Antibody-drug conjugate
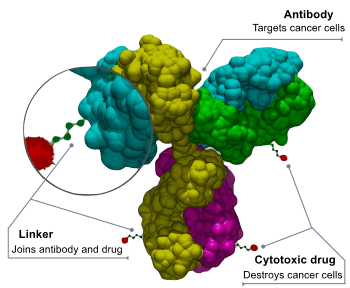
Antibody-drug conjugates or ADCs are an important class of highly potent biopharmaceutical drugs designed as a targeted therapy for the treatment of people with cancer. Unlike chemotherapy, ADCs are intended to target and kill only the cancer cells and spare healthy cells. ADCs are complex molecules composed of an antibody linked to a biologically active cytotoxic (anticancer) payload or drug.[1] Antibody-drug conjugates are examples of bioconjugates and immunoconjugates.
By combining the targeting capabilities of monoclonal antibodies with the cancer-killing ability of cytotoxic drugs, antibody-drug conjugates allow for discrimination between healthy and diseased tissue. In contrast to traditional chemotherapeutic drugs, antibody-drug conjugates target only cancer cells so that healthy cells are less severely affected.[2][3]
Mechanism of action
In developing antibody-drug conjugates, an anticancer drug is coupled to an antibody that specifically targets a certain tumor marker (e.g. a protein that, ideally, is only to be found in or on tumor cells). Antibodies track these proteins down in the body and attach themselves to the surface of cancer cells. The biochemical reaction between the antibody and the target protein (antigen) triggers a signal in the tumor cell, which then absorbs or internalizes the antibody together with the cytotoxin. After the ADC is internalized, the cytotoxic drug is released and kills the cancer.[4] Due to this targeting, ideally the drug has lower side effects and gives a wider therapeutic window than other chemotherapeutic agents. These advantages have led to ADC technologies being featured in many publications, notably The New York Times,[5][6] as well as numerous scientific journals.
Marketed ADC Products
To date, only four ADCs have received market approval. However, after a request from the U.S. Food and Drug Administration (FDA), Pfizer/Wyeth, the developer and marketer of the first ADC to receive marketing approval in 2001 for the treatment of patients with acute myelogenous leukemia (Gemtuzumab ozogamicin, trade name: Mylotarg), withdrew the drug from the market in June 2010[7] (although still marketed in Japan[8]). It was re-introduced into the US market in 2017.[9] The second and third marketed ADCs are Brentuximab vedotin (trade name: Adcetris, marketed by Seattle Genetics and Millennium/Takeda) and Trastuzumab emtansine (trade name: Kadcyla, marketed by Genentech and Roche).
Brentuximab vedotin[10] was granted accelerated approval by the U.S. Food and Drug Administration (FDA) on August 19, 2011 for relapsed HL and relapsed sALCL and conditional Marketing authorization from the European Medicines Agency in October 2012 for relapsed or refractory HL and relapsed or refractory sALCL. Trastuzumab emtansine (ado-trastuzumab emtansine or T-DM1) was approved in February 2013 for the treatment of people with HER2-positive metastatic breast cancer (mBC) who have received prior treatment with trastuzumab (Herceptin, Genentech and Roche) and a taxane chemotherapy.[11][12] The newest ADC, Inotuzumab ozogamicin,[13] was approved by the European Commission as monotherapy for the treatment of adults with relapsed or refractory CD22-positive B-cell precursor acute lymphoblastic leukemia (ALL) on June 30, 2017 under the trade name Besponsa®(Pfizer/Wyeth).[14] On August 17, 2017 Inotuzumab ozogamicin was also approved by the US FDA for the treatment of adults with relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL)[15]
Cleavable and Non-Cleavable Linkers
A stable link between the antibody and cytotoxic (anti-cancer) agent is a crucial aspect of an ADC.[16] A highly stable ADC linker will ensure that less of the cytotoxic payload falls off in circulation, driving an improved safety profile, and will also ensure that more of the payload arrives at the cancer cell, driving enhanced efficacy. Linkers are based on chemical motifs including disulfides, hydrazones or peptides (cleavable), or thioethers (noncleavable) and control the distribution and delivery of the cytotoxic agent to the target cell. Cleavable and noncleavable types of linkers have been proven to be safe in preclinical and clinical trials. Brentuximab vedotin includes an enzyme-sensitive cleavable linker that delivers the potent and highly toxic antimicrotubule agent monomethyl auristatin E or MMAE, a synthetic antineoplastic agent, to human specific CD30-positive malignant cells. Because of its high toxicity MMAE, which inhibits cell division by blocking the polymerization of tubulin, cannot be used as a single-agent chemotherapeutic drug. However, the combination of MMAE linked to an anti-CD30 monoclonal antibody (cAC10, a cell membrane protein of the tumor necrosis factor or TNF receptor) proved to be stable in extracellular fluid, cleavable by cathepsin and safe for therapy. Trastuzumab emtansine, the other approved ADC, is a combination of the microtubule-formation inhibitor mertansine (DM-1), a derivative of the Maytansine, and antibody trastuzumab (Herceptin/ Genentech/Roche) attached by a stable, non-cleavable linker.
