Tetanus vaccine
Tetanus vaccine, also known as tetanus toxoid (TT), is an inactive vaccine used to prevent tetanus.[1] During childhood, five doses are recommended, with a sixth given during adolescence.[1] Additional doses every ten years are recommended.[2] After three doses, almost everyone is initially immune.[1] In those who are not up to date on their tetanus immunization, a booster should be given within 48 hours of an injury.[3] In those with high-risk injuries who are not fully immunized, tetanus antitoxin may also be recommended.[1] Making sure pregnant women are up to date on their tetanus immunization can prevent neonatal tetanus.[1]
 Tetanus vaccination is often administered via combination DPT vaccines | |
| Vaccine description | |
|---|---|
| Target disease | Tetanus |
| Type | Toxoid |
| Clinical data | |
| MedlinePlus | a682198 |
| License data |
|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| ChemSpider |
|
| | |
The vaccine is very safe, including during pregnancy and in those with HIV/AIDS.[1] Redness and pain at the site of injection occur in between 25% and 85% of people.[1] Fever, feeling tired, and minor muscle pain occurs in less than 10% of people.[1] Severe allergic reactions occur in less than one in 100,000 people.[1]
A number of vaccine combinations include the tetanus vaccine, such as DTaP and Tdap, which contain diphtheria, tetanus, and pertussis vaccines, and DT and Td, which contain diphtheria and tetanus vaccines.[4] DTaP and DT are given to children less than seven years old, while Tdap and Td are given to those seven years old and older.[4][5] The lowercase d and p denote lower strengths of diphtheria and pertussis vaccines.[4]
Tetanus antiserum was developed in 1890, with its protective effects lasting a few weeks.[6][7] The tetanus toxoid vaccine was developed in 1924, and came into common use for soldiers in World War II.[1][8] Its use resulted in a 95% decrease in the rate of tetanus.[1] It is on the World Health Organization's List of Essential Medicines, the safest and most effective medicines needed in a health system.[9] The wholesale cost in the developing world is between 0.17 and 0.65 USD per dose as of 2014.[10] In the United States, a course of tetanus vaccine is between 25 and 50 USD.[11][12]
Medical uses
Effectiveness
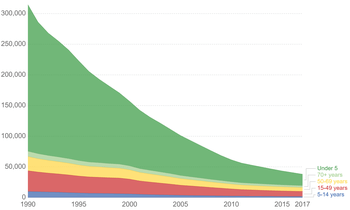
Following vaccination, 95% of people are protected from diphtheria, 80% to 85% from pertussis, and 100% from tetanus.[14] Globally deaths from tetanus in newborns decreased 787,000 in 1988 to 58,000 in 2010, and 34,000 deaths in 2015 (a 96% decrease from 1988).[2][15]
In the 1940s, before the vaccine, there was about 550 cases of tetanus per year in the United States which has decreased to about 30 cases per year in the 2000s.[2] Nearly all cases are among those who have never received a vaccine, or adults who don't stay up to date on their 10-year booster shots.[16]
Pregnancy
Guidelines on prenatal care in the United States specify that women should receive a dose of the Tdap vaccine during each pregnancy, preferably between weeks 27 and 36, to allow antibody transfer to the fetus.[17][18] All postpartum women who have not previously received the Tdap vaccine are recommended to get it prior to discharge after delivery.[19] It is recommended for pregnant women who have never received the tetanus vaccine (i.e., neither DTP or DTaP, nor DT as a child or Td or TT as an adult) to receive a series of three Td vaccinations starting during pregnancy to ensure protection against maternal and neonatal tetanus. In such cases, Tdap is recommended to be substituted for one dose of Td, again preferably between 27 and 36 weeks of gestation, and then the series completed with Td.[17][18]
Specific types
The first vaccine is given in infancy. The baby is injected with the DTaP vaccine, which is three inactive toxins in one injection. DTaP protects against diphtheria, pertussis, and tetanus. This vaccine is safer than the previously used DTP.[4] Another option is DT, which is a combination of diphtheria and tetanus vaccines. This is given as an alternative to infants who have conflicts with the DTaP vaccine.[14] Quadrivalent, pentavalent, and hexavalent formulations contain DTaP with one or more of the additional vaccines: inactivated polio virus vaccine (IPV), Haemophilus influenzae type b conjugate, Hepatitis B, with the availability varying in different countries.[20][21][22]
For the every ten-year booster Td or Tdap may be used, though Tdap is more expensive.[18]
Schedule
Because DTaP and DT are administered to children less than a year old, the recommended location for injection is the anterolateral thigh muscle. However, these vaccines can be injected into the deltoid muscle if necessary.
