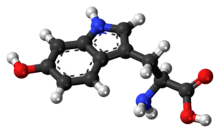
5-Hydroxytryptophan
5-Hydroxytryptophan (5-HTP), also known as oxitriptan, is a naturally occurring amino acid and chemical precursor as well as a metabolic intermediate in the biosynthesis of the neurotransmitter serotonin.
.svg.png) | |
 | |
| Names | |
|---|---|
| IUPAC name
2-amino-3-(5-hydroxy-1H-indol-3-yl)propanoic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.022.193 |
IUPHAR/BPS |
|
| KEGG | |
| MeSH | 5-Hydroxytryptophan |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
| C11H12N2O3 | |
| Molar mass | 220.228 g·mol−1 |
| Density | 1.484 g/mL |
| Melting point | 298 to 300 °C (568 to 572 °F; 571 to 573 K) |
| Boiling point | 520.6 °C (969.1 °F; 793.8 K) |
| Pharmacology | |
| N06AX01 (WHO) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Uses
5-HTP is sold over the counter in the United States, Canada, the Netherlands, and the United Kingdom as a dietary supplement for use as an antidepressant, appetite suppressant, and sleep aid. It is also marketed in many European countries for the indication of major depression under the trade names Cincofarm, Levothym, Levotonine, Oxyfan, Telesol, Tript-OH, and Triptum.[1]
A 2002 review concluded that although the data evaluated suggests that 5-HTP is more effective than placebo in the treatment of depression, the evidence was insufficient to be conclusive due to a lack of clinical data meeting the rigorous standards of today.[2] More and larger studies using current methodologies are needed to determine if 5-HTP is truly effective in treating depression.[3][4] In small controlled trials 5-HTP has also been reported to augment the antidepressant efficacy of the antidepressant clomipramine.[5][6][7]
5-HTP is sometimes taken by people coming down from MDMA to relieve post-MDMA dysphoria.[8] As 5-HTP is a necessary precursor for the brain to produce more serotonin, and MDMA use depletes a person's natural serotonin levels, it is believed that taking 5-HTP after consuming MDMA will speed up serotonin production. DanceSafe claims that the anecdotal evidence is widespread and that the theory is physiologically reasonable.[9] Backing up this approach is research conducted by Wang, et al. in 2007, which observed that MDMA-induced depletions of 5-HT (serotonin) were restored in rats after administration of 5-HTP, and suggested that this approach might be clinically useful in abstinent MDMA users.[8]
At high doses, or in combination with carbidopa, it has been used off-label to treat obesity (by promoting weight loss).[10][11]
In clinical trials of various design, 5-HTP has also been reported to treat fibromyalgia,[12] myoclonus,[13] migraine,[14] and cerebellar ataxia.[15] However, these clinical findings, as for all therapeutic findings with 5-HTP, are preliminary and need confirmation in larger trials.
