Beryllium-8
 | |
| General | |
|---|---|
| Name, symbol | Beryllium-8,8Be |
| Neutrons | 4 |
| Protons | 4 |
| Nuclide data | |
| Natural abundance | Trace |
| Half-life | 6.7(17)×10−17 s |
| Decay products | 4He |
| Isotope mass | 8.00530510(4) u |
| Spin | 0 |
| Decay modes | |
| Decay mode | Decay energy (MeV) |
| α | 6.8(17)×10−6 |
| Complete table of nuclides | |
Beryllium-8 is an isotope of beryllium with 4 neutrons and 4 protons, and four electrons when its oxidation state is 0. It is one of the radionuclides.
Formation
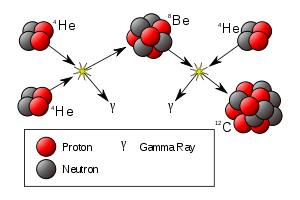
In stellar nucleosynthesis, two helium-4 nuclei may collide and fuse into a single beryllium-8 nucleus. Beryllium-8 has an extremely short half-life (6.7(17)×10−17 seconds), and decays back into two helium-4 nuclei. If the beryllium-8 collides with a helium-4 nucleus before decaying, they can fuse into a carbon-12 nuclei. This is called the triple-alpha process, or "helium burning".[1]
Theory
George Gamow, in his Big Bang theory, predicted that Be-8 decays too fast to sustain nuclear fusion. This would mean that only Hydrogen, Helium and Lithium would be produced and not the elements which have a higher mass.[2] Fred Hoyle subsequently theorized the Triple-alpha process which would allow for higher elements to be created in stars.
References
| Lighter: Beryllium-7 |
Beryllium-8 is an isotope of beryllium |
Heavier: Beryllium-9 |
| Decay product of: Carbon-9 (β+, p) boron-9 (p, α) |
Decay chain of beryllium-8 |
Decays to: helium-4 |