Monarch butterfly
The monarch butterfly or simply monarch (Danaus plexippus) is a milkweed butterfly (subfamily Danainae) in the family Nymphalidae.[4] Other common names depending on region include milkweed, common tiger, wanderer, and black veined brown.[5] It may be the most familiar North American butterfly, and is considered an iconic pollinator species.[6] Its wings feature an easily recognizable black, orange, and white pattern, with a wingspan of 8.9–10.2 cm (3 1⁄2–4 in)[7] A Müllerian mimic, the viceroy butterfly is similar in color and pattern, but is markedly smaller and has an extra black stripe across each hindwing.
| Monarch butterfly | |
|---|---|
 | |
| Female | |
 | |
| Male | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Lepidoptera |
| Family: | Nymphalidae |
| Genus: | Danaus |
| Species: | D. plexippus |
| Binomial name | |
| Danaus plexippus | |
 | |
| Synonyms | |
The eastern North American monarch population is notable for its annual southward late-summer/autumn migration from the northern and central United States and southern Canada to Florida and Mexico.[4] During the fall migration, monarchs cover thousands of miles, with a corresponding multi-generational return north. The western North American population of monarchs west of the Rocky Mountains often migrates to sites in southern California but has been found in overwintering Mexican sites as well.[8][9] Monarchs have been bred on the International Space Station.[10]
Taxonomy

The name "monarch" is believed to be given in honor of King William III of England, whose secondary title Prince of Orange makes a reference to the butterfly's main color.[11] The monarch was originally described by Carl Linnaeus in his Systema Naturae of 1758 and placed in the genus Papilio.[12] In 1780, Jan Krzysztof Kluk used the monarch as the type species for a new genus Danaus.
Danaus (Ancient Greek Δαναός), a great-grandson of Zeus, was a mythical king in Egypt or Libya, who founded Argos; Plexippus (Πλήξιππος) was one of the 50 sons of Aegyptus, the twin brother of Danaus. In Homeric Greek, his name means "one who urges on horses", i.e., "rider" or "charioteer". In the 10th edition of Systema Naturae, at the bottom of page 467,[13] Linnaeus wrote that the names of the Danai festivi, the division of the genus to which Papilio plexippus belonged, were derived from the sons of Aegyptus. Linnaeus divided his large genus Papilio, containing all known butterfly species, into what we would now call subgenera. The Danai festivi formed one of the "subgenera", containing colorful species, as opposed to the Danai candidi, containing species with bright white wings. Linnaeus wrote: "Danaorum Candidorum nomina a filiabus Danai Aegypti, Festivorum a filiis mutuatus sunt." (English: "The names of the Danai candidi have been derived from the daughters of Danaus, those of the Danai festivi from the sons of Aegyptus.")
Robert Michael Pyle suggested Danaus is a masculinized version of Danaë (Greek Δανάη), Danaus's great-great-granddaughter, to whom Zeus came as a shower of gold, which seemed to him a more appropriate source for the name of this butterfly.[14]
There are three species of monarch butterflies:
- D. plexippus, described by Linnaeus in 1758, is the species known most commonly as the monarch butterfly of North America. Its range actually extends worldwide and can be found in Hawaii, Australia, New Zealand, Spain and the Pacific Islands.
- D. erippus, the southern monarch, was described by Pieter Cramer in 1775. This species is found in tropical and subtropical latitudes of South America, mainly in Brazil, Uruguay, Paraguay, Argentina, Bolivia, Chile and southern Peru. The South American monarch and the North American monarch may have been one species at one time. Some researchers believe the southern monarch separated from the monarch's population some 2 mya, at the end of the Pliocene. Sea levels were higher, and the entire Amazonas lowland was a vast expanse of brackish swamp that offered limited butterfly habitat.[15]
- D. cleophile, the Jamaican monarch, described by Jean Baptiste Godart in 1819, ranges from Jamaica to Hispaniola.[16]
Six subspecies and two color morphs of D. plexippus have been identified:[5]
- D. p. plexippus – nominate subspecies, described by Linnaeus in 1758, is the migratory subspecies known from most of North America.
- D. p. nigrippus (Richard Haensch, 1909) – as forma: Danais [sic] archippus f. nigrippus. Hay-Roe et al. in 2007 identified this taxon as a subspecies:[19]
- D. p. megalippe (Jacob Hübner, [1826]) – nonmigratory subspecies, and is found from Florida and Georgia southwards, throughout the Caribbean and Central America to the Amazon River.
- D. p. leucogyne (Arthur G. Butler, 1884) − St. Thomas
- D. p. portoricensis Austin Hobart Clark, 1941 − Puerto Rico
- D. p. tobagi Austin Hobart Clark, 1941 − Tobago
The percentage of the white morph in Oahu is nearing 10%. On other Hawaiian islands, the white morph occurs at a relatively low frequency. White monarchs (nivosus) have been found throughout the world, including Australia, New Zealand, Indonesia, and the United States.[17]
Some taxonomists disagree on these classifications.[15][19]
Monarchs belong in the subfamily Danainae of the family Nymphalidae; Danainae was formerly considered a separately family Danaidae.[20]
A 2015 paper identified genes from wasp bracoviruses in the genome of the North American monarch[21] leading to articles about monarch butterflies being genetically modified organisms.[22][23]
Description

Commonly and easily mistaken for the similar viceroy butterfly – the two species are Müllerian mimics, the monarch's wingspan ranges from 8.9 to 10.2 centimetres (3.5–4.0 in).[7] The uppersides of the wings are tawny orange, the veins and margins are black, and there are two series of small white spots in the margins. Monarch forewings also have a few orange spots near their tips. Wing undersides are similar, but the tips of forewings and hindwings are yellow brown instead of tawny orange and the white spots are larger.[24] The shape and color of the wings change at the beginning of the migration and appear redder and more elongated than later migrants.[25] Wings size and shape differ between migratory and non-migratory monarchs. Monarchs from eastern North America have larger and more angular forewings than those in the western population.[10]
Monarch flight has been described as "slow and sailing".[26] Monarch flight speed has been estimated by a number of researchers. One scientist examined all prior estimates and concluded their flight speed is approximately 9 km/h or 5.5 mph.[27] For comparison, the average human jogs at a rate of 9.7–12.9 km/h (6–8 mph).
Adults are sexually dimorphic. Males are slightly larger than females[10][24] and have a black patch or spot of androconial scales on each hindwing (in some butterflies, these patches disperse pheromones, but are not known to do so in monarchs). The male's black wing veins are lighter and narrower than those of females.[28]
One variation, the "white monarch", observed in Australia, New Zealand, Indonesia and the United States, is called nivosus by lepidopterists. It is grayish white in all areas of its wings that are normally orange and is only about 1% or less of all monarchs, but populations as high as 10% exist on Oahu in Hawaii.[17]
The monarch has six legs like all insects, but uses only its middle legs and hindlegs as the forelegs are vestigial, as in all Nymphalidae, and held against its body.[29]

Detailed measurements
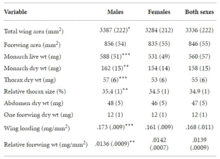
A study in 2015 examined a preserved collection of male and female monarch specimens from eastern North America to evaluate the sex-based differences in fine-scale wing and body structure.[30] The study found significant differences in overall wing size and in the physical dimensions of wings. Males tended to have larger wings than females, and were heavier than females, on average. Both males and females had similar thorax dimensions (wing muscles are contained in the thorax). Female monarchs tended to have thicker wings, which is thought to convey greater tensile strength. This would make female wings less likely to be damaged during migration. Also, females had lower wing loading than males (wing loading is a value derived from the ratio of wing size to body mass), which would mean females require less energy to fly.[31]
Distribution and habitat

The range of the western and eastern populations of D. plexippus plexippus expands and contracts depending upon the season. The range differs between breeding areas, migration routes, and winter roosts.[10]:(p18) However, no genetic differences between the western and eastern monarch populations exist;[32] reproductive isolation has not led to subspeciation of these populations, as it has elsewhere within the species' range.[10]:(p19)
In the Americas, the monarch ranges from southern Canada through northern South America.[4] It has also been found in Bermuda, Cook Islands,[33] Hawaii,[34][35] Cuba,[36] and other Caribbean islands[10]:(p18) the Solomons, New Caledonia, New Zealand,[37] Papua New Guinea,[38] Australia, the Azores, the Canary Islands, Madeira, Gibraltar,[39] the Philippines, and North Africa.[40] It appears in the UK in some years as an accidental migrant.[41]
Overwintering populations of D. plexippus plexippus are found in Mexico, California, along the Gulf Coast, year round in Florida, and in Arizona where the habitat has the specific conditions necessary for their survival.[42][43] On the US East Coast, they have overwintered as far north as Lago Mar, Virginia Beach, Virginia.[44] Their wintering habitat typically provides access to streams, plenty of sunlight (enabling body temperatures that allow flight), and appropriate roosting vegetation, and is relatively free of predators. Overwintering, roosting butterflies have been seen on basswoods, elms, sumacs, locusts, oaks, osage-oranges, mulberries, pecans, willows, cottonwoods, and mesquites.[45] While breeding, monarch habitats can be found in agricultural fields, pasture land, prairie remnants, urban and suburban residential areas, gardens, trees, and roadsides – anywhere where there is access to larval host plants.[46] Habitat restoration is a primary goal in monarch conservation efforts. Habitat requirements change during migration. During the fall migration, butterflies must have access to nectar-producing plants. During the spring migration, butterflies must have access to larval food plants and nectar plants.