5-HTP's drawbacks as a drug
The short half-life (<2h)[16] of 5-HTP may inherently limit the therapeutic potential of 5-HTP,[17] as the systemic 5-HTP exposure levels will fluctuate substantially, even with relatively frequent dosing. Such exposure fluctuations are usually associated with increased adverse event burden, resulting from Cmax drug spikes, and decreased clinical efficacy resulting from sub-therapeutic exposure for large parts of the day. It has been proposed that 5-HTP dosage forms achieving prolonged delivery would be more effective,[17] as is generally the situation with short-acting active pharmaceutical ingredients.[18]
Side Effects
Potential side effects of 5-HTP include heartburn, stomach pain, nausea, vomiting, diarrhea, drowsiness, sexual problems, vivid dreams or nightmares, and muscle problems.[19] Because 5-HTP has not been thoroughly studied in a clinical setting, possible side effects and interactions with other drugs are not well known. According to the US National Institute of Health TOXNET, 5-HTP has not been associated with serotonin syndrome or any serious adverse events in humans.[20] Across multiple studies, 5-HTP has also been reported to not cause any noticeable hematological or cardiovascular changes.[21] 5-HTP has also been associated with eosinophilia, but later studies have not found any causal connection.[22]
Interactions
When combined with antidepressants of the MAOI or SSRI class, very high parenteral doses of 5-HTP can cause acute serotonin syndrome in rats.[23][24] It is unclear if such findings have clinical relevance, as most drugs will cause serious adverse events or death in rodents at very high doses. In humans 5-HTP has never been clinically associated with serotonin syndrome, although a case report suggests 5-HTP can precipitate mania when added to an MAOI.[25]
When combined with carbidopa (as a treatment for symptoms of Parkinson's disease), 5-HTP causes nausea and vomiting; however this can be alleviated via administration of granisetron.[26] As mentioned below under pharmacology, cases of scleroderma-like illness have been reported in patients using carbidopa and 5-HTP.[27]
Oral 5-HTP results in an increase in urinary 5-HIAA, a serotonin metabolite, indicating that 5-HTP is peripherally metabolized to serotonin, which is then metabolized. This might cause a false positive test in tests looking for carcinoid syndrome.[28][29] Due to the conversion of 5-HTP into serotonin by the liver, there could be a risk of heart valve disease from serotonin's effect on the heart, as based on preclinical findings.[30][31] However, 5-HTP has not been associated with cardiac toxicity in humans.[22][21][20][32]
It has been suggested that 5-HTP may cause eosinophilia-myalgia syndrome (EMS), a serious condition which results in extreme muscle tenderness, myalgia, and blood abnormalities. However, there is evidence to show that EMS was likely caused by a contaminant in certain 5-HTP supplements.[33]
Production
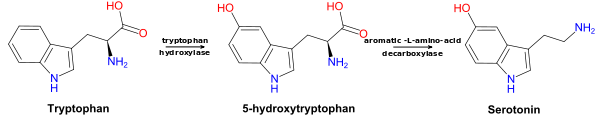
5-HTP is produced from the amino acid tryptophan through the action of the enzyme tryptophan hydroxylase. Tryptophan hydroxylase is one of the biopterin-dependent aromatic amino acid hydroxylases. Production of 5-HTP is the rate-limiting step in 5-HT synthesis. 5-HTP is normally rapidly converted to 5-HT by amino acid decarboxylase.[34]
Absorption
After oral administration, 5-HTP is absorbed by the upper intestine.[16] The mode of absorption is not known, but presumably involves active transport via amino acid transporters. 5-HTP is adequately absorbed via oral cavity.[35] With a decarboxylase inhibitor, the bioavailability of 5-HTP can be higher than 50%.[36]
Pharmacokinetics
5-HTP is rapidly absorbed with Tmax of ~1.5h, and rapidly eliminated with a half-life of ~1.5-2h. Co-administration of a decarboxylase inhibitor (e.g. carbidopa, benserazide) doubles the half-life of 5-HTP, to 3-4h,[37][16] and enhances exposure several fold, depending on the dosing regimen.[16][38]
Metabolism
5-HTP is decarboxylated to serotonin (5-hydroxytryptamine or 5-HT) by the enzyme aromatic-L-amino-acid decarboxylase with the help of vitamin B6.[39] This reaction occurs both in nervous tissue and in the liver.[40] 5-HTP crosses the blood–brain barrier,[41] while 5-HT does not. Excess 5-HTP, especially when administered with vitamin B6, is thought to be metabolized and excreted.[42][43]
| 5-HTP | AAAD | Serotonin | |
.svg.png) |
.svg.png) | ||
| PLP | |||
 | |||
Pharmacology
The psychoactive action of 5-HTP is derived from its increase in production of serotonin in central nervous system tissue.[44]
Research shows that co-administration with carbidopa greatly increases plasma 5-HTP levels.[45] Other studies have indicated the risk of a scleroderma-like condition resulting from the combination of 5-HTP and carbidopa.[46]
Regulatory Status
There are currently no approved drug products containing 5-HTP approved by the FDA.[47] All available 5-HTP products are nutraceuticals and are as such not regulated or verified for purity, integrity, or clinical efficacy or safety, mandating caution regarding human consumption.[48]
5-HTP Slow-Release
5-HTP's fast-in, fast-out pharmacokinetics is impractical for chronic drug therapy. Research conducted at Duke University in mice have demonstrated that 5-HTP when administered as slow-release appears to gain drug properties.[49] Slow-Release delivery attenuates or abolishes the peaks and valleys in 5-HTP exposure during treatment.[50] Slow-release delivery of 5-HTP markedly improved the safety profile of 5-HTP and conferred stable plasma exposure of 5-HTP and strong and sustained enhancement of brain serotonin function.[49] This discovery indicates that 5-HTP slow-release medications represent a new avenue for treatment of brain disorders responsive to serotonergic enhancement.
