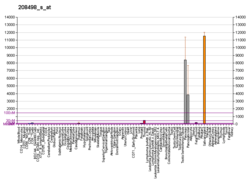
Alpha-amylase 2B is an enzyme that in humans is encoded by the AMY2B gene.[5][6]
Amylases are secreted proteins that hydrolyze 1,4-alpha-glucoside bonds in oligosaccharides and polysaccharides, and thus catalyze the first step in digestion of dietary starch and glycogen. The human genome has a cluster of several amylase genes that are expressed at high levels in either salivary gland or pancreas. This gene encodes an amylase isoenzyme produced by the pancreas.[6]
Further reading
- Kaczmarek MJ, Rosenmund H (1977). "The action of human pancreatic and salivary isoamylases on starch and glycogen". Clin. Chim. Acta. 79 (1): 69–73. doi:10.1016/0009-8981(77)90462-4. PMID 890964.
- Groot PC, Mager WH, Henriquez NV, et al. (1991). "Evolution of the human alpha-amylase multigene family through unequal, homologous, and inter- and intrachromosomal crossovers". Genomics. 8 (1): 97–105. doi:10.1016/0888-7543(90)90230-R. PMID 2081604.
- Yokouchi H, Horii A, Emi M, et al. (1990). "Cloning and characterization of a third type of human alpha-amylase gene, AMY2B". Gene. 90 (2): 281–6. doi:10.1016/0378-1119(90)90191-S. PMID 2401405.
- Gumucio DL, Wiebauer K, Caldwell RM, et al. (1988). "Concerted evolution of human amylase genes". Mol. Cell. Biol. 8 (3): 1197–205. PMC 363264. PMID 2452973.
- Samuelson LC, Wiebauer K, Gumucio DL, Meisler MH (1988). "Expression of the human amylase genes: recent origin of a salivary amylase promoter from an actin pseudogene". Nucleic Acids Res. 16 (17): 8261–76. doi:10.1093/nar/16.17.8261. PMC 338557. PMID 2458567.
- Tomita N, Horii A, Doi S, et al. (1989). "A novel type of human alpha-amylase produced in lung carcinoid tumor". Gene. 76 (1): 11–8. doi:10.1016/0378-1119(89)90003-6. PMID 2701942.
- Groot PC, Bleeker MJ, Pronk JC, et al. (1989). "The human alpha-amylase multigene family consists of haplotypes with variable numbers of genes". Genomics. 5 (1): 29–42. doi:10.1016/0888-7543(89)90083-9. PMID 2788608.
- Groot PC, Bleeker MJ, Pronk JC, et al. (1988). "Human pancreatic amylase is encoded by two different genes". Nucleic Acids Res. 16 (10): 4724. doi:10.1093/nar/16.10.4724. PMC 336663. PMID 3260028.
- Tricoli JV, Shows TB (1984). "Regional assignment of human amylase (AMY) to p22----p21 of chromosome 1". Somat. Cell Mol. Genet. 10 (2): 205–10. doi:10.1007/BF01534909. PMID 6608795.
- Nagase T, Nakayama M, Nakajima D, et al. (2001). "Prediction of the coding sequences of unidentified human genes. XX. The complete sequences of 100 new cDNA clones from brain which code for large proteins in vitro". DNA Res. 8 (2): 85–95. doi:10.1093/dnares/8.2.85. PMID 11347906.
- Aughsteen AA (2001). "A comparative immunohistochemical study on amylase localization in the rat and human exocrine pancreas". Saudi medical journal. 22 (5): 410–5. PMID 11376382.
- Koyama I, Komine S, Iino N, et al. (2001). "alpha-Amylase expressed in human liver is encoded by the AMY-2B gene identified in tumorous tissues". Clin. Chim. Acta. 309 (1): 73–83. doi:10.1016/S0009-8981(01)00501-0. PMID 11408008.
- Strausberg RL, Feingold EA, Grouse LH, et al. (2003). "Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences". Proc. Natl. Acad. Sci. U.S.A. 99 (26): 16899–903. doi:10.1073/pnas.242603899. PMC 139241. PMID 12477932.
- Ota T, Suzuki Y, Nishikawa T, et al. (2004). "Complete sequencing and characterization of 21,243 full-length human cDNAs". Nat. Genet. 36 (1): 40–5. doi:10.1038/ng1285. PMID 14702039.
- Iafrate AJ, Feuk L, Rivera MN, et al. (2004). "Detection of large-scale variation in the human genome". Nat. Genet. 36 (9): 949–51. doi:10.1038/ng1416. PMID 15286789.
- Gerhard DS, Wagner L, Feingold EA, et al. (2004). "The status, quality, and expansion of the NIH full-length cDNA project: the Mammalian Gene Collection (MGC)". Genome Res. 14 (10B): 2121–7. doi:10.1101/gr.2596504. PMC 528928. PMID 15489334.