Life cycle
The monarch butterfly undergoes four stages of complete metamorphosis:
Eggs
The eggs are derived from materials ingested as a larva and from the spermatophores received from males during mating.[47] Eggs are laid singly on the underside of a young leaf of a milkweed plant during the spring and summer months.[48] The eggs are cream colored or light green, ovate to conical in shape, and about 1.2×0.9 mm in size. The eggs weigh less than 0.5 mg each and have raised ridges that form longitudinally from the point to apex to the base. Though each egg is 1⁄1000 the mass of the female, she may lay up to her own mass in eggs. Females lay smaller eggs as they age. Larger females lay larger eggs.[47] The number of eggs laid by a female, who may mate several times, ranges from 290 to 1180.[49] Females lay their eggs on the underside of the milkweed leaves; the offspring's consumption of the milkweed benefits health and helps defend them against predators.[50][51] Eggs take 3 to 8 days to develop and hatch into larva or caterpillars.[10]:(p21) Monarchs will lay eggs along the southern migration route.[52]
Larvae
The caterpillar goes through five major, distinct stages of growth and after each one, it molts. Each caterpillar, or instar, that molts is larger than the previous as it eats and stores energy in the form of fat and nutrients to carry it through the nonfeeding pupal stage. Each instar usually lasts about 3 to 5 days, depending on various factors such as temperature and food availability.[4]

The first instar caterpillar that emerges out of the egg is pale green and translucent. It lacks banding coloration or tentacles. The larvae or caterpillar eats its egg case and begins to feed on milkweed. It is during this stage of growth that the caterpillar begins to sequester cardenolides. The circular motion a caterpillar uses while eating milkweed prevents the flow of latex that could entrap it. The first instar is usually between 2 and 6S mm long.
The second instar larva develops a characteristic pattern of white, yellow and black transverse bands. It is no longer translucent but is covered in short setae. Pairs of black tentacles begin to grow. One pair grows on the thorax and another pair on the abdomen. Like the first instar, second instar larvae usually eat holes in the middle of the leaf, rather than at the edges. The second instar is usually between 6 mm and 1 cm long.
The third instar larva has more distinct bands and the two pairs of tentacles become longer. Legs on the thorax differentiate into a smaller pair near the head and larger pairs further back. These third-stage caterpillars begin to eat along the leaf edges. The third instar is usually between 1 and 1.5 cm long.
The fourth instar has a different banding pattern. It develops white spots on the prolegs near the back of the caterpillar. It is usually between 1.5 and 2.5 cm long.
The fifth instar larva has a more complex banding pattern and white dots on the prolegs, with front legs that are small and very close to the head. A caterpillar at this stage has an enormous appetite, being able to consume a large milkweed leaf in a day. Its length ranges from 2.5 to 4.5 cm.[4]
.jpg)
At this stage of development, it is relatively large compared to the earlier instars. The caterpillar completes its growth. At this point, it is 4.5 cm long (large specimens can reach 5 cm) and 7 to 8 mm wide, and weighs about 1.5 grams. This can be compared to the first instar, which was 2 to 6 mm long and 0.5 to 1.5 mm wide. Fifth-instar larvae increase in weight 2000 times from first instars. Fifth-stage instar larva can chew through the petiole or midrib of milkweed leaves and stop the flow of latex. After this, they eat more leaf tissue. Before pupation, larvae must consume milkweed to increase their mass. Larvae stop feeding and search for a pupation site.
Pupa

To prepare for the pupa or chrysalis stage, the caterpillar chooses a safe place for pupation, where it spins a silk pad on a downward-facing horizontal surface. At this point, it turns around and securely latches on with its last pair of hindlegs and hangs upside down, in the form of the letter J. After "J-hanging" for about 12–16 hours, it will suddenly straighten out its body and go into peristalsis some seconds before its skin splits behind its head. It then sheds its skin over a period of a few minutes, revealing a green chrysalis. At first, the chrysalis is long, soft, and somewhat amorphous, but over a few hours it compacts into its distinct shape – an opaque, pale-green chrysalis with small golden dots near the bottom, and a gold-and-black rim around the dorsal side near the top. At first, its exoskeleton is soft and fragile, but it hardens and becomes more durable within about a day. At this point, it is about 2.5 cm (1") long and 10–12 mm (3/8–7/16") wide, weighing about 1.2 grams. At normal summer temperatures, it matures in 8–15 days (usually 11–12 days). During this pupal stage, the adult butterfly forms inside. Within a day or so before emerging is due, the exoskeleton first becomes translucent and the chrysalis more bluish. Finally, within 12 hours or so, it becomes transparent, revealing the black and orange colors of the butterfly inside before it ecloses (emerges).
Adult
An adult butterfly emerges after about two weeks as a chrysalis, and hangs upside down for a few hours until its wings are dry. Fluids are pumped into the wings, and they expand, dry, and stiffen. The monarch expands and retracts its wings, and once conditions allow, it then flies and feeds on a variety of nectar plants. During the breeding season adults reach sexual maturity in four or five days. However, the migrating generation does not reach maturity until overwintering is complete.[53] Monarchs typically live for two to five weeks during their breeding season.[10]:(pp22-23) Larvae growing in high densities are smaller, have lower survival, and weigh less as adults compared with those growing in lower densities.[54] Monarch metamorphosis from egg to adult occurs during the warm summer temperatures in as little as 25 days, extending to as many as seven weeks during cool spring conditions. During the development, both larvae and their milkweed hosts are vulnerable to weather extremes, predators, parasites and diseases; commonly fewer than 10% of monarch eggs and caterpillars survive.[10]:(pp21-22) However, this is a natural attrition rate for most butterflies, since they are low on the food chain.
Reproduction
Healthy males are more likely to mate than unhealthy ones. Females and males typically mate more than once. Females that mate several times lay more eggs.[55] Mating for the overwintering populations occurs in the spring, prior to dispersion. Mating is less dependent on pheromones than other species in its genus.[56] Male search and capture strategies may influence copulatory success, and human-induced changes to the habitat can influence monarch mating activity at overwintering sites.[57]
Courtship occurs in two phases. During the aerial phase, a male pursues and often forces a female to the ground. During the ground phase, the butterflies copulate and remain attached for about 30 to 60 minutes.[58] Only 30% of mating attempts end in copulation, suggesting that females may be able to avoid mating, though some have more success than others.[59][60] During copulation, a male transfers his spermatophore to a female. Along with sperm, the spermatophore provides a female with nutrition, which aids her in egg laying. An increase in spermatophore size increases the fecundity of female monarchs. Males that produce larger spermatophores also fertilize more females' eggs.[61]
Pictorial life cycle
 Egg
Egg.jpg) Caterpillar
Caterpillar Feeding on milkweed
Feeding on milkweed Beginning pupation
Beginning pupation Chrysalis
Chrysalis- Emerging from chrysalis
 Mating
Mating Laying eggs
Laying eggs
Larval host plants
The host plants used by the monarch caterpillar include:

- Asclepias angustifolia – Arizona milkweed[62]
- Asclepias asperula – antelope horns milkweed[62]
- Asclepias californica – California milkweed[62]
- Asclepias cordifolia – heartleaf milkweed[62]
- Asclepias curassavica
- Asclepias eriocarpa – woolly pod milkweed[62]
- Asclepias erosa – desert milkweed[62]
- Asclepias exaltata – poke milkweed[62]
- Asclepias fascicularis – Mexican whorled milkweed[62]
- Asclepias humistrata – sandhill/pinewoods milkweed[62]
- Asclepias incarnata – swamp milkweed[62]
- Asclepias nivea – Caribbean milkweed[63]
- Asclepias oenotheroide – zizotes milkweed[62]
- Asclepias perennis – aquatic milkweed[62]
- Asclepias speciosa – showy milkweed[62]
- Asclepias subulata – rush milkweed[62]
- Asclepias syriaca – common milkweed.[62][64]
- Asclepias tuberosa – butterfly weed[62]
- Asclepias variegata – white milkweed[62]
- Asclepias verticillata – whorled milkweed[62]
- Asclepias vestita – woolly milkweed[62]
- Asclepias viridis – green antelopehorn milkweed[62]
- Calotropis gigantea – crown flower[65]
- Calotropis procera
- Cynanchum laeve – sand vine milkweed[66]
- Sarcostemma clausa – white vine[34][67]
Asclepias curassavica, or tropical milkweed, is often planted as an ornamental in butterfly gardens. Year-round plantings in the USA are controversial and criticised, as they may be the cause of new overwintering sites along the U.S. Gulf Coast, leading to year-round breeding of monarchs.[68] This is thought to adversely affect migration patterns, and to cause a dramatic buildup of the dangerous parasite, Ophryocystis elektroscirrha.[69] New research also has shown that monarch larvae reared on tropical milkweed show reduced migratory development (reproductive diapause), and when migratory adults are exposed to tropical milkweed, it stimulates reproductive tissue growth.[70]
Monarch caterpillars do not favor butterfly weed (Asclepias tuberosa) because the leaves of that milkweed species contain little toxin (cardiac glycosides).[71] Some other milkweeds may have similar characteristics.
Adult food sources

Although larvae eat only milkweed, adult monarchs feed on the nectar of many plants including:
- Apocynum cannabinum – Indian hemp
- Asclepias sp. – milkweeds
- Aster sp. – asters
- Cirsium sp. – thistles
- Daucus carota – wild carrot
- Dipsacus sylvestris – teasel
- Echinacea sp. – coneflowers
- Erigeron canadensis – horseweed
- Eupatorium maculatum – spotted Joe-Pye weed
- Eupatorium perfoliatum – common boneset
- Hesperis matronalis – dame's rocket
- Liatris sp. – blazing stars
- Medicago sativa – alfalfa
- Solidago sp. – goldenrod
- Syringa vulgaris – lilac
- Trifolium pratense – red clover
- Vernonia altissima – tall ironweed[43]
Monarchs obtain moisture and minerals from damp soil and wet gravel, a behavior known as mud-puddling. The monarch has also been noticed puddling at an oil stain on pavement.[43]
Migration
In North America, monarchs migrate both north and south on an annual basis, in a long-distance journey that is fraught with risks.[4] The population east of the Rocky Mountains attempts to migrate to the sanctuaries of the Mariposa Monarca Biosphere Reserve in Mexico and parts of Florida. The western population tries to reach overwintering destinations in various coastal sites in central and southern California. The overwintered population of those east of the Rockies may reach as far north as Texas and Oklahoma during the spring migration. The second, third and fourth generations return to their northern locations in the United States and Canada in the spring.[72] Captive-raised monarchs appear capable of migrating to overwintering sites in Mexico,[73] though they have a much lower migratory success rate than wild monarchs do.[74] See section on captive-rearing below. Recent discoveries have located monarch overwintering sites in Arizona.[75]
Vision
Physiological experiments suggest that monarch butterflies view the world through a tetrachromatic system.[76] Like humans, their retina contain three types of opsin proteins, expressed in distinct photoreceptor cells, each of which absorbs light at a different wavelength. Unlike humans, one of those types of photoreceptor cells corresponds to a wavelength in the ultraviolet range; the other two correspond to blue and green.[77] In addition to these three photoreceptors cells in the main retina, monarch butterfly eyes contain orange filtering pigments that filter the light reaching some but not all green-absorbing opsins, thereby making a fourth photoreceptor cell sensitive to longer wavelength light.[76] The combination of filtered and unfiltered green opsins permits the butterflies to distinguish yellow from orange colors.[76] The ultraviolet opsin protein has also been detected in the dorsal rim region of monarch eyes. One study suggests that this allows the butterflies the ability to detect ultraviolet polarized skylight in order to orient themselves with the sun for their long migratory flight.[78]
These butterflies are capable of distinguishing colors based on their wavelength only, and not based on intensity; this phenomenon is termed “true color vision.” This is important for many butterfly behaviors, including seeking nectar for nourishment, choosing a mate, and finding milkweed to lay eggs on. One study found that floral color is more easily recognized at a distance by butterflies searching for nectar than floral shape. This is may be because flowers have highly contrasting colors to the green background of a vegetative landscape.[79] On the other hand, leaf shape is important for oviposition so that the butterflies can ensure their eggs are being laid on milkweed.