The availability of better and more stable linkers has changed the function of the chemical bond. The type of linker, cleavable or noncleavable, lends specific properties to the cytotoxic (anti-cancer) drug. For example, a non-cleavable linker keeps the drug within the cell. As a result, the entire antibody, linker and cytotoxic (anti-cancer) agent enter the targeted cancer cell where the antibody is degraded to the level of an amino acid. The resulting complex – amino acid, linker and cytotoxic agent – now becomes the active drug. In contrast, cleavable linkers are catalyzed by enzymes in the cancer cell where it releases the cytotoxic agent. The difference is that the cytotoxic payload delivered via a cleavable linker can escape from the targeted cell and, in a process called "bystander killing", attack neighboring cancer cells.[17]
Another type of cleavable linker, currently in development, adds an extra molecule between the cytotoxic drug and the cleavage site. This linker technology allows researchers to create ADCs with more flexibility without worrying about changing cleavage kinetics. Researchers are also developing a new method of peptide cleavage based on Edman degradation, a method of sequencing amino acids in a peptide.[18] Future direction in the development of ADCs also include the development of site-specific conjugation (TDCs)[19] and novel conjugation techniques[20][21] to further improve stability and therapeutic index, α emitting immunoconjugates[22] and antibody-conjugated nanoparticles.[23]
Non-natural amino acids
The first generation of ADCs use linking technologies that conjugate drugs non-selectively to cysteine or lysine residues in the antibody, resulting in a heterogeneous mixture of ADCs. This approach leads to suboptimal safety and efficacy properties and makes optimization of the biological, physical and pharmacological properties of an ADC challenging.[24] The site-specific incorporation of unnatural amino acids into the antibody generates a site for controlled and stable attachment of the drug. This enables the production of homogeneous ADCs with the antibody precisely linked to the drug and controlled ratios of antibody to drug, allowing the selection of a best-in-class ADC.[24] An Escherichia coli-based open cell-free synthesis (OCFS), developed by Stanford Professor James R. Swartz, allows the synthesis of proteins containing site-specifically incorporated non-natural amino acids and has been optimized for predictable high-yield protein synthesis and folding at any scale with straightforward downstream purification processes. The absence of a cell wall allows the addition of non-natural factors to the open system in order to manipulate transcription, translation and folding to provide precise modulation of the protein expression process.[25]
Other disease areas
The majority of the ADCs currently under development or in clinical trials are for oncological and hematological indications.[26] This is primarily driven by the availability of monoclonal antibodies targeting various types of cancer. However, some drug developers are also looking to expanding the application of ADCs beyond oncology and hematology to other important disease areas.[27][28]
References
- ↑ Antibody-Drug Conjugates Stage a Comeback
- ↑ Dijoseph, JF; Armellino, DC; Boghaert, ER; Khandke, K; Dougher, MM; Sridharan, L; Kunz, A; Hamann, PR; Gorovits, B; Udata, C; Moran, JK; Popplewell, AG; Stephens, S; Frost, P; Damle, NK (2004). "Antibody-targeted chemotherapy with CMC-544: A CD22-targeted immunoconjugate of calicheamicin for the treatment of B-lymphoid malignancies". Blood. 103 (5): 1807–14. doi:10.1182/blood-2003-07-2466. PMID 14615373.
