Ammonium hexachloroiridate(IV)
 | |
2PtCl6Xray.tif.png) | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.037.264 |
| EC Number | 241-007-0 |
PubChem CID |
|
| |
| |
| Properties | |
| H8N2Cl6Ir | |
| Molar mass | 441.01 |
| Appearance | brown crystals |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Ammonium hexachloroiridate(IV) is the inorganic compound with the formula (NH4)2[IrCl6]. This dark brown solid is the ammonium salt of the iridium(IV) complex [IrCl6]2−. It is a commercially important iridium compound[1] one of the most common complexes of iridium(IV). A related but ill-defined compound is iridium tetrachloride, which is often used interchangeably.[2]
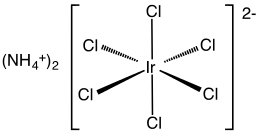
Structure
The compound has been characterized by X-ray crystallography. The salt crystallizes in a cubic motif like that of ammonium hexachloroplatinate. The [IrCl6]2− centers adopt octahedral molecular geometry.[3]
Uses
It is a key intermediate in the isolation of iridium from ores. Most other metals form insoluble sulfides when aqueous solutions of their chlorides are treated with hydrogen sulfide, but [IrCl6]2− resists ligand substitution. Upon heating under hydrogen, the solid salt converts to the metal:[1]
- (NH4)2[IrCl6] + 2 H2 → Ir + 6 HCl + 2 NH3
References
- 1 2 Renner, H.; Schlamp, G.; Kleinwächter, I.; Drost, E.; Lüschow, H. M.; Tews, P.; Panster, P.; Diehl, M.; et al. (2002). "Platinum group metals and compounds". Ullmann's Encyclopedia of Industrial Chemistry. Wiley. doi:10.1002/14356007.a21_075.
- ↑ Thomas R. B. Mitchell (2001). "Iridium(IV) Chloride". e-EROS Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.ri050.
- ↑ Bokii, G.B.; Ussikov, P.I. "Roentgenographische Untersuchung der Struktur des Ammonium-Chlor-Iridats (N H4)2IrCl6 Doklady Akademii Nauk SSSR 1940, vol. 26, p782-p784.