Beyond the perception of color, the ability to remember certain colors is essential in the life of monarch butterflies. Researchers have found that these insects can easily learn to associate color and, to a lesser extent shape, with sugary food rewards. When searching for nectar, color is the first cue that draws the insect’s attention toward a potential food source, and shape is a secondary characteristic that promotes the process. When searching for a place to lay one’s eggs, the roles of color and shape are switched. There may also be a difference between male and female butterflies from other species in terms of the ability to learn certain colors; however, there is no differences between the sexes for monarch butterflies.[79]
Defense against predators
In both caterpillar and butterfly form, monarchs are aposematic—warding off predators with a bright display of contrasting colors to warn potential predators of their undesirable taste and poisonous characteristics.

Large larvae are able to avoid wasp predation by dropping from the plant or by jerking their bodies.[80]
Aposematism
Monarchs are foul tasting and poisonous due to the presence of cardenolides in their bodies, which the caterpillars ingest as they feed on milkweed.[56] Monarchs and other cardenolide resistant insects rely on a resistant form of the Na+/ K+-ATPase enzyme to tolerate significantly higher concentrations of cardenolides than nonresistant species.[81] By ingesting a large amount of plants in the genus Asclepias, primarily milkweed, monarch caterpillars are able to sequester cardiac glycosides, or more specifically cardenolides, which are steroids that act in heart-arresting ways similar to digitalis.[82] It has been found that monarchs are able to sequester cardenolides most effectively from plants of intermediate cardenolide content rather than those of high or low content.[83]
Additional studies have shown that different species of milkweed have different effects on growth, virulence, and transmission of parasites.[84] One species, Asclepias curassavica, appears to reduce the symptoms of Ophryocystis elektroscirrha (OE) infection. There are two possible explanations for this: that it promotes overall monarch health to boost the monarch's immune system; or that chemicals from the plant have a direct negative effect on the OE parasites.[84] A. curassavica does not cure or prevent the infection with OE, it merely allows infected monarchs to live longer, and this would allow infected monarchs to spread the OE spores for longer periods. For the average home butterfly garden, this scenario will only add more OE to the local population.[85]

After the caterpillar becomes a butterfly, the toxins shift to different parts of the body. Since many birds attack the wings of the butterfly, having three times the cardiac glycosides in the wings leaves predators with a very foul taste and may prevent them from ever ingesting the body of the butterfly.[82] In order to combat predators that remove the wings only to ingest the abdomen, monarchs keep the most potent cardiac glycosides in their abdomens.[86]
Mimicry
Monarchs share the defense of noxious taste with the similar-appearing viceroy butterfly in what is perhaps one of the most well-known examples of mimicry. Though long purported to be an example of Batesian mimicry, the viceroy is actually reportedly more unpalatable than the monarch, making this a case of Müllerian mimicry.[87]
Human interaction
The monarch is the state insect of Alabama,[88] Idaho,[89] Illinois,[90] Minnesota,[91] Texas,[92] Vermont,[93] and West Virginia.[94] Legislation was introduced to make it the national insect of the United States,[95] but this failed in 1989[96] and again in 1991.[97]
A growing number of homeowners are establishing butterfly gardens; monarchs can be attracted by cultivating a butterfly garden with specific milkweed species and nectar plants. Efforts are underway to establish these monarch waystations. [98]
An IMAX film, Flight of the Butterflies, describes the story of the Urquharts, Brugger and Trail to document the then unknown monarch migration to Mexican overwintering areas.[99]
Sanctuaries and reserves have been created at overwintering locations in Mexico and California to limit habitat destruction. These sites can generate significant tourism revenue.[100]
Organizations and individuals participate in tagging programs. Tagging information is used to study migration patterns.[101]
The 2012 novel by Barbara Kingsolver, Flight Behavior, deals with the appearance of a large population in the Appalachians.[102]
Captive rearing
One of the most direct ways humans are interacting with monarchs is by rearing them in captivity, which has become increasingly popular, although there are risks to this activity, and this has become a controversial topic. On one hand there are many positive aspects of captive rearing. Monarchs are bred in schools and used for butterfly releases at hospices, memorial events and weddings.[103] Memorial services for the September 11 attacks include the release of captive-bred monarchs.[104][105][106] Monarchs are used in schools and nature centers for educational purposes.[107] Many homeowners raise monarchs in captivity as a hobby and for educational purposes.[108]
On the other hand this practice becomes problematic when monarchs are "mass-reared". Stories in the Huffington Post in 2015 and Discover magazine in 2016 have summarized the controversy around this issue.[109][110] The frequent media reports of monarch declines has empowered many homeowners to attempt to rear as many monarchs as possible in their homes and then release them to the wild in an effort to "boost the monarch population". Some individuals have taken this practice to the extreme, with massive operations that rear thousands of monarchs at once, like one in Linn County, Iowa.[111] However, the practice of rearing "large" numbers of monarchs in captivity for release into the wild is not condoned by monarch scientists, because of the risks of genetic issues and disease spread.[112] One of the biggest concerns of mass-rearing is the potential for spreading the monarch parasite, Ophryocystis elektroscirrha, into the wild. This parasite can rapidly build up in captive monarchs, especially if they are housed together. The spores of the parasite also can quickly contaminate all housing equipment, so that all subsequent monarchs reared in the same containers then become infected. One researcher stated that rearing more than 100 monarchs constitutes "mass-rearing" and should not be done.[113]
In addition to the disease risks, researchers believe these captive-reared monarchs are not as fit as wild ones, owing to the unnatural conditions they are raised in. Homeowners often raise monarchs in plastic or glass containers in their kitchens, basements, porches, etc., and under artificial lighting and controlled temperatures. Such conditions would not mimic what the monarchs are used to in the wild, and may result in adult monarchs that are unsuited for the realities of their wild existence. In support of this, a recent study by a citizen scientist found that captive-reared monarchs have a lower migration success rate than wild monarchs do.[74]
A study published in 2019 shed light on the fitness of captive-reared monarchs, by testing reared and wild monarchs on a tethered flight apparatus that assessed navigational ability.[114] In that study, monarchs that were reared to adulthood in artificial conditions showed a reduction in navigational ability. This happened even with monarchs that were brought into captivity from the wild for a few days. A few captive-reared monarchs did show proper navigation. This study revealed the fragility of monarch development - if the conditions are not suitable, their ability to properly migrate could be impaired. The same study also examined the genetics of a collection of reared monarchs purchased from a butterfly breeder, and found they were dramatically different from wild monarchs, so much so that the lead author described them as "franken-monarchs."[115]
An unpublished study in 2019 compared behavior of captive-reared versus wild monarch larvae.[116] The study showed that reared larvae exhibited more defensive behavior than wild larvae. The reason for this is unknown, but it could relate to the fact that reared larvae are frequently handled and/or disturbed.
Genome
The monarch was the first butterfly to have its genome sequenced.[10]:(p12) The 273-million base pair draft sequence includes a set of 16,866 protein-coding genes. The genome provides researchers insights into migratory behavior, the circadian clock, juvenile hormone pathways and microRNAs that are differentially expressed between summer and migratory monarchs.[117][118][119] More recently, the genetic basis of monarch migration and warning coloration has been described.[120]
There is no genetic differentiation between the migratory populations of eastern and western North America.[10]:(p16) Recent research has identified the specific areas in the genome of the monarch that regulate migration. There appears to be no genetic difference between a migrating and nonmigrating monarch but the gene is expressed in migrating monarchs but not expressed in nonmigrating monarchs.[32]
Conservation status
The monarch butterfly is not currently listed under the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) or protected specifically under U.S. domestic laws.[121] On 14 August 2014, the Center for Biological Diversity and the Center for Food Safety filed a legal petition requesting Endangered Species Act protection for the monarch and its habitat,[10] based largely on the long-term trends observed at overwintering sites. The U.S. Fish and Wildlife Service initiated a status review of the monarch butterfly under the Endangered Species Act with a due date for information submission of 3 March 2015. The decision on whether to list the monarch is still pending, and a new deadline for completion of an internal FWS species status report is December 2020.
