Methylprednisolone
Methylprednisolone, sold under the brand name Depo-Medrol among others, is a corticosteroid medication used to suppress the immune system and decrease inflammation.[2] Conditions in which it is used include skin diseases, rheumatic disorders, allergies, asthma, croup, COPD, certain cancers, multiple sclerosis, and as add-on therapy for tuberculosis.[2] It is given by mouth, by injection into a vein or muscle, or applied to the skin.[2]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Medrol, Depo-Medrol, Solu-Medrol, others |
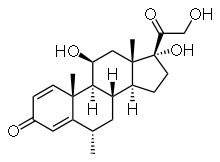
| Other names | (6α, 11β)-11,17,21-trihydroxy-6-methyl-pregna-1,4-diene-3,20-dione |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682795 |
| License data |
|
| Pregnancy category | |
| Routes of administration | IV, IM, by mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 78% |
| Metabolism | liver primarily, kidney, tissues; CYP450: 3A4 substrate |
| Elimination half-life | urine; Half-life: 18-26h (biological) |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.001.343 |
| Chemical and physical data | |
| Formula | C22H30O5 |
| Molar mass | 374.471 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
InChI
| |
| (verify) | |
Serious side effects may include mental health problems and an increased risk of infection.[2] Common side effects with long-term use include osteoporosis, cataracts, weakness, easy bruising, and yeast infections.[2] For pregnant women, short-term use around the time of delivery is safe for the baby; however, long-term use during pregnancy may result in harm.[1] Methylprednisolone is in the glucocorticoid family of medication.[2]
Methylprednisolone was approved for medical use in 1955.[3] It is on the World Health Organization's List of Essential Medicines, the safest and most effective medicines needed in a health system.[4] Methylprednisolone is available as a generic medication.[2] The wholesale cost in the developing world is about US$3.81–9.35 per 500 mg vial.[5] In the United States, a course of treatment typically costs less than US$25.[6] In 2017, it was the 157th most commonly prescribed medication in the United States, with more than four million prescriptions.[7][8]
Medical uses
.jpg)
Like most adrenocortical steroids, methylprednisolone is typically used for its anti-inflammatory effects. However, glucocorticoids have a wide range of effects, including changes to metabolism and immune responses. The list of medical conditions for which methylprednisolone is prescribed is rather long and is similar to other corticosteroids such as prednisolone. Common uses include arthritis therapy and short-term treatment of bronchial inflammation or acute bronchitis due to various respiratory diseases. It is used both in the treatment of acute periods and long-term management of autoimmune diseases, most notably systemic lupus erythematosus. It is also used as a treatment for multiple sclerosis, where it can favour recovery from acute attacks.[9]
Another potential use of methylprednisolone is for vestibular neuritis.[10]
After egg retrieval for a cycle of in vitro fertilization, methylprednisolone may be prescribed to prevent the body from rejecting the embryos being transferred, up to the time of implantation.[11][12]
Side effects
Long-term use of methylprednisolone, as with all corticosteroids, can be associated with hyperglycemia, decreased resistance to infection, swelling of face, weight gain, congestive cardiac insufficiency, fluid and sodium retention, edema, hypertension, increased eye pressure, glaucoma, osteoporosis, and psychosis, especially when used at high doses.[13][14] The most serious side effect occurs after the adrenal glands cease natural production of cortisol, which methylprednisolone will replace. Abrupt cessation of the drug after this occurs can result in a condition known as Addisonian crisis, which can be fatal. To prevent this, the drug is usually prescribed with a tapering dose, including a predosed "dose pack" detailing a specific number of tablets to take at designated times over a several-day period. Pharmacists sometimes advise that this drug may cause sleeplessness and "down" moods.
Measles and chicken pox are very dangerous and potentially fatal for people on methylprednisolone therapy. Exposure to these infections is especially risky for people who are not immune to them. Exposures like these should be reported to a physician immediately, and may be treated with prophylactic immunoglobulin. Also, live, attenuated vaccines can be bad for people taking immunosuppressive doses of methylprednisolone. The exception to this rule is patients receiving complete corticosteroid replacement therapy, e.g., for Addison's disease, who may follow standard immunization protocols.
Mechanism of action
Unbound glucocorticoids cross cell membranes and bind with high affinity to specific cytoplasmic receptors, modifying transcription and protein synthesis. By this mechanism, glucocorticoids can inhibit leukocyte infiltration at the site of inflammation, interfere with mediators of inflammatory response, and suppress humoral immune responses. The anti-inflammatory actions of corticosteroids are thought to involve phospholipase A2 inhibitory proteins, lipocortins, which control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes.
Formulations
Methylprednisolone acetate (Depo-Medrol) is a suspension administered by IM or intra-articular injection only.[15] It has the potential to cause subcutaneous atrophy in the area administered.[15] Due to the acetate in this formulation, it is important to remember that it should not be given intravenously.[15] The only formulation that can be given intravenously is methylprednisolone succinate (Solu-medrol).[13] Medrol dose packs are prepackaged forms of regular methylprednisolone for oral use.[14]
See also
- Guanacastepene A
- Dexamethasone
References
- "Methylprednisolone Use During Pregnancy". Drugs.com. 20 August 2019. Retrieved 20 February 2020.
- "Methylprednisolone". The American Society of Health-System Pharmacists. Archived from the original on 27 November 2016. Retrieved 8 December 2016.
- Fischer, Janos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 486. ISBN 978-3-527-60749-5. Archived from the original on 2016-12-22.
- World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- "Methylprednisolone". International Drug Price Indicator Guide. Retrieved 8 December 2016.
- Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 202. ISBN 978-1-284-05756-0.
- "The Top 300 of 2020". ClinCalc. Retrieved 11 April 2020.
- "Methylprednisolone - Drug Usage Statistics". ClinCalc. Retrieved 11 April 2020.
- Filippini, G.; Brusaferri, F.; Sibley, W. A.; Citterio, A.; Ciucci, G.; Midgard, R.; Candelise, L. (2000). "Corticosteroids or ACTH for acute exacerbations in multiple sclerosis". The Cochrane Database of Systematic Reviews (4): CD001331. doi:10.1002/14651858.CD001331. ISSN 1469-493X. PMID 11034713.
- Strupp M, Zingler VC, Arbusow V, Niklas D, Maag KP, Dieterich M, Bense S, Theil D, Jahn K, Brandt T (July 2004). "Methylprednisolone, valacyclovir, or the combination for vestibular neuritis". N. Engl. J. Med. 351 (4): 354–61. doi:10.1056/NEJMoa033280. PMID 15269315.
- "Medications Commonly Used During the IVF Cycle". Continuum Reproductive Center. Archived from the original on 6 September 2013. Retrieved 14 June 2013.
- "In Vitro Fertilization (IVF)". Reproductive Medicine Associates of Michigan. Archived from the original on 15 May 2013. Retrieved 14 June 2013.
- "Solu-medrol- methylprednisolone sodium succinate injection, powder, for solution Solu-medrol- methylprednisolone sodium succinate kit". DailyMed. 16 December 2019. Retrieved 19 February 2020.
- "Medrol- methylprednisolone tablet". DailyMed. 9 September 2019. Retrieved 19 February 2020.
- "Depo-Medrol- methylprednisolone acetate injection, suspension". DailyMed. 18 January 2019. Retrieved 19 February 2020.
External links
- "Methylprednisolone". Drug Information Portal. U.S. National Library of Medicine.
- "methylprednisolone" at medicinenet.com