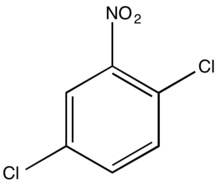
1,4-Dichloro-2-nitrobenzene
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,4-Dichloro-2-nitrobenzene | |
| Other names
Nitro-P-dichlorobenzene | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.749 |
| MeSH | C503932 |
PubChem CID |
|
| RTECS number | CZ5260000 |
| UNII | |
| UN number | 1578 |
| |
| |
| Properties | |
| C6H3Cl2NO2 | |
| Molar mass | 192.00 |
| Appearance | yeloow flakes |
| Density | 1.67 |
| Melting point | 52-54 °C |
| Boiling point | 266-269 °C |
| 95 mg/l | |
| Hazards | |
| GHS pictograms |    |
| GHS signal word | Danger |
| H302, H336, H351, H361, H370, H372, H373, H400, H410 | |
| P201, P202, P260, P261, P264, P270, P271, P273, P281, P301+312, P304+340, P307+311, P308+313, P312, P314, P321, P330, P391, P403+233, P405, P501 | |
| Flash point | 135°C |
| 465°C | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
1,4-Dichloro-2-nitrobenzene is an organic compound with the formula C6H3Cl2NO2. One of several isomers of dichloronitrobenzene, it is a colorless solid that is insoluble in water. It is produced by nitration of 1,4-dichlorobenzene. It is a precursor to many derivatives of commercial interest. Hydrogenation gives 1,4-dichloroaniline. Nucleophiles displace the chloride adjacent to the nitro group: ammonia gives the aniline derivative, aqueous base gives the phenol derivative, and methoxide gives the anisole derivative. These compounds are respectively 4-chloro-2-nitroaniline, 4-chloro-2-nitrophenol, and 4-chloro-2-nitroanisole.[1] Isomeric with this compound is 1,2-dichloro-4-nitrobenzene.
References
- ↑ Gerald Booth (2007). "Nitro Compounds, Aromatic" in Ullmann's Encyclopedia of Industrial Chemistry Wiley-VCH, Weinheim, 2005.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.