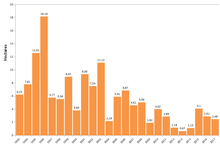
The number of monarchs overwintering in Mexico has shown a long-term downward trend. Since 1995, coverage numbers have been as high as 18 hectares (44 acres) during the winter of 1996–1997, but on average about 6 hectares (15 acres). Coverage declined to its lowest point to date (0.67 hectares (1.66 acres)) during the winter of 2013–2014, but rebounded to 4.01 hectares (10 acres) in 2015–2016. The average population of monarchs in 2016 was estimated at 200 million. Historically, on average there are 300 million monarchs. The 2016 increase was attributed to favorable breeding conditions in the summer of 2015. However, coverage declined by 27% to 2.91 hectares (7.19 acres) during the winter of 2016–2017. Some believe this was because of a storm that had occurred during March 2016 in the monarchs' previous overwintering season,[122] though this seems unlikely since most current research shows that the overwintering colony sizes do not predict the size of the next summer breeding population.[123]
A study in 2016 claimed that the long-term trend in the size of the overwintering sites is cause for concern. After a ten-fold drop in the overwintering numbers of the eastern monarch butterfly population over the last decade, this study claimed there was an 11%–57% probability that this population will go quasi-extinct over the next 20 years.[124] According to Xerces Society, the monarch population in California decreased 86 percent in 2018, going from millions of butterflies to tens of thousands of butterflies.[125]
In Ontario, Canada, the monarch butterfly is listed as a Species of Special Concern.[126] In fall 2016, the Committee on the Status of Endangered Wildlife in Canada proposed that the monarch be listed as endangered in Canada, as opposed to its current listing as a "species of concern" in that country. This move, once enacted, would protect critical monarch habitat in Canada, such as major fall accumulation areas in southern Ontario, but it would also have implications for citizen scientists who work with monarchs, and for classroom activities. If the monarch were federally protected in Canada, these activities could be limited, or require federal permits.[127] In Nova Scotia, the monarch is listed as "Endangered" at the provincial level, as of 2017. This decision (as well as the Ontario decision) appears to be because of the presumption that the overwintering colony declines in Mexico translate into declines in the breeding range in Canada.[128] Two recent studies have been conducted examining long-term trends in monarch abundance in Canada, using either butterfly atlas records[129] or citizen science butterfly surveys,[130] and neither shows evidence of a population decline in Canada.
Threats
There is increasing concern related to the ongoing decline of monarchs at their overwintering sites; based on a 2014 twenty-year comparison, the overwintering numbers west of the Rocky Mountains have dropped more than 50 percent since 1997 and the overwintering numbers east of the Rockies have declined by more than 90 percent since 1995.[10]
In February 2015, the U.S. Fish and Wildlife Service provided a statistic showing that nearly a billion monarchs have vanished from the overwintering sites since 1990. At that time, one of the main reasons cited was the herbicides used by farmers and homeowners on milkweed, a plant used as a food source, a home and a nursery by the monarchs.[131] A 2016 study also attributed the last decade's ten-fold decline in overwintering numbers of the eastern monarch population to the loss of breeding habitat, namely the many species of milkweed (Asclepias species) that developing larvae require for food; however, scientists believe there are other factors as well. A number of researchers believe milkweed loss during the breeding season is the cause because declines in milkweed abundance are highly correlated with the adoption of herbicide-tolerant genetically modified corn and soybeans, which now constitute 89% and 94% of these crops, respectively, in the U.S.[124] However, correlative evidence does not prove causation, and other possible causes of the overwintering declines have been proposed. A 2018 study has suggested that the decline in milkweed predates the arrival of GM crops.[132]
Habitat loss due to herbicide use
A number of conservationists attribute the disappearance of milkweed species to agricultural practices in the Midwest, where genetically modified seeds are bred to resist herbicides that eliminate milkweed nearby. Growers eliminate milkweed that previously grew between the rows of food crops. Corn and soybeans are resistant to the effect of the herbicide glyphosate. The increased use of these crop strains is correlated with the decline in monarch populations between 1999 and 2010.[133][134] Chip Taylor, director of Monarch Watch at the University of Kansas, said the Midwest milkweed habitat "is virtually gone" with 120–150 million acres lost.[135][136] To help fight this problem, Monarch Watch encourages the planting of "Monarch Waystations".[108] The Natural Resources Defense Council filed a suit in 2015 against the EPA, in which it is argued that the agency ignored warnings about the dangers of glyphosate usage for monarchs.[137]
Losses during migration
While herbicide-use has been proposed as one factor causing the decline in overwintering numbers of eastern monarchs, it is not the only possibility. Another is that the monarchs are experiencing problems reaching Mexico. This idea has been embraced by a number of leading monarch researchers, largely because of recent evidence showing that the number of breeding (adult) monarchs has not declined in the last two decades, based on long-term citizen science data.[138][139][140] The lack of long-term declines in the numbers of breeding, and migratory monarchs, yet the clear declines in overwintering numbers, implies there is a disconnect between these life stages, that must be growing. One expert has proposed that a large and growing threat to migrating monarchs is mortality from car strikes.[141] New research published in 2019 appears to be bearing this out - a study of road mortality in northern Mexico showed shockingly high mortality from just two "hotspots" each year, amounting to 200,000 monarchs killed.[142]
Predators
While monarchs have a wide range of natural predators, none of these are suspected of causing harm to the overall population, or are the cause of the long-term declines in winter colony sizes.
Larvae feed exclusively on milkweed and consume protective cardiac glycosides. Toxin levels in Asclepias species vary. Not all monarchs are unpalatable, but exhibit Batesian or automimics. Cardiac glycosides levels are higher in the abdomen and wings. Some predators can differentiate between these parts and consume the most palatable ones.[143] Bird predators include brown thrashers, grackles, robins, cardinals, sparrows, scrub jays, pinyon jays,[143] black-headed grosbeak, and orioles.[34]
Several species of birds have acquired methods that allow them to ingest monarchs without experiencing the ill effects associated with the cardiac glycosides. The oriole is able to eat the monarch through an exaptation of its feeding behavior that gives it the ability to identify cardenolides by taste and reject them.[144] The grosbeak, on the other hand, has developed an insensitivity to secondary plant poisons that allows it to ingest monarchs without vomiting. As a result, orioles and grosbeaks will periodically have high levels of cardenolides in their bodies, and they will be forced to go on periods of reduced monarch consumption. This cycle effectively reduces potential predation of monarchs by 50 percent and indicates that monarch aposematism has a legitimate purpose.[144]
Some mice are able to withstand large doses of the toxin. Overwintering adults become less toxic over time making them more vulnerable to predators. In Mexico, about 14% of the overwintering monarchs are eaten by birds and mice.[42]
In North America, eggs and first-instar larvae of the monarch are eaten by larvae and adults of the introduced Asian lady beetle (Harmonia axyridis).[145] The Chinese mantis (Tenodera sinensis) will consume the larvae once the gut is removed thus avoiding cardenolides.[146] Wasps commonly consume larvae.[147]
One monarch researcher emphasizes that predation on eggs, larvae or adults is natural, since monarchs are part of the food chain, thus people should not take steps to kill predators of monarchs.[148]
On Oahu, a white morph of the monarch has emerged. This is because of the introduction, in 1965 and 1966, of two bulbul species, Pycnonotus cafer and Pycnonotus jocosus. They are now the most common insectivore birds, and probably the only ones preying on insects as large as the monarch. Monarchs in Hawaii are known to have low cardiac glycoside levels, but the birds may also be tolerant of the chemical. The two species hunt the larvae and some pupae from the branches and undersides of leaves in milkweed bushes. The bulbuls also eat resting and ovipositing adults, but rarely flying ones. Because of its color, the white morph has a higher survival rate than the orange one. This is either because of apostatic selection (i.e., the birds have learned the orange monarchs can be eaten), because of camouflage (the white morph matches the white pubescence of milkweed or the patches of light shining through foliage), or because the white morph does not fit the bird's search image of a typical monarch, so is thus avoided.[149]
Parasites

Parasites include the tachinid flies Sturmia convergens[150] and Lespesia archippivora. Lesperia-parasitized butterfly larvae suspend, but die prior to pupation. The fly's maggot lowers itself to the ground, forms a brown puparium and then emerges as an adult.[151]
Monarch chrysalises are parasitized by pteromalid wasps, specifically Pteromalus cassotis.[152] These wasps lay their eggs in the pupae while the chrysalis is still soft. Up to 400 adults emerge from the chrysalis after 14–20 days,[152] killing the monarch.
The bacterium Micrococcus flacidifex danai also infects larvae. Just before pupation, the larvae migrate to a horizontal surface and die a few hours later, attached only by one pair of prolegs, with the thorax and abdomen hanging limp. The body turns black shortly after. The bacterium Pseudomonas aeruginosa has no invasive powers, but causes secondary infections in weakened insects. It is a common cause of death in laboratory-reared insects.[151]
The protozoan Ophryocystis elektroscirrha is another parasite of the monarch. It infects the subcutaneous tissues and propagates by spores formed during the pupal stage. The spores are found over all of the body of infected butterflies, with the greatest number on the abdomen. These spores are passed, from female to caterpillar, when spores rub off during egg laying and are then ingested by caterpillars. Severely infected individuals are weak, unable to expand their wings, or unable to eclose, and have shortened lifespans, but parasite levels vary in populations. This is not the case in laboratory rearing, where after a few generations, all individuals can be infected.[153] Infection with this parasite creates an effect known as culling whereby migrating monarchs that are infected are less likely to complete the migration. This results in overwintering populations with lower parasite loads.[154] Owners of commercial butterfly breeding operations claim that they take steps to control this parasite in their practices,[155] although this claim is doubted by most scientists who study monarchs.
