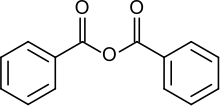
Benzoic anhydride
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Benzoic anhydride | |
| Other names
Benzoic acid anhydride Benzoyl anhydride Benzoyl benzoate | |
| Identifiers | |
3D model (JSmol) |
|
| 516726 | |
| ChemSpider | |
| ECHA InfoCard | 100.002.084 |
| EC Number | 202-291-1 |
PubChem CID |
|
| |
| |
| Properties | |
| C 14H 10O 3 | |
| Molar mass | 226.23 g mol−1 |
| Appearance | White to off-white solid |
| Density | 1.1989 g cm−3 at 15 °C |
| Melting point | 42 °C (108 °F; 315 K) |
| Boiling point | 360 °C (680 °F; 633 K) |
| -124.9·10−6 cm3/mol | |
| Hazards | |
| NFPA 704 | |
| Flash point | 113[2] °C (235 °F; 386 K) |
| Related compounds | |
Related compounds |
Benzoic acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Benzoic anhydride is the acid anhydride of benzoic acid. It is the simplest symmetrical aromatic acid anhydride. It can be produced by dehydrating benzoic acid. It provides a convenient way to prepare benzoic esters, which are more stable to hydrolysis than acetic esters and thus efficient at protecting alcohol function groups in a molecule.[3]
References
- ↑ "Sciencelab msds".
- ↑ "aldrich product page".
- ↑ Organic Synthesis. Academic press. p. 605.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.
