 × 10−12 s [< 0.1 MeV] |
n | 15 B |
0− | ||
| 17B[n 5] | 5 | 12 | 17.04699(18) | 5.08(5) ms | β−, n (63.0%) | 16 C |
(3/2−) | ||
| β− (22.1%) | 17 C | ||||||||
| β−, 2n (11.0%) | 15 C | ||||||||
| β−, 3n (3.5%) | 14 C | ||||||||
| β−, 4n (0.40%) | 13 C | ||||||||
| 18B | 5 | 13 | 18.05617(86)# | < 26 ns | n | 17 B |
(4−)# | ||
| 19B[n 5] | 5 | 14 | 19.06373(43)# | 2.92(13) ms | β− | 19 C |
(3/2−)# | ||
Notes
- The precision of the isotope abundances and atomic mass is limited through variations. The given ranges should be applicable to any normal terrestrial material.
- Commercially available materials may have been subjected to an undisclosed or inadvertent isotopic fractionation. Substantial deviations from the given mass and composition can occur.
- Values marked # are not purely derived from experimental data, but at least partly from systematic trends. Spins with weak assignment arguments are enclosed in parentheses.
- Uncertainties are given in concise form in parentheses after the corresponding last digits. Uncertainty values denote one standard deviation, except isotopic composition and standard atomic mass from IUPAC, which use folical uncertainties.[5]
- Nuclide masses are given by IUPAP Commission on Symbols, Units, Nomenclature, Atomic Masses and Fundamental Constants (SUNAMCO).
- Isotope abundances are given by IUPAC Commission on Isotopic Abundances and Atomic Weights (CIAAW).
- Neutrinos from Boron-8 beta decays within the sun are an important background to dark matter direct detection experiments.[6] They are the first component of the neutrino floor that dark matter direct detection experiments are expected to eventually encounter.
Applications
Boron-10
Boron-10 is used in boron neutron capture therapy (BNCT) as an experimental treatment of some brain cancers.
References
Notes
- 1 2 "Atomic Weights and Isotopic Compositions for All Elements". National Institute of Standards and Technology. Retrieved 2008-09-21.
- ↑ Szegedi, S.; Váradi, M.; Buczkó, Cs. M.; Várnagy, M.; Sztaricskai, T. (1990). "Determination of boron in glass by neutron transmission method". Journal of Radioanalytical and Nuclear Chemistry Letters. 146 (3): 177. doi:10.1007/BF02165219.
- ↑ Meija, J.; et al. (2016). "Atomic weights of the elements 2013 (IUPAC Technical Report)". Pure and Applied Chemistry. 88 (3): 265–91. doi:10.1515/pac-2015-0305.
- ↑ "Universal Nuclide Chart". nucleonica. (Registration required (help)).
- ↑ "2.5.7. Standard and expanded uncertainties". Engineering Statistics Handbook. Retrieved 2010-09-16.
- ↑ Cerdeno, David G.; Fairbairn, Malcolm; Jubb, Thomas; Machado, Pedro; Vincent, Aaron C.; Boehm, Celine (2016). "Physics from solar neutrinos in dark matter direct detection experiments". JHEP. 2016 (05): 118. arXiv:1604.01025. Bibcode:2016JHEP...05..118C. doi:10.1007/JHEP05(2016)118.
General references
- Isotope masses from:
- G. Audi; A. H. Wapstra; C. Thibault; J. Blachot; O. Bersillon (2003). "The NUBASE evaluation of nuclear and decay properties" (PDF). Nuclear Physics A. 729: 3–128. Bibcode:2003NuPhA.729....3A. doi:10.1016/j.nuclphysa.2003.11.001. Archived from the original (PDF) on 2008-09-23.
- Isotopic compositions and standard atomic masses from:
- J. R. de Laeter; J. K. Böhlke; P. De Bièvre; H. Hidaka; H. S. Peiser; K. J. R. Rosman; P. D. P. Taylor (2003). "Atomic weights of the elements. Review 2000 (IUPAC Technical Report)". Pure and Applied Chemistry. 75 (6): 683–800. doi:10.1351/pac200375060683.
- M. E. Wieser (2006). "Atomic weights of the elements 2005 (IUPAC Technical Report)". Pure and Applied Chemistry. 78 (11): 2051–2066. doi:10.1351/pac200678112051. Lay summary.
- Half-life, spin, and isomer data selected from the following sources. See editing notes on this article's talk page.
- G. Audi; A. H. Wapstra; C. Thibault; J. Blachot; O. Bersillon (2003). "The NUBASE evaluation of nuclear and decay properties" (PDF). Nuclear Physics A. 729: 3–128. Bibcode:2003NuPhA.729....3A. doi:10.1016/j.nuclphysa.2003.11.001. Archived from the original (PDF) on 2008-09-23.
- National Nuclear Data Center. "NuDat 2.1 database". Brookhaven National Laboratory. Retrieved 23 February 2017.
- N. E. Holden (2004). "Table of the Isotopes". In D. R. Lide. CRC Handbook of Chemistry and Physics (85th ed.). CRC Press. Section 11. ISBN 978-0-8493-0485-9.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.