Confusion of host plants
The black swallow-wort (Cynanchum louiseae) and pale swallow-wort (Cynanchum rossicum) plants are problematic for monarchs in North America. Monarchs lay their eggs on these relatives of native vining milkweed (Cynanchum laeve) because they produce stimuli similar to milkweed. Once the eggs hatch, the caterpillars are poisoned by the toxicity of this invasive plant from Europe.[156]
Loss of overwintering habitat
The area of forest occupied has been declining and reached its lowest level in two decades in 2013. The decline is continuing but is expected to increase during the 2013–2014 season. Mexican environmental authorities continue to monitor illegal logging of the oyamel trees. The oyamel is a major species of evergreen on which the overwintering butterflies spend a significant time during their winter diapause, or suspended development.[157]
A 2014 study acknowledged that while "the protection of overwintering habitat has no doubt gone a long way towards conserving monarchs that breed throughout eastern North America", their research indicates that habitat loss on breeding grounds in the United States is the main cause of both recent and projected population declines.[158]
Climate
Climate variations during the fall and summer affect butterfly reproduction. Rainfall, and freezing temperatures affect milkweed growth. Omar Vidal, director general of WWF-Mexico, said "The monarch's lifecycle depends on the climatic conditions in the places where they breed. Eggs, larvae and pupae develop more quickly in milder conditions. Temperatures above 95°F can be lethal for larvae, and eggs dry out in hot, arid conditions, causing a drastic decrease in hatch rate."[159] If a monarch's body temperatures is below 86 °F a monarch cannot fly. To warm up they will sit in the sun or rapidly shiver their wings to warm themselves.[160]
There is concern that climate change will dramatically affect the monarch migration. A study from 2015 examined the impact of warming temperatures on the breeding range of the monarch, and showed that in the next 50 years the monarch host plant will expand its range further north into Canada, and that the monarchs will follow this.[161] While this will expand the breeding locations of the monarch, this will also have the effect of increasing the distance that monarchs must travel to reach their overwintering destination in Mexico, and this could result in greater mortality during the migration.[162]
Milkweeds grown at increased temperatures have been shown to contain higher cardenolide concentrations making the leaves too toxic for the monarch caterpillars, but these increased concentrations are likely in response to increased insect herbivory which is also caused by the increased temperatures, so it is unknown whether increased temperatures in isolation will make milkweed too toxic for monarch caterpillars.[163] Additionally, milkweed grown at carbon dioxide levels of 760 parts per million (ppm) plants were found to produce a different mix of the toxic cardenolides, one that was less effective against monarch parasites.[164]
Conservation efforts
Although numbers of breeding monarchs in eastern North America have apparently not decreased, reports of declining numbers of overwintering butterflies have inspired efforts to conserve the species.[138][139][140] Because of concerns over the overwintering numbers, the Center for Biological Diversity, the Center for Food Safety, the Xerces Society and Lincoln Brower have filed a petition to the United States Department of the Interior to protect the monarch by having it federally protected.[10]
On 20 June 2014, President Barack Obama issued a presidential memorandum entitled "Creating a Federal Strategy to Promote the Health of Honey Bees and Other Pollinators". The memorandum established a Pollinator Health Task Force, to be co-chaired by the Secretary of Agriculture and the Administrator of the Environmental Protection Agency, and stated:
The number of migrating Monarch butterflies sank to the lowest recorded population level in 2013–14, and there is an imminent risk of failed migration.[165]
In May 2015, the Pollinator Health Task Force issued a "National Strategy to Promote the Health of Honey Bees and Other Pollinators". The strategy lays out current and planned federal actions to achieve three goals, two of which are:
• Monarch Butterflies: Increase the Eastern population of the monarch butterfly to 225 million butterflies occupying an area of approximately 15 acres (6 hectares) in the overwintering grounds in Mexico, through domestic/international actions and public-private partnerships, by 2020.
• Pollinator Habitat Acreage: Restore or enhance 7 million acres of land for pollinators over the next 5 years through Federal actions and public/private partnerships.
Many of the priority projects that the national strategy identifies will focus on the I-35 corridor extending for 1,500 miles (2,400 km) from Texas to Minnesota that provides spring and summer breeding habitats in the monarch's key migration corridor.[166]
There have been a number of national and local efforts underway to establish pollinator habitat along highways and roadways, although this effort is controversial. Conservationists are lobbying transportation departments and utilities to reduce their use of herbicides and specifically encourage milkweed to grow along roadways and power lines. Reducing roadside mowing and application of herbicides during the butterfly breeding season will encourage milkweed growth.[167] Conservationists lobby agriculture companies to set aside areas that remain unsprayed to allow the butterflies to breed.[134] This practice is controversial because of the high risk of butterfly mortality near roads, as several studies have shown that millions of monarchs and other butterflies are killed by cars every year.[141] There is also evidence that monarch larvae living near roads experience physiological stress conditions, as evidenced by elevations in their heart rate.[168]
A 2020 resource from the Cooperative Research Programs of the Transportation Research Board developed products for roadway corridors to provide habitat for monarch butterflies and developed tools for roadside managers to optimize potential habitat for monarch butterflies in their road right-of-ways.[169]
Butterfly gardening
While there are few scientific studies on the subject, the practice of butterfly gardening and creating "Monarch Waystations" is commonly thought to increase the populations of butterflies.[170] Efforts to increase monarch populations by establishing butterfly gardens and waystations require particular attention to the butterfly's food preferences and population cycles, as well to the conditions needed to propagate milkweed.
For example, in the Washington, D.C. area and elsewhere in the northeastern United States, monarchs prefer to reproduce on common milkweed (Asclepias syriaca), especially when its foliage is soft and fresh. Because monarch reproduction in that area peaks in late summer when milkweed foliage is old and tough, A. syriaca needs to be cut back in June - August to assure that it will be regrowing rapidly when monarch reproduction reaches its peak.[62][64][71]
Further, monarch caterpillars do not favor butterfly weed (Asclepias tuberosa) because the leaves of that milkweed species contain little toxin (cardiac glycosides).[71] In addition, milkweed seed may need a period of cold treatment (cold stratification) before it will germinate.[62][64]
See also
- Monarch butterfly migration
- Lepidoptera migration
- Monarch Butterfly Biosphere Reserve
- Peninsula Point Light, Michigan
- Butterfly house (conservatory)
- Lepidoptera
- Butterflies
References
- Monarch, NatureServe Explorer
- Committee On Generic Nomenclature, Royal Entomological Society of London (2007) [1934]. The Generic Names of British Insects. Royal Entomological Society of London Committee on Generic Nomenclature, Committee on Generic Nomenclature. British Museum (Natural History). Dept. of Entomology. p. 20.
- Scudder, Samuel H.; William M. Davis; Charles W. Woodworth; Leland O. Howard; Charles V. Riley; Samuel W. Williston (1989). The butterflies of the eastern United States and Canada with special reference to New England. The author. p. 721. ISBN 978-0-665-26322-4.
- Agrawal, Anurag (7 March 2017). Monarchs and Milkweed: A Migrating Butterfly, a Poisonous Plant, and Their Remarkable Story of Coevolution. Princeton University Press. ISBN 9781400884766.
- Danaus plexippus, funet.fi
- "Conserving Monarch Butterflies and their Habitats". USDA. 2015.
- Garber, Steven D. (1998). The Urban Naturalist. Courier Dover Publications. pp. 76–79. ISBN 978-0-486-40399-1.
- Groth, Jacob (10 November 2000). "Monarch Migration Study". Swallowtail Farms. Retrieved 21 July 2014.
- "Monarch Migration". Monarch Joint Venture. 2013.
- "Petition to protect the Monarch butterfly (Danaus plexippus plexippus) under the endangered species act" (PDF). Xerces Society. Retrieved 1 September 2014.
- Adams, Jean Ruth (1992). Insect Potpourri: Adventures in Entomology. CRC Press. pp. 28–29. ISBN 978-1-877743-09-2.
- Linnaeus, Carl (1758). Systema Naturae (in Latin). 1. Stockholm: Laurentius Salvius. p. 471. OCLC 174638949. Retrieved 5 June 2012.
- Linnaeus, C. (1758). Systema Naturae ed. X: 467 (in BHL)
- Pyle, Robert Michael (2001). Chasing Monarchs: Migrating with the Butterflies of Passage. Houghton Mifflin Books. pp. 148–149. ISBN 978-0-618-12743-6.
- Smith, David A.; Gugs Lushai and John A. Allen (2005). "A classification of Danaus butterflies (Lepidoptera: Nymphalidae) based upon data from morphology and DNA". Zoological Journal of the Linnean Society. 144 (2): 191–212. doi:10.1111/j.1096-3642.2005.00169.x.
- "IUCN Red List of Threatened Species, Version 2014.1". IUCN Red List of Threatened Species. Retrieved 13 July 2014.
- Gibbs, Lawrence; Taylor, O.R. (1998). "The White Monarch". Department of Entomology University of Kansas. Retrieved 17 July 2014.
- Groth, Jacob (10 November 2000). "Monarch Migration Study". Swallowtail Farms. Retrieved 22 July 2014.
- Hay-Roe, Miriam M.; Lamas, Gerardo; Nation, James L. (2007). "Pre- and postzygotic isolation and Haldane rule effects in reciprocal crosses of Danaus erippus and Danaus plexippus (Lepidoptera: Danainae), supported by differentiation of cuticular hydrocarbons, establish their status as separate species". Biological Journal of the Linnean Society. 91 (3): 445–453. doi:10.1111/j.1095-8312.2007.00809.x.
- Ackery, P. R.; Vaine-Wright, R. I. (1984). Milkweed butterflies, their cladistics and biology: being an account of the natural history of the Danainae, subfamily of the Lepidoptera, Nymphalidae. British Museum (Natural History), London. p. 17. ISBN 978-0-565-00893-2.
- Gasmi, Laila; Boulain, Helene; Gauthier, Jeremy; Hua-Van, Aurelie; Musset, Karine; Jakubowska, Agata K.; Aury, Jean-Marc; Volkoff, Anne-Nathalie; Huguet, Elisabeth (17 September 2015). "Recurrent Domestication by Lepidoptera of Genes from Their Parasites Mediated by Bracoviruses". PLOS Genet. 11 (9): e1005470. doi:10.1371/journal.pgen.1005470. PMC 4574769. PMID 26379286.
- Le Page, Michael (17 September 2015). "If viruses transfer wasp genes into butterflies, are they GM?". The New Scientist. Retrieved 13 July 2016.
- Main, Douglas (17 September 2015). "Wasps Have Genetically Modified Butterflies, Using Viruses". Newsweek. Retrieved 13 July 2016.
- Braby, Michael F. (2000). Butterflies of Australia: Their Identification, Biology and Distribution. CSIRO Publishing. pp. 597–599. ISBN 978-0-643-06591-8.
- Satterfield, Dara A.; Davis, Andrew K. (April 2014). "Variation in wing characteristics of monarch butterflies during migration: Earlier migrants have redder and more elongated wings". Animal Migration. 2 (1). doi:10.2478/ami-2014-0001.
- Klots, Alexander B. (1951). A Field Guide to the Butterflies of North America, East of the Great Plains (Tenth ed.). Boston: Houghton Mifflin Company. pp. 78, 79. ISBN 978-0395078655.
- "monarchscience". Akdavis6.wixsite.com. 31 December 2016. Retrieved 6 January 2017.
- "Monarch, Danaus plexippus". Archived from the original on 15 December 2012. Retrieved 27 August 2008.
- Darby, Gene (1958). What is a Butterfly. Chicago: Benefic Press. p. 10.
- Davis, A.K., and M. Holden. 2015. Measuring intraspecific variation in flight-related morphology of monarch butterflies (Danaus plexippus): which sex has the best flying gear? Journal of Insects Article ID 591705, 6 pages. https://www.hindawi.com/archive/2015/591705/
- "monarchscience". Akdavis6.wixsite.com. 3 November 2015.