The World Health Organization (WHO) recommends six doses in childhood starting at six weeks of age.[1] Four doses of DTaP are to be given in early childhood.[14] The first dose should be around two months of age, the second at four months, the third at six, and the fourth from fifteen to eighteen months of age. There is a recommended fifth dose to be administered to four- to six-year-olds.[14]
Td and Tdap are for older children, adolescents, and adults and can be injected into the deltoid muscle.[14] These are boosters and are recommended every ten years. It is safe to have shorter intervals between a single dose of Tdap and a dose of the Td booster.[23]
Additional doses
Booster shots are important because lymphocyte production (antibodies) is not at a constant high rate of activity. This is because after the introduction of the vaccine when lymphocyte production is high, the production activity of white blood cells will start to decline. The decline in activity of the T-helper cells means that there must be a booster to help keep the white blood cells active.[24]
Td and Tdap are the booster shots given every ten years to maintain immunity for adults nineteen years of age to sixty-five years of age.[4][18]
Tdap is given as a one-time, first-time-only dose that includes the tetanus, diphtheria, and acellular pertussis vaccinations.[4] This should not be administered to those who are under the age of eleven or over the age of sixty-five.
Td is the booster shot given to people over the age of seven and includes the tetanus and diphtheria toxoids. However, Td has less of the diphtheria toxoid, which is why the "d" is lowercase and the "T" is capitalized.[4]
It is important to understand that booster shots should be administered before the age of sixty-five and that one of these booster shots should be Tdap while the rest are Td.[14]
In 2020, the US Centers for Disease Control and Prevention (CDC) Advisory Committee on Immunization Practices (ACIP) recommended that either tetanus and diphtheria toxoids (Td) vaccine or Tdap to be used for the decennial Td booster, tetanus prevention during wound management, and for additional required doses in the catch-up immunization schedule if a person has received at least one Tdap dose.[18]
Side effects
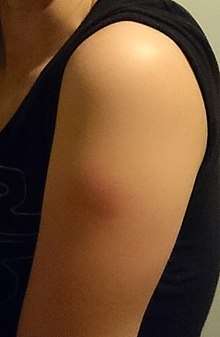
Common side effects of the tetanus vaccine include fever, redness, and swelling with soreness or tenderness around the injection site (one of five people have redness or swelling). Body aches and tiredness have been reported following Tdap. Td / Tdap can cause painful swelling of the entire arm in one of 500 people.[14][25] Tetanus toxoid containing vaccines (DTaP, DTP, Tdap, Td, DT) may cause brachial neuritis at a rate of one out of every 100,000 to 200,000 doses.[2][26]
Mechanism of action
The type of vaccination for this disease is called artificial active immunity. This type of immunity is generated when a dead or weakened version of the disease enters the body, causing an immune response which includes the production of antibodies. This is beneficial because it means that if the disease is ever introduced into the body, the immune system will recognize the antigen and produce antibodies more rapidly.[27]
History
The first vaccine for passive immunology was discovered by a group of German scientists under the leadership of Emil von Behring in 1890. The first inactive tetanus toxoid was discovered and produced in 1924. A more effective adsorbed version of the vaccine, created in 1938, was proven to be successful when it was used to prevent tetanus in the military during World War II.[14] DTP (which is the combined vaccine for diphtheria, tetanus, and pertussis) was first used in 1948, and was continued until 1991, when it was replaced with an acellular form of the pertussis vaccine due to safety concerns.[28] Half of those who received the DTP vaccine had redness, swelling, and pain around the injection site[14], which convinced researchers to find a replacement vaccine.
Two new vaccines were launched in 1992. These combined tetanus and diphtheria with acellular pertussis (TDaP or DTaP), which could be given to adolescents and adults (as opposed to previously when the vaccine was only given to children).[14]
References
- World Health Organization (February 2017). "Tetanus vaccines: WHO position paper – February 2017". Weekly epidemiological record. 92 (6): 53–76. hdl:10665/254583. PMID 28185446. Lay summary (PDF).