PDB gallery |
|---|
1b2y: STRUCTURE OF HUMAN PANCREATIC ALPHA-AMYLASE IN COMPLEX WITH THE CARBOHYDRATE INHIBITOR ACARBOSE 1bsi: HUMAN PANCREATIC ALPHA-AMYLASE FROM PICHIA PASTORIS, GLYCOSYLATED PROTEIN 1c8q: STRUCTURE SOLUTION AND REFINEMENT OF THE RECOMBINANT HUMAN SALIVARY AMYLASE 1cpu: SUBSITE MAPPING OF THE ACTIVE SITE OF HUMAN PANCREATIC ALPHA-AMYLASE USING SUBSTRATES, THE PHARMACOLOGICAL INHIBITOR ACARBOSE, AND AN ACTIVE SITE VARIANT 1hny: The structure of human pancreatic alpha-amylase at 1.8 angstroms resolution and comparisons with related enzymes 1jxj: Role of mobile loop in the mechanism of human salivary amylase 1jxk: Role of the mobile loop in the mechanism of human salivary amylase 1kb3: Three Dimensional Structure Analysis of the R195A Variant of Human Pancreatic Alpha Amylase 1kbb: Mechanistic Analyses of Catalysis in Human Pancreatic alpha-Amylase: Detailed Kinetic and Structural Studies of Mutants of Three Conserved Carboxylic Acids 1kbk: Mechanistic Analyses of Catalysis in Human Pancreatic Alpha-Amylase: Detailed Kinetic and Structural Studies of Mutants of Three Conserved Carboxylic Acids 1kgu: THREE DIMENSIONAL STRUCTURE ANALYSIS OF THE R337A VARIANT OF HUMAN PANCREATIC ALPHA-AMYLASE 1kgw: THREE DIMENSIONAL STRUCTURE ANALYSIS OF THE R337Q VARIANT OF HUMAN PANCREATIC ALPHA-MYLASE 1kgx: Three Dimensional Structure Analysis of the R195Q Variant of Human Pancreatic Alpha Amylase 1mfu: Probing the role of a mobile loop in human salivary amylase: Structural studies on the loop-deleted mutant 1mfv: Probing the role of a mobile loop in human slaivary amylase: Structural studies on the loop-deleted enzyme 1nm9: Crystal structure of recombinant human salivary amylase mutant W58A 1q4n: Structural studies of Phe256Trp of human salivary alpha-amylase: implications for the role of a conserved water molecule and its associated chain in enzyme activity 1smd: HUMAN SALIVARY AMYLASE 1u2y: In situ extension as an approach for identifying novel alpha-amylase inhibitors, structure containing D-gluconhydroximo-1,5-lactam 1u30: In situ extension as an approach for identifying novel alpha-amylase inhibitors, structure containing maltosyl-alpha (1,4)-D-gluconhydroximo-1,5-lactam 1u33: In situ extension as an approach for identifying novel alpha-amylase inhibitors 1xcw: Acarbose Rearrangement Mechanism Implied by the Kinetic and Structural Analysis of Human Pancreatic alpha-Amylase in Complex with Analogues and Their Elongated Counterparts 1xcx: Acarbose Rearrangement Mechanism Implied by the Kinetic and Structural Analysis of Human Pancreatic alpha-Amylase in Complex with Analogues and Their Elongated Counterparts 1xd0: Acarbose Rearrangement Mechanism Implied by the Kinetic and Structural Analysis of Human Pancreatic alpha-Amylase in Complex with Analogues and Their Elongated Counterparts 1xd1: Acarbose Rearrangement Mechanism Implied by the Kinetic and Structural Analysis of Human Pancreatic alpha-Amylase in Complex with Analogues and Their Elongated Counterparts 1xgz: Structure of the N298S variant of human pancreatic alpha-amylase 1xh0: Structure of the N298S variant of human pancreatic alpha-amylase complexed with acarbose 1xh1: Structure of the N298S variant of human pancreatic alpha-amylase complexed with chloride 1xh2: Structure of the N298S variant of human pancreatic alpha-amylase complexed with chloride and acarbose 1xv8: Crystal Structure of Human Salivary Alpha-Amylase Dimer 1z32: Structure-function relationships in human salivary alpha-amylase: Role of aromatic residues 2cpu: SUBSITE MAPPING OF THE ACTIVE SITE OF HUMAN PANCREATIC ALPHA-AMYLASE USING SUBSTRATES, THE PHARMACOLOGICAL INHIBITOR ACARBOSE, AND AN ACTIVE SITE VARIANT 3cpu: SUBSITE MAPPING OF THE ACTIVE SITE OF HUMAN PANCREATIC ALPHA-AMYLASE USING SUBSTRATES, THE PHARMACOLOGICAL INHIBITOR ACARBOSE, AND AN ACTIVE SITE VARIANT |