Dietary sources
Though 5-HTP is found in food only in insignificant quantities, it is a chemical involved intermediately in the metabolism of tryptophan, an amino acid found in all unfractionated foods, with lower total amino acid content correlating with increased tryptophan absorption.[51]
The seeds of the Griffonia simplicifolia, a climbing shrub native to West Africa and Central Africa, are used as an herbal supplement for their 5-hydroxytryptophan (5-HTP) content.[52][53][54] In one 2010 trial, Griffonia simplicifolia extract appeared to increase satiety in overweight women.[55]
See also
- Cardiac fibrosis
- Melatonin
- N-Acetylserotonin
- Selective serotonin reuptake inhibitor
- Serotonin
- Tryptophan
References
- Swiss Pharmaceutical Society (2000). Index Nominum 2000: International Drug Directory (Book with CD-ROM). Boca Raton: Medpharm Scientific Publishers. ISBN 978-3-88763-075-1.
- Shaw K, Turner J, Del Mar C (2002). Shaw KA (ed.). "Tryptophan and 5-hydroxytryptophan for depression" (PDF). The Cochrane Database of Systematic Reviews (1): CD003198. doi:10.1002/14651858.CD003198. PMID 11869656.
- 5-Hydroxytryptophan (5-HTP) University of Maryland Medical Center. 2011. Accessed: 9 January 2012.
- Iovieno N, Dalton ED, Fava M, Mischoulon D (May 2011). "Second-tier natural antidepressants: review and critique". Journal of Affective Disorders. 130 (3): 343–57. doi:10.1016/j.jad.2010.06.010. PMID 20579741.
- van Praag HM (1982). "Serotonin precursors in the treatment of depression". Advances in Biochemical Psychopharmacology. 34: 259–86. PMID 6753514.
- van Praag HM, van den Burg W, Bos ER, Dols LC (1974). "5-hydroxytryptophan in combination with clomipramine in "therapy-resistant" depressions". Psychopharmacologia. 38 (3): 267–9. doi:10.1007/BF00421379. PMID 4547418.
- Nardini M, De Stefano R, Iannuccelli M, Borghesi R, Battistini N (1983). "Treatment of depression with L-5-hydroxytryptophan combined with chlorimipramine, a double-blind study". International Journal of Clinical Pharmacology Research. 3 (4): 239–50. PMID 6381336.
- Wang X, Baumann MH, Dersch CM, Rothman RB (August 2007). "Restoration of 3,4-methylenedioxymethamphetamine-induced 5-HT depletion by the administration of L-5-hydroxytryptophan". Neuroscience. 148 (1): 212–20. doi:10.1016/j.neuroscience.2007.05.024. PMID 17629409.
- "Ecstasy and Depression". DanceSafe.Org. nd. Retrieved 21 September 2015.
- Halpern B, Oliveira ES, Faria AM, Halpern A, Melo ME, Cercato C, Mancini MC (July 2010). "Combinations of drugs in the Treatment of Obesity". Pharmaceuticals. 3 (8): 2398–2415. doi:10.3390/ph3082398. PMC 4033931. PMID 27713360.
- Hendricks EJ (2017). "Off-label drugs for weight management". Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy. 10: 223–234. doi:10.2147/DMSO.S95299. PMC 5473499. PMID 28652791.