- ↑ Mullard, Asher (2013). "Maturing antibody–drug conjugate pipeline hits 30". Nature Reviews Drug Discovery. 12 (5): 329–32. doi:10.1038/nrd4009. PMID 23629491.
- ↑ Chari, Ravi V. J.; Martell, Bridget A.; Gross, Jonathan L.; Cook, Sherrilyn B.; Shah, Sudhir A.; Blättler, Walter A.; McKenzie, Sara J.; Goldmacher, Victor S. (1992). "Immunoconjugates containing novel maytansinoids: promising anticancer drugs". Cancer Research. 52 (1): 127–31. PMID 1727373.
- ↑ , Pollack A. May 31, 2012. In print on June 1, 2012, on page B1 of the New York edition with the headline: A One-Two Punch.
- ↑ , Published June 3, 2012
- ↑ FDA: Pfizer Voluntarily Withdraws Cancer Treatment Mylotarg from U.S. Market, US FDA
- ↑ Pharmaceuticals and Medical Devices Safety Information, No. 277, February 2011 (Technical report). Pharmaceuticals and Medical Devices Agency of Japan, 2011.
- ↑ "Approved Drugs > FDA Approves Gemtuzumab Ozogamicin for CD33-positive AML". fda.gov. Silver Spring, USA: U.S. Food and Drug Administration. 1 September 2017. Retrieved 6 September 2017.
- ↑ Brentuximab vedotin (SGN35), ADC Review/Journal of Antibody-drug Conjugates
- ↑ FDA Approves Genentech's Kadcyla® (Ado-Trastuzumab Emtansine), the First Antibody-Drug Conjugate for Treating Her2-Positive Metastatic Breast Cancer
- ↑ Ado-trastuzumab emtansine (U.S. Department of Health and Human Services | National Institutes of Health | National Cancer Institute.)
- ↑ Inotuzumab ozogamicin (drug description), ADC Review/Journal of Antibody-drug Conjugates
- ↑ BESPONSA® Approved in the EU for Adult Patients with Relapsed or Refractory B-cell Precursor Acute Lymphoblastic Leukemia
- ↑ U.S. FDA Approves Inotuzumab Ozogamicin for Treatment of Patients with R/R B-cell precursor Acute Lymphoblastic Leukemia, ADC Review/Journal of Antibody-drug Conjugates, August 17, 2017
- ↑ Beck, A.; Goetsch, L.; Dumontet, C.; Corvaïa, N. (2017). "Strategies and challenges for the next generation of antibody-drug conjugates". Chem. Soc. Rev. 19 (16): 315–337. doi:10.1038/nrd.2016.268.
- ↑ Kovtun, YV; Goldmacher, VS (2007). "Cell killing by antibody-drug conjugates". Cancer Letters. 255 (2): 232–40. doi:10.1016/j.canlet.2007.04.010. PMID 17553616.
- ↑ Bąchor, R; Kluczyk, A; Stefanowicz, P; Szewczuk, Z (2013). "New method of peptide cleavage based on Edman degradation". Molecular diversity. 17 (3): 605–11. doi:10.1007/s11030-013-9453-y. PMC 3713267. PMID 23690169.
- ↑ Axup, J. Y.; Bajjuri, K. M.; Ritland, M.; Hutchins, B. M.; Kim, C. H.; Kazane, S. A.; Halder, R.; Forsyth, J. S.; Santidrian, A. F.; Stafin, K.; Lu, Y.; Tran, H.; Seller, A. J.; Biroc, S. L.; Szydlik, A.; Pinkstaff, J. K.; Tian, F.; Sinha, S. C.; Felding-Habermann, B.; Smider, V. V.; Schultz, P. G. (2012). "Synthesis of site-specific antibody-drug conjugates using unnatural amino acids". Proceedings of the National Academy of Sciences. 109 (40): 16101–6. doi:10.1073/pnas.1211023109. PMC 3479532. PMID 22988081.