- Hamborsky J, Kroger A, Wolfe S, eds. (2015). "Chapter 21: Tetanus". Epidemiology and Prevention of Vaccine-Preventable Diseases (13th ed.). Centers for Disease Control and Prevention (CDC). Archived from the original on 2017-04-13.
- "Puncture wounds: First aid". Mayo Clinic. Feb 4, 2015. Archived from the original on 8 December 2015. Retrieved 6 December 2015.
- "Vaccines: VPD-VAC/Tetanus/main page". Centers for Disease Control and Prevention (CDC). Archived from the original on February 12, 2015. Retrieved February 10, 2015.
- "Recommended Child and Adolescent Immunization Schedule for ages 18 years or younger, United States, 2020". Centers for Disease Control and Prevention (CDC). 3 February 2020. Retrieved 6 February 2020.
- Gradmann, C.; Simon, J. (2010). Evaluating and Standardizing Therapeutic Agents, 1890-1950. Springer. p. 53. ISBN 9780230285590.
- Jones, Bernard R. (2016). Pharmacology for Student and Pupil Nurses and Students in Associated Professions. Elsevier. p. 162. ISBN 9781483141312.
- Macera, Caroline (2012). Introduction to Epidemiology: Distribution and Determinants of Disease. Nelson Education. p. 251. ISBN 9781285687148. Archived from the original on 2016-03-05.
- World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- "Vaccine, Tetanus Toxoid". International Drug Price Indicator Guide. Retrieved 6 December 2015.
- Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 317. ISBN 9781284057560.
- "CDC Vaccine Price List". Centers for Disease Control and Prevention (CDC). 1 January 2020. Retrieved 28 January 2020.
- "Deaths from tetanus, by age". Our World in Data. Retrieved 13 January 2020.
- Centers for Disease Control and Prevention (2011). "Tetanus". Epidemiology and Prevention of Vaccine-Preventable Diseases (CDC, Epidemiology and Prevention of Vaccine-Preventable Diseases) (pdf). Washington, D.C: Public Health Foundation. ISBN 0-01-706609-3. Archived (PDF) from the original on March 6, 2008. Retrieved January 4, 2016.
- World Health Organization (9 May 2018). "Tetanus". World Health Organization (WHO). Retrieved 12 January 2020.
- "About Tetanus". Centers for Disease Control and Prevention (CDC). Archived from the original on 2014-11-11. Retrieved 2014-12-11.
- "Updated recommendations for use of tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis vaccine (Tdap) in pregnant women--Advisory Committee on Immunization Practices (ACIP), 2012" (PDF). MMWR Morb. Mortal. Wkly. Rep. 62 (7): 131–5. February 2013. PMC 4604886. PMID 23425962.
- Havers FP, Moro PL, Hunter P, Hariri S, Bernstein H (January 2020). "Use of Tetanus Toxoid, Reduced Diphtheria Toxoid, and Acellular Pertussis Vaccines: Updated Recommendations of the Advisory Committee on Immunization Practices - United States, 2019" (PDF). MMWR Morb. Mortal. Wkly. Rep. 69 (3): 77–83. doi:10.15585/mmwr.mm6903a5. PMID 31971933.
- "Pregnancy Guidelines and Recommendations by Vaccine - Tetanus, Diphtheria, and Pertussis (Tdap); & Tetanus and Diphtheria (Td)". Centers for Disease Control and Prevention (CDC). August 2016. Retrieved 15 January 2020.
- "2016 Binational Immunization Resource Tool for Children from Birth Through 18 Years" (PDF). Centers for Disease Control and Prevention (CDC). Archived (PDF) from the original on 2017-02-11. Retrieved 2016-12-22.
- "Licensure of a diphtheria and tetanus toxoids and acellular pertussis adsorbed, inactivated poliovirus, and haemophilus B conjugate vaccine and guidance for use in infants and children". MMWR Morb. Mortal. Wkly. Rep. 57 (39): 1079–80. October 2008. PMID 18830213. Archived from the original on 2016-11-07. Retrieved 2016-12-22.
- Baldo V, Bonanni P, Castro M, Gabutti G, Franco E, Marchetti F, Prato R, Vitale F (2014). "Combined hexavalent diphtheria-tetanus-acellular pertussis-hepatitis B-inactivated poliovirus-Haemophilus influenzae type B vaccine; Infanrix™ hexa: twelve years of experience in Italy". Hum Vaccin Immunother. 10 (1): 129–37. doi:10.4161/hv.26269. PMC 4181021. PMID 24004825.