- Zhan, Shuai; Merlin, Christine; Boore, Jeffrey L.; Reppert, Steven M. (23 November 2012). "The monarch butterfly genome yields insights into long-distance migration". Cell. 147 (5): 1171–1185. doi:10.1016/j.cell.2011.09.052. PMC 3225893. PMID 22118469.
- Gerald McCormack (7 December 2005). "Cook Islands' Largest Butterfly – the Monarch". Cook Islands Biodiversity.
- Scott, James A. (1986). The Butterflies of North America. Stanford University Press, Stanford, CA. ISBN 0-8047-2013-4
- Brower, Lincoln P.; Malcolm, Stephen B. (1991). "Animal Migrations: Endangered Phenomena". Am. Zool. 31 (1): 265–276. doi:10.1093/icb/31.1.265.
- Davis, Donald (27 November 2014). "DPLEX-L:59250 THE possibility of a trans-Gulf migration, oil rigs, Dr. Gary Ross, and more". Monarch Watch. University of Kansas.
- "Monarch Sightings Map". Monarch Butterfly New Zealand Trust. Retrieved 15 December 2014.
- "The lonely flight of the monarch butterfly". NewsAdvance.com, Lynchburg, Virginia Area. Retrieved 7 October 2014.
- Gibraltar Ornithological and Natural History Society "Provisional species list of the Lepidoptera" Archived 25 September 2015 at the Wayback Machine
- Pais, Miguel. "Northwestern African sightings of D. plexippus, Maroc>Pappilons>Danaus plexippus". Google Maps. Retrieved 19 September 2014.
- Coombes, Simon. "1995 Monarch Invasion of the UK". butterfly-guide.co.uk.
- Cech, Rick and Tudor, Guy (2005). Butterflies of the East Coast. Princeton University Press, Princeton, NJ. ISBN 0-691-09055-6
- Iftner, David C.; Shuey, John A. and Calhoun, John C. (1992). Butterflies and Skippers of Ohio. College of Biological Sciences and The Ohio State University. ISBN 0-86727-107-8
- "Monarch Butterfly Journey North". Annenberg Learner. Archived from the original on 14 October 2017. Retrieved 16 July 2017.
- Pyle, Robert Michael (2014). Chasing monarchs: Migrating with the butterflies of passage. Yale University Press. p. 2. ISBN 978-0395828205.
- Halpern, Sue (2002). Four Wings and a Prayer. Kindle edition location 1594. New York, New York: Random House. ISBN 978-0-307-78720-0.CS1 maint: ref=harv (link)
- Oberhauser (2004), p. 3
- "Monarch Butterfly Life Cycle and Migration". National Geographic Education. 24 October 2008. Retrieved 15 August 2013.
- Oberhauser (2004), p. 23
- Lefevre, T.; Chiang, A.; Li, H; Li, J; de Castillejo, C.L.; Oliver, L.; Potini, Y.; Hunter, M.D.; de Roode, J.C. (2012). "Behavioral resistance against a protozoan parasite in the monarch butterfly" (PDF). Journal of Animal Ecology. 81 (1): 70–9. doi:10.1111/j.1365-2656.2011.01901.x. hdl:2027.42/89483. PMID 21939438.
- "The other butterfly effect – A youth reporter talks to Jaap de Roode". TED Blog. 25 November 2014. Retrieved 12 December 2014.
- Oberhauser (2004), p. 51
- "Reproduction". Monarch Lab. Regents of the University of Minnesota. Retrieved 13 December 2014.
- Flockhart, D. T. Tyler; Martin, Tara G.; Norris, D. Ryan (2012). "Experimental Examination of Intraspecific Density-Dependent Competition during the Breeding in Monarch Butterflies (Danaus plexippus)". PLOS One. 7 (9): e45080. Bibcode:2012PLoSO...745080F. doi:10.1371/journal.pone.0045080. PMC 3440312. PMID 22984614.
- Oberhauser, K.S. (1989). "Effects of spermatophores on male and female monarch butterfly reproductive success". Behav. Ecol. Sociobiol. 25 (4): 237–246. doi:10.1007/bf00300049.
- "ADW: Danaus plexippus: Information". Retrieved 27 August 2008.
- Solensky, Michelle J. (November 2004). "The Effect of Behavior and Ecology on Male Mating Success in Overwintering Monarch Butterflies (Danaus plexippus)". Journal of Insect Behavior. 17 (6): 723–743. doi:10.1023/b:joir.0000048985.58159.0d. ISSN 0892-7553.
- Emmel, Thomas C. (1997). Florida's Fabulous Butterflies. p. 44, World Publications, ISBN 0-911977-15-5
- Oberhauser (2004), pp. 61–68.
- Frey, D.; Leong, K.L.H.; Peffer, E.; Smidt, R.K.; Oberhauser, K.S. (1998). "Mating patterns of overwintering monarch butterflies (Danaus plexippus (L.)) in California" (PDF). J. Lepid. Soc. 52: 84–97.
- Solensky, M.J.; K.S. Oberhauser (2009). "Sperm Precedence in Monarch Butterflies (Danaus plexippus)". Behavioral Ecology. 20 (2): 328–34. doi:10.1093/beheco/arp003.
- "Plant Milkweed for Monarchs" (PDF). MONARCH JOINT VENTURE Partnering across the U.S. to conserve the monarch migration. Monarch Joint Venture. Archived from the original (PDF) on 21 May 2015. Retrieved 7 July 2015.
- "Asclepias nivea". butterfly gardening & all things milkweed. Archived from the original on 7 July 2015. Retrieved 7 July 2015.
- Higgins, Adrian (27 May 2015). "A gardener's guide to saving the monarch". Home & Garden. The Washington Post. Archived from the original on 29 May 2015. Retrieved 29 May 2015.
Higgins, Adrian (27 May 2015). "7 milkweed varieties and where to find them". Home & Garden. The Washington Post. Archived from the original on 29 May 2015. Retrieved 29 May 2015.
Gomez, Tony. "Asclepias Syriaca: Common Milkweed for Monarch Caterpillars". MonarchButterflyGarden.net. Archived from the original on 16 March 2015. Retrieved 29 May 2015.
"Common Milkweed: Asclepias syriaca L." (PDF). Plant Guide. United States Department of Agriculture: Natural Resources Conservation Service. Archived from the original (PDF) on 27 March 2015. Retrieved 29 May 2015.
"Asclepias syriaca". butterfly gardening & all things milkweed. Archived from the original on 7 July 2015. Retrieved 7 July 2015. - "Butterfly Society of Hawaii". Butterfly Society of Hawaii. Retrieved 6 January 2017.
- Butterfly Gardening. kansasnativeplants.com
- Wagner, David L. (2005). Caterpillars of Eastern North America. Princeton University Press, Princeton, NJ. ISBN 0-691-12144-3
- Howard, Elizabeth; Aschen, Harlen; Davis, Andrew K. (2010). "Citizen Science Observations of Monarch Butterfly Overwintering in the Southern United States". Psyche: A Journal of Entomology. 2010: 1. doi:10.1155/2010/689301.
- Satterfield, D. A.; Maerz, J. C.; Altizer, S (2015). "Loss of migratory behaviour increases infection risk for a butterfly host". Proceedings of the Royal Society B: Biological Sciences. 282 (1801): 20141734. doi:10.1098/rspb.2014.1734. PMC 4308991. PMID 25589600.
- Majewska, Ania A.; Altizer, Sonia (16 August 2019). "Exposure to Non-Native Tropical Milkweed Promotes Reproductive Development in Migratory Monarch Butterflies". Insects. 10 (8): 253. doi:10.3390/insects10080253. PMC 6724006. PMID 31426310.
- Abugattas, Alonzo (3 January 2017). "Monarch Way Stations". Capital Naturalist. Archived from the original on 5 June 2017. Retrieved 5 June 2017 – via Blogger.
- "North American Monarch Conservation Plan" (PDF). Commission for Environmental Cooperation. Retrieved 17 October 2014.
- Taylor, O.R. (28 June 1998), Monarch Watch 1999 Season Recoveries, pp. 1–11
- Steffy, Gayle (2015). "Trends Observed in Fall Migrant Monarch Butterflies (Lepidoptera: Nymphalidae) East of the Appalachian Mountains at an Inland Stopover in Southern Pennsylvania over an Eighteen Year Period". Annals of the Entomological Society of America. 108 (5): 718. doi:10.1093/aesa/sav046.
- "Monarch butterflies are a steady presence in Arizona". Arizona Daily Star. Retrieved 15 January 2016.
- Blackiston, Douglas; Briscoe, Adriana D.; Weiss, Martha R. (1 February 2011). "Color vision and learning in the monarch butterfly, Danaus plexippus (Nymphalidae)". The Journal of Experimental Biology. 214 (Pt 3): 509–520. doi:10.1242/jeb.048728. ISSN 1477-9145. PMID 21228210.
- Stalleicken, Julia; Labhart, Thomas; Mouritsen, Henrik (March 2006). "Physiological characterization of the compound eye in monarch butterflies with focus on the dorsal rim area". Journal of Comparative Physiology A. 192 (3): 321–331. doi:10.1007/s00359-005-0073-6. ISSN 0340-7594. PMID 16317560.
- Sauman, Ivo; Briscoe, Adriana D.; Zhu, Haisun; Shi, Dingding; Froy, Oren; Stalleicken, Julia; Yuan, Quan; Casselman, Amy; Reppert, Steven M. (5 May 2005). "Connecting the Navigational Clock to Sun Compass Input in Monarch Butterfly Brain". Neuron. 46 (3): 457–467. doi:10.1016/j.neuron.2005.03.014. ISSN 0896-6273. PMID 15882645.
- Cepero, Laurel C.; Rosenwald, Laura C.; Weiss, Martha R. (1 July 2015). "The Relative Importance of Flower Color and Shape for the Foraging Monarch Butterfly (Lepidoptera: Nymphalidae)". Journal of Insect Behavior. 28 (4): 499–511. doi:10.1007/s10905-015-9519-z. ISSN 1572-8889.
