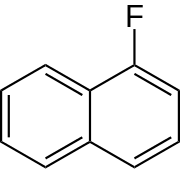
1-Fluoronaphthalene
 | |
| Names | |
|---|---|
| Other names
α-fluoronaphthalene | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.005.717 |
| EC Number | 206-287-0 |
PubChem CID |
|
| |
| |
| Properties | |
| C 10H 7F | |
| Appearance | Colorless liquid |
| Melting point | −13 °C (9 °F; 260 K) |
| Boiling point | 215 °C (419 °F; 488 K) |
| insoluble | |
| Hazards | |
| GHS pictograms |  |
| GHS signal word | WARNING |
| Flash point | 65 °C (149 °F; 338 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
1-Fluornaphthalene is an organofluorine chemical compound from the group of naphthalene derivatives and fluoroaromatics. Its chemical formula is C
10H
7F.[1]
Synthesis
1-Fluoronaphthalene can be obtained by reacting naphthalene with Selectfluor.[2]
Properties
1-Fluornaphthalene is a colorless, combustible liquid, which is insoluble in water.[3]
Applications
1-Fluoronaphthalene was used for the tert-butyllithium-mediated synthesis of 6-substituted phenanthridines. It has also been used in the synthesis of LY248686, a potent inhibitor of serotonin and norepinephrine uptake.
See also
References
- ↑ "1-Fluoronaphthalene". Sigma Aldrich. sigmaaldrich.com. Retrieved 13 June 2017.
- ↑ Atta-Ur-Rahman (2006). Advances in Organic Synthesis: Modern Organofluorine Chemistry-Synthetic Aspects. Bentham Science Publishers. p. 246. ISBN 1-60805-198-6.
- ↑ "1-FLUORONAPHTHALENE". CAMEO Chemicals. cameochemicals.noaa.gov. Retrieved 13 June 2017.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.