- Talbot, Elizabeth A (2010). The Safety of Immunizing with Tetanus–diphtheria–acellular Pertussis Vaccine (Tdap) Less than 2 Years following Previous Tetanus Vaccination: Experience during a Mass Vaccination Campaign of Healthcare Personnel during a Respiratory Illness Outbreak. pp. 8001–7.
- Veronesi, R (1981). Tetanus: Important New Concepts. Amsterdam: Excerpta Medica.
- "Tdap (Tetanus, Diphtheria, Pertussis) Vaccine Information Statement". Centers for Disease Control and Prevention (CDC). 24 February 2015. Archived from the original on 28 December 2019. Retrieved 28 December 2019.
- "Update: vaccine side effects, adverse reactions, contraindications, and precautions. Recommendations of the Advisory Committee on Immunization Practices (ACIP)" (PDF). MMWR Recomm Rep. 45 (RR-12): 1–35. September 1996. PMID 8801442.
- "Vaccines & Immunizations". U.S. Centers for Disease Control and Prevention (CDC). Archived from the original on 2011-12-22. Retrieved 27 November 2011.
- "Whooping Cough: Vaccine Combined with Tetanus, Diphtheria". Archived from the original on June 6, 2017. Retrieved May 1, 2017.
Further reading
- Liang JL, Tiwari T, Moro P, Messonnier NE, Reingold A, Sawyer M, Clark TA (April 2018). "Prevention of Pertussis, Tetanus, and Diphtheria with Vaccines in the United States: Recommendations of the Advisory Committee on Immunization Practices (ACIP)" (PDF). MMWR Recomm Rep. 67 (2): 1–44. doi:10.15585/mmwr.rr6702a1. ISSN 1057-5987. PMC 5919600. PMID 29702631.
- Kahn KE, Black CL, Ding H, Williams WW, Lu PJ, Fiebelkorn AP, Havers F, D'Angelo DV, Ball S, Fink RV, Devlin R (September 2018). "Influenza and Tdap Vaccination Coverage Among Pregnant Women - United States, April 2018" (PDF). MMWR Morb. Mortal. Wkly. Rep. 67 (38): 1055–1059. doi:10.15585/mmwr.mm6738a3. PMC 6188122. PMID 30260946.
- World Health Organization (2018). The immunological basis for immunization series: module 3: tetanus: update 2018. World Health Organization (WHO). hdl:10665/275340. ISBN 9789241513616. License: CC BY-NC-SA 3.0 IGO.
- Ramsay M (ed.). "Chapter 30: Tetanus". Immunisation against infectious disease. Public Health England.
- Hamborsky J, Kroger A, Wolfe S, eds. (2015). "Chapter 21: Tetanus". Epidemiology and Prevention of Vaccine-Preventable Diseases (13th ed.). Washington D.C.: U.S. Centers for Disease Control and Prevention (CDC). ISBN 978-0990449119.
- Roush SW, Baldy LM, Hall MA, eds. (March 2019). "Chapter 16: Tetanus". Manual for the surveillance of vaccine-preventable diseases. Atlanta GA: U.S. Centers for Disease Control and Prevention (CDC).
External links
- "Td (Tetanus, Diphtheria) Vaccine Information Statement". Centers for Disease Control and Prevention (CDC). 11 April 2017.
- "DTaP (Diphtheria, Tetanus, Pertussis) Vaccine Information Statement". Centers for Disease Control and Prevention (CDC). 24 August 2018.
- "ACIP DTaP Vaccine Recommendations". Centers for Disease Control and Prevention (CDC).
- Tetanus Toxoid at the US National Library of Medicine Medical Subject Headings (MeSH)
- Diphtheria-Tetanus Vaccine at the US National Library of Medicine Medical Subject Headings (MeSH)
- Diphtheria-Tetanus-Pertussis Vaccine at the US National Library of Medicine Medical Subject Headings (MeSH)
- Diphtheria-Tetanus-acellular Pertussis Vaccines at the US National Library of Medicine Medical Subject Headings (MeSH)
| Wikimedia Commons has media related to Tetanus vaccination. |