- Oberhauser (2004), p. 44
- Karageorgi, Marianthi; Groen, Simon C.; Sumbul, Fidan; Pelaez, Julianne N.; Verster, Kirsten I.; Aguilar, Jessica M.; Hastings, Amy P.; Bernstein, Susan L.; Matsunaga, Teruyuki; Astourian, Michael; Guerra, Geno (October 2019). "Genome editing retraces the evolution of toxin resistance in the monarch butterfly". Nature. 574 (7778): 409–412. doi:10.1038/s41586-019-1610-8. ISSN 1476-4687. PMC 7039281. PMID 31578524.
- Parsons, J.A. (1965). "A Digitallis-like Toxin in the Monarch Butterfly Danaus plexippus L". J. Physiol. 178 (2): 290–304. doi:10.1113/jphysiol.1965.sp007628. PMC 1357291. PMID 14298120.
- Malcolm, S.B.; L.P. Brower (1989). "Evolutionary and ecological implications of cardenolide sequestration in the monarch butterfly". Experientia. 45 (3): 284–295. doi:10.1007/BF01951814.
- de Rood, J.C.; De Castillejo, C. L.; Faits, T; Alizon, S (2011). "Virulence evolution in response to anti-infection resistance: toxic food plants can select for virulent parasites of monarch butterflies". Journal of Evolutionary Biology. 24 (4): 712–722. doi:10.1111/j.1420-9101.2010.02213.x. PMID 21261772.
- "Is tropical milkweed really medicinal? (answer: yes, and that's really really bad for your garden)". monarchscience. Retrieved 12 December 2019.
- Glazier, Lincoln; Susan Glazier (1975). "Localization of Heart Poisons in the Monarch Butterfly". Science. 188 (4183): 19–25. Bibcode:1975Sci...188...19B. doi:10.1126/science.188.4183.19. PMID 17760150.
- Ritland, D.; L. P. Brower (1991). "The viceroy butterfly is not a Batesian mimic". Nature. 350 (6318): 497–498. Bibcode:1991Natur.350..497R. doi:10.1038/350497a0.
Viceroys are as unpalatable as monarchs, and significantly more unpalatable than queens from representative Florida populations.
- "Official Alabama Insect". Alabama Emblems, Symbols and Honors. Alabama Department of Archives & History. 12 July 2001. Retrieved 19 March 2007.
- "Idaho Symbols, Insect: Monarch Butterfly". Idaho State Symbols, Emblems, and Mascots. SHG resources, state handbook & guide. Retrieved 26 March 2008.
- "State Symbol: Illinois Official Insect — Monarch Butterfly (Danaus plexippus)". [Illinois] State Symbols. Illinois State Museum. Retrieved 26 March 2008.
- "Minnesota State Symbols" (PDF). Minnesota House of Representatives. Retrieved 26 March 2008.
- "Texas State Symbols". The Texas State Library and Archives. Retrieved 26 March 2008.
- "(Vermont) State Butterfly". Vermont Department of Libraries. Archived from the original on 18 May 2008. Retrieved 26 March 2008.
- "West Virginia Statistical Information, General State Information" (PDF). Official West Virginia Web Portal. Archived from the original (PDF) on 11 April 2008. Retrieved 26 March 2008.
- Wade, Nicholas (1 June 1990). "Choosing a National Bug". The New York Times. Retrieved 27 September 2017.
- "Designating the monarch butterfly as the national insect. (1989 - H.J.Res. 411)". GovTrack.us.
- "Designating the monarch butterfly as the national insect. (1991 - H.J.Res. 200)". GovTrack.us.
- "Monarch Watch: Monarch Waystation Program". University of Kansas, Entomology Department. Archived from the original on 18 November 2019. Retrieved 26 February 2019.
- "Flight of the Butterflies". Reuben H. Fleet Science Center. Archived from the original on 17 February 2013. Retrieved 18 February 2013.
- "Saving Butterflies: Insect Ecologist Spearheads Creation of Oases for Endangered Butterflies". ScienceDaily. 1 January 2005. Archived from the original on 4 June 2008. Retrieved 27 May 2008.
- "Monarch Monitoring Project". Cap May Bird Observatory. 2008. Archived from the original on 20 December 2008. Retrieved 5 December 2008.
- HarperCollins Inc; UK, Faber and Faber
- "Live butterfly release for funerals and butterfly weddings". Frangrant Acres Butterfly Farm. Retrieved 14 November 2014.
- "In Memory of 9/11 "Wings of Hope"". gayandciha.com. 4 September 2014. Retrieved 14 November 2014.
- Johnson, Pam (24 August 2010). "Join Branford Rotary's 9/11 Town Green Event/Butterfly Release". Shure Publishing. The Day. Retrieved 14 November 2014.
- "Ormond Beach Flying US Flags On Granada Bridge T0 Mark 9\17". NewsDaytonaBeach.com. WNDB Local News First. 2013. Archived from the original on 29 November 2014. Retrieved 14 November 2014.
- "Monarch Butterfly release at Children's Museum of Fond du Lac". FDL Reporter. Retrieved 14 November 2014.
- "Monarch Watch". University of Kansas, Entomology Department. Archived from the original on 20 July 2012. Retrieved 20 February 2014.CS1 maint: unfit url (link)
- John Platt TakePart (14 October 2015). "When Butterflies Shouldn't Fly Free". The Huffington Post. Retrieved 12 January 2017.
- "Are We Loving Monarchs to Death? – The Crux". Blogs.discovermagazine.com. 21 June 2016. Retrieved 12 January 2017.
- Love, Orlan (25 August 2016). "Monarch Moonshot: Officials hope to make Linn County center of butterfly production and habitat". The Gazette. Retrieved 12 January 2017.
- Rearing Monarchs Responsibly: A conservationist’s guide to raising monarchs for science and education. Monarch Joint Venture, University of Minnesota
- "monarchscience". Akdavis6.wixsite.com. 7 September 2015.
- Tenger-Trolander, Ayşe; Lu, Wei; Noyes, Michelle; Kronforst, Marcus R. (16 July 2019). "Contemporary loss of migration in monarch butterflies". Proceedings of the National Academy of Sciences. 116 (29): 14671–14676. doi:10.1073/pnas.1904690116. ISSN 0027-8424. PMC 6642386. PMID 31235586.
- Maeckle |, Monika (2 July 2019). "Study of "genetic franken monarchs" provokes online ire and debate". texasbutterflyranch. Retrieved 12 December 2019.
- "Effects of captive-rearing on caterpillar anti-predator behavior - an inside look at some preliminary data". monarchscience. Retrieved 12 December 2019.
- Zhan, Shuai; Merlin, Christine; Boore, Jeffrey L.; Reppert, Steven M. (November 2011). "The Monarch Butterfly Genome Yields Insights into Long-Distance Migration". Cell. 147 (5): 1171–85. doi:10.1016/j.cell.2011.09.052. PMC 3225893. PMID 22118469.
- Stensmyr, Marcus C.; Hansson, Bill S. (November 2011). "A Genome Befitting a Monarch". Cell. 147 (5): 970–2. doi:10.1016/j.cell.2011.11.009. PMID 22118454.
- Johnson, Carolyn Y. (23 November 2011). "Monarch butterfly genome sequenced". Boston Globe. Boston, MA. Retrieved 9 January 2012.
- Zhan, Shuia; Zhang, Wei; Niitepold, Kristjan; Hsu, Jeremy; Haeger, Juan Fernandez; Zalucki, Myron P.; Altizer, Sonia; de Roode, Jacobus C.; Reppert, Stephen M.; Kronforst, Marcus R. (1 October 2014). "The genetics of Monarch butterfly migration and warning coloration". Nature. 514 (7522): 317–321. Bibcode:2014Natur.514..317Z. doi:10.1038/nature13812. PMC 4331202. PMID 25274300.
- "Monarch Butterfly". fws.gov.
- (1) Howard, Elizabeth (26 February 2016). "Monarch Population Size Announced". Journey North. Archived from the original on 28 February 2016. Retrieved 5 June 2017.
(2) "Eastern Monarch Population Numbers Drop 27%". News. The Monarch Joint Venture. 16 February 2017. Archived from the original on 5 June 2017. Retrieved 5 June 2017.
(3) "Where Are All The Monarchs?". Monarch Lab. University of Minnesota Extension. 14 July 2016. Archived from the original on 22 August 2016. Retrieved 5 June 2017. - "Another monarch study published showing the spring migration holds the key to everything". monarchscience. Retrieved 12 December 2019.
- Semmens, Brice X.; Semmens, Darius J.; Thogmartin, Wayne E.; Wiederholt, Ruscena; López-Hoffman, Laura; Diffendorfer, Jay E.; Pleasants, John M.; Oberhauser, Karen S.; Taylor, Orley R. (2016). "Quasi-extinction risk and population targets for the Eastern, migratory population of monarch butterflies (Danaus plexippus)". Scientific Reports. 6: 23265. Bibcode:2016NatSR...623265S. doi:10.1038/srep23265. PMC 4800428. PMID 26997124.
- "The Monarch butterfly population in California has plummeted 86% in one year".
- "Monarch". Government of Ontario. Retrieved 16 September 2015.
- "monarchscience". Akdavis6.wixsite.com. 18 December 2016. Retrieved 12 January 2017.
- "Monarchs now listed as an endangered species in Nova Scotia Canada - a prelude for things to come elsewhere". monarchscience. Retrieved 12 December 2019.
- Crewe, Tara L.; Mitchell, Greg W.; Larrivée, Maxim (2019). "Size of the Canadian Breeding Population of Monarch Butterflies Is Driven by Factors Acting During Spring Migration and Recolonization". Frontiers in Ecology and Evolution. 7. doi:10.3389/fevo.2019.00308. ISSN 2296-701X.
- Flockhart, D. T. Tyler; Larrivée, Maxim; Prudic, Kathleen L.; Norris, D. Ryan (21 June 2019). "Estimating the annual distribution of monarch butterflies in Canada over 16 years using citizen science data". FACETS. 4: 238–253. doi:10.1139/facets-2018-0011.
- Fears, Darryl (26 August 2015). "As pesticides wipe out Monarch butterflies in the U.S., illegal logging is doing the same in Mexico". The Washington Post.