- ↑ Lyon, R.P.; Setter, J.R.; Bovee, T.D.; Doronina, S.O.; Hunter, J.H.; Anderson M.E.; Balasubramanian, C.L.; Duniho, S.M.; Leiske, C.I.; Li, F.; Senter, P.D. (2014). "Self-hydrolyzing maleimides improve the stability and pharmacological properties of antibody-drug conjugates". Bioconjugate Chem. 32 (10): 1059–1062. doi:10.1038/nbt.2968.
- ↑ Kolodych, S.; Koniev, O.; Baatarkhuu, Z.; Bonnefoy, J.-Y.; Debaene, F.; Chienférani, S.; Dorsselaer, A.; Wagner, A. (2015). "CBTF: new amine-to-thiol coupling reagent for preparation of antibody conjugates with increased plasma stability". Bioconjugate Chem. 26 (2): 197–200. doi:10.1021/bc500610g.
- ↑ Wulbrand, C; Seidl, C; Gaertner, FC; Bruchertseifer, F; Morgenstern, A; Essler, M; Senekowitsch-Schmidtke, R (2013). Multhoff, Gabriele, ed. "Alpha-particle emitting 213Bi-anti-EGFR immunoconjugates eradicate tumor cells independent of oxygenation". PLoS ONE. 8 (5): e64730. doi:10.1371/journal.pone.0064730. PMC 3665541. PMID 23724085.
- ↑ Cardoso, MM; Peça, IN; Roque, AC (2012). "Antibody-conjugated nanoparticles for therapeutic applications". Current Medicinal Chemistry. 19 (19): 3103–27. doi:10.2174/092986712800784667. PMID 22612698.
- 1 2 Axup, JY; Bajjuri, KM; Ritland, M; et al. (October 2012). "Synthesis of site-specific antibody-drug conjugates using unnatural amino acids". Proc. Natl. Acad. Sci. U.S.A. 109 (40): 16101–6. doi:10.1073/pnas.1211023109. PMC 3479532. PMID 22988081.
- ↑ Zawada, JF; Yin, G; Steiner, AR; Yang, J; Naresh, A; Roy, SM; Gold, DS; Heinsohn, HG; Murray, CJ (2011). "Microscale to Manufacturing Scale-up of Cell-Free Cytokine Production—A New Approach for Shortening Protein Production Development Timelines". Biotechnol Bioeng. 108 (7): 1570–8. doi:10.1002/bit.23103. PMC 3128707. PMID 21337337.
- ↑ Flygare, John A.; Pillow, Thomas H.; Aristoff, Paul (2013). "Antibody-Drug Conjugates for the Treatment of Cancer". Chemical Biology & Drug Design. 81: 113–121. doi:10.1111/cbdd.12085.
- ↑ Lehar, Sophie M.; Pillow, Thomas; Xu, Min; Staben, Leanna; Kajihara, Kimberly K.; Vandlen, Richard; DePalatis, Laura; Raab, Helga; Hazenbos, Wouter L.; Morisaki, J. Hiroshi; Kim, Janice; Park, Summer; Darwish, Martine; Lee, Byoung-Chul; Hernandez, Hilda; Loyet, Kelly M.; Lupardus, Patrick; Fong, Rina; Yan, Donghong; Chalouni, Cecile; Luis, Elizabeth; Khalfin, Yana; Plise, Emile; Cheong, Jonathan; Lyssikatos, Joseph P.; Strandh, Magnus; Koefoed, Klaus; Andersen, Peter S.; Flygare, John A.; Tan, Man Wah; Brown, Eric J.; Mariathasan, Sanjeev (2015). "Novel antibody–antibiotic conjugate eliminates intracellular S. aureus". Nature. Springer Nature. 527 (7578): 323–328. doi:10.1038/nature16057. Retrieved 2017-05-11.
- ↑ Ambrx Collaborates with Merck to Design and Develop Biologic Drug Conjugates "Archived copy". Archived from the original on 2013-01-07. Retrieved 2013-06-30.