- Puzey, J. R.; Dalgleish, H. J.; Boyle, J. H. (5 February 2019). "Monarch butterfly and milkweed declines substantially predate the use of genetically modified crops". Proceedings of the National Academy of Sciences. 116 (8): 3006–3011. doi:10.1073/pnas.1811437116. ISSN 0027-8424. PMC 6386695. PMID 30723147.
- Pleasants, John M.; Oberhauser, Karen S. (2012). "Milkweed loss in agricultural fields because of herbicide use: effect on the monarch butterfly population" (PDF). Insect Conservation and Diversity. 6 (2): 135–144. doi:10.1111/j.1752-4598.2012.00196.x. Archived from the original (PDF) on 4 September 2014.
- Brennen, Shannon. "For Love of Nature: Annual monarch butterfly migration in peril". The News & Advance, Lynchburg, Virginia. Retrieved 4 July 2014.
- Conniff, Richard (1 April 2013). "Tracking the Causes of Sharp Decline of the Monarch Butterfly". Yale University.
- Wines, Michael (13 March 2013) Monarch Migration Plunges to Lowest Level in Decades. The New York Times
- "NRDC Sues EPA Over Demise of Monarch Butterfly Population". NBC. 2015.
- Ries, Leslie; Taron, Douglas J.; Rendón-Salinas, Eduardo (2015). "The Disconnect Between Summer and Winter Monarch Trends for the Eastern Migratory Population: Possible Links to Differing Drivers". Annals of the Entomological Society of America. 108 (5): 691. doi:10.1093/aesa/sav055.
- Inamine, Hidetoshi; Ellner, Stephen P.; Springer, James P.; Agrawal, Anurag A. (2016). "Linking the continental migratory cycle of the monarch butterfly to understand its population decline". Oikos. 125 (8): 1081. doi:10.1111/oik.03196.
- Davis, Andrew K. (2012). "Are migratory monarchs really declining in eastern North America? Examining evidence from two fall census programs". Insect Conservation and Diversity. 5 (2): 101. doi:10.1111/j.1752-4598.2011.00158.x.
- "monarchscience". Akdavis6.wixsite.com. 24 August 2016. Retrieved 6 January 2017.
- Mora Alvarez, Blanca Xiomara; Carrera-Treviño, Rogelio; Hobson, Keith A. (2019). "Mortality of Monarch Butterflies (Danaus plexippus) at Two Highway Crossing "Hotspots" During Autumn Migration in Northeast Mexico". Frontiers in Ecology and Evolution. 7. doi:10.3389/fevo.2019.00273. ISSN 2296-701X.
- Barbosa, Pedro; Deborah Kay Letourneau (1988). "5". Novel Aspects of Insect-plant Interactions. Wiley-Interscience. pp. 29–31. ISBN 978-0-471-83276-8.
- Brower, Lincoln (1988). "Avian Predation on the Monarch Butterfly and Its Implications for Mimicry Theory". American Society of Naturalists. 131: S4–S6. doi:10.1086/284763.
- Koch, R. L.; W. D. Hutchison; R. C. Venette; G. E. Heimpel (October 2003). "Susceptibility of immature monarch butterfly, Danaus plexippus (Lepidoptera: Nymphalidae: Danainae), to predation by Harmonia axyridis (Coleoptera: Coccinellidae)". Biological Control. 28 (2): 265–270. doi:10.1016/S1049-9644(03)00102-6.
- Rafter, Jamie; Anurag Agruwal; Evan Preisser (2013). "Chinese mantids gut caterpillars: avoidance of prey defense?". Ecological Entomology. 38 (1): 78–82. doi:10.1111/j.1365-2311.2012.01408.x.
- Zalucki, Myron P.; Malcolm, Stephen B.; Paine, Timothy D.; Hanlon, Christopher C.; Brower, Lincoln P.; Clarke, Anthony R. (2001). "It's the first bites that count: Survival of first-instar monarchs on milkweeds". Austral Ecology. 26 (5): 547–555. doi:10.1046/j.1442-9993.2001.01132.x.
- "monarchscience". Akdavis6.wixsite.com. 5 April 2016.
- Stimson, John; Mark Berman (1990). "Predator induced colour polymorphism in Danaus plexippus L. (Lepidoptera: Nymphalidae) in Hawaii". Heredity. 65 (3): 401–406. doi:10.1038/hdy.1990.110. Lay summary.
- Clarke, A.R.; Zalucki, M.P. (2001). "Taeniogonalos raymenti Carmean & Kimsey (Hymenoptera: Trigonalidae) reared as a hyperparasite of Sturmia convergens (Weidemann) (Diptera: Tachinidae), a primary parasite of Danaus plexippus (L.) (Lepidoptera: Nymphalidae)". Pan-Pacific Entomologist. 77 (?): 68–70.
- Brewer, Jo; Gerard M. Thomas (1966). "Causes of death encountered during rearing of Danaus plexippus (Danaidae)" (PDF). Journal of the Lepidopterists' Society. 20 (4): 235–238. Archived from the original (PDF) on 25 March 2009. Retrieved 13 April 2008. Lay summary.
- Stenoien, Carl; McCoshum, Shaun; Caldwell, Wendy; De Anda, Alma; Oberhauser, Karen (January 2015). "New reports that Monarch butterflies (Lepidoptera: Nymphalidae, Linnaeus) are hosts for a pupal parasitoid (Hymenoptera: Chalcidoidea, Walker)". Journal of the Kansas Entomological Society. 88 (1): 16–26. doi:10.2317/JKES1402.22.1.
- Leong, K. L. H.; M. A. Yoshimura, H. K. Kaya and H. Williams (1997). "Instar susceptibility of the Monarch butterfly (Danaus plexippus) to the neogregarine parasite, Ophryocystis elektroscirrha". Journal of Invertebrate Pathology. 69 (1): 79–83. CiteSeerX 10.1.1.494.9827. doi:10.1006/jipa.1996.4634. PMID 9028932.
- Bartel, Rebecca; Oberhauser, Karen; De Roode, Jacob; Atizer, Sonya (February 2011). "Monarch butterfly migration and parasite transmission in eastern North America". Ecology. 92 (2): 342–351. doi:10.1890/10-0489.1. PMID 21618914.
- "Do My Monarch Butterflies Have OE? Ophryocystis elektroscirrha". Butterfly Fun Facts. Retrieved 24 March 2015.
- Invasive species alert: Black swallow-wort (Cynanchum louisea) and pale swallow-wort (Cynanchum rossicum). monarchjointventure.org]
- Paz, Fátima (18 June 2014) En espera de aprobación de la Profepa por tala ilegal en la Reserva de la Mariposa Monarca. cambiodemichoacan.com.mx
- "Habitat Loss on Breeding Grounds Cause of Monarch Decline, U of G Study Finds". University of Guelph. 4 June 2014. Retrieved 17 March 2015.
- Monarch Population Hits Lowest Point in More Than 20 Years. WWF (29 January 2014)
- "BASIC FACTS ABOUT MONARCH BUTTERFLIES".
- Lemoine, Nathan P. (2015). "Climate Change May Alter Breeding Ground Distributions of Eastern Migratory Monarchs (Danaus plexippus) via Range Expansion of Asclepias Host Plants". PLOS One. 10 (2): e0118614. Bibcode:2015PLoSO..1018614L. doi:10.1371/journal.pone.0118614. PMC 4338007. PMID 25705876.
- "monarchscience". Akdavis6.wixsite.com. 10 August 2015.
- Hahn, Philip G.; Agrawal, Anurag A.; Sussman, Kira I.; Maron, John L. (January 2019). "Population Variation, Environmental Gradients, and the Evolutionary Ecology of Plant Defense against Herbivory". The American Naturalist. 193 (1): 20–34. doi:10.1086/700838. ISSN 1537-5323. PMID 30624107.
- "Climate change, pesticides put monarch butterflies at risk of extinction". National Geographic. 21 December 2018. Retrieved 31 December 2018.
- Obama, President Barack (20 June 2014). "Presidential Memorandum – Creating a Federal Strategy to Promote the Health of Honey Bees and Other Pollinators". Office of the Press Secretary. Washington, D.C.: The White House. Retrieved 2 May 2018.
- Pollinator Health Task Force (19 May 2015). "National Strategy to Promote the Health of Honey Bees and Other Pollinators" (PDF). Washington, D.C.: The White House. Retrieved 2 May 2018.
- "Tennessee Makes Way for Monarchs". New York Times. Retrieved 16 September 2019.
- Davis, Andrew K.; Schroeder, Hayley; Yeager, Ian; Pearce, Jana (31 May 2018). "Effects of simulated highway noise on heart rates of larval monarch butterflies, Danaus plexippus: implications for roadside habitat suitability". Biology Letters. 14 (5): 20180018. doi:10.1098/rsbl.2018.0018. PMC 6012697. PMID 29743264.
- Read "Evaluating the Suitability of Roadway Corridors for Use by Monarch Butterflies" at NAP.edu.
- (1) Glassberg, J. (1995). Enjoying butterflies more: attract butterflies to your backyard. Marietta, Ohio: Bird Watcher's Digest Press. ISBN 1880241080. LCCN 96202681. OCLC 35808599. Retrieved 9 March 2020 – via Google Books.
(2) "Butterfly Gardening: Introduction". University of Kansas: Monarch Watch. Archived from the original on 2 February 2020. Retrieved 9 March 2020.
(3) "Monarch Garden Plants" (PDF). San Francisco, California: Pollinator Partnership. Archived from the original (PDF) on 9 March 2020. Retrieved 9 March 2020.
(4) "Monarch Waystation Program". University of Kansas: Monarch Watch. Archived from the original on 2 June 2017. Retrieved 5 June 2017.
External links
| Wikimedia Commons has media related to Danaus plexippus. |
| Wikispecies has information related to Danaus plexippus |
| Wikisource has original text related to this article: |
- Australian Museum fact sheet on Monarch butterflies
- Mission Monarch (Canada)
- Monarch Butterfly Metamorphosis: Time-lapse video (2:42 minutes)
- Monarch butterfly on the UF / IFAS Featured Creatures website
- Monarch Joint Venture – Partnering Across the U.S. to Conserve the Monarch Butterfly Migration
- Monarch Migration Maps
- Monarch Watch
- PBS NOVA Documentary "The Incredible Journey of the Butterflies"
- USGS description of Monarch butterfly
- Monarch Butterfly data at NatureServe Explorer