- Caruso I, Sarzi Puttini P, Cazzola M, Azzolini V (1990). "Double-blind study of 5-hydroxytryptophan versus placebo in the treatment of primary fibromyalgia syndrome". J Int Med Res. 18 (3): 201–9. doi:10.1177/030006059001800304. PMID 2193835.
- Thal LJ, Sharpless NS, Wolfson L, Katzman R (1980). "Treatment of myoclonus with L-5-hydroxytryptophan and carbidopa: clinical, electrophysiological, and biochemical observations". Ann Neurol. 7: 570–6. doi:10.1002/ana.410070611. PMID 6969054.
- Boiardi A, Crenna P, Merati B, Negri S, Tansini E, Bussone G (1981). "5-OH-Tryptophane in migraine: clinical and neurophysiological considerations". J Neurol. 225 (1): 41–6. doi:10.1007/bf00313460. PMID 6164755.
- Trouillas P, Brudon F, Adeleine P (November 1988). "Improvement of cerebellar ataxia with levorotatory form of 5-hydroxytryptophan. A double-blind study with quantified data processing". Arch Neurol. 45 (11): 1217–22. doi:10.1001/archneur.1988.00520350055016. PMID 3190503.
- Gijsman HJ, van Gerven JM, de Kam ML, Schoemaker RC, Pieters MS, Weemaes M, de Rijk R, van der Post J, Cohen AF (April 2002). "Placebo-controlled comparison of three dose-regimens of 5-hydroxytryptophan challenge test in healthy volunteers". Journal of Clinical Psychopharmacology. 22 (2): 183–9. doi:10.1097/00004714-200204000-00012. PMID 11910264.
- Jacobsen JP, Krystal AD, Krishnan KR, Caron MG (November 2016). "Adjunctive 5-Hydroxytryptophan Slow-Release for Treatment-Resistant Depression: Clinical and Preclinical Rationale". Trends in Pharmacological Sciences. 37 (11): 933–944. doi:10.1016/j.tips.2016.09.001. PMC 5728156. PMID 27692695.
- Thombre AG (September 2005). "Assessment of the feasibility of oral controlled release in an exploratory development setting". Drug Discovery Today. 10 (17): 1159–66. doi:10.1016/S1359-6446(05)03551-8. PMID 16182208.
- "5-HTP". U.S. National Library of Medicine. Retrieved 7 June 2015.
- "5-HYDROXYTRYPTOPHAN - National Library of Medicine HSDB Database".
- Byerley WF, Judd LL, Reimherr FW, Grosser BI (June 1987). "5-Hydroxytryptophan: a review of its antidepressant efficacy and adverse effects". J Clin Psychopharmacol. 7 (3): 127–37. doi:10.1097/00004714-198706000-00002. PMID 3298325.
- Das YT, Bagchi M, Bagchi D, Preuss HG (2004). "Safety of 5-hydroxy-L-tryptophan". Toxicol Lett. 150 (1): 111–22. doi:10.1016/j.toxlet.2003.12.070. PMID 15068828.
- Ma Z, Zhang G, Jenney C, Krishnamoorthy S, Tao R (July 2008). "Characterization of serotonin-toxicity syndrome (toxidrome) elicited by 5-hydroxy-l-tryptophan in clorgyline-pretreated rats". European Journal of Pharmacology. 588 (2–3): 198–206. doi:10.1016/j.ejphar.2008.04.004. PMC 4242171. PMID 18499101.
- Izumi T, Iwamoto N, Kitaichi Y, Kato A, Inoue T, Koyama T (February 2006). "Effects of co-administration of a selective serotonin reuptake inhibitor and monoamine oxidase inhibitors on 5-HT-related behavior in rats". European Journal of Pharmacology. 532 (3): 258–64. doi:10.1016/j.ejphar.2005.12.075. PMID 16488409.
- Pardo JV (2012). "Mania following addition of hydroxytryptophan to monoamine oxidase inhibitor". General Hospital Psychiatry. 34 (1): 102.e13–4. doi:10.1016/j.genhosppsych.2011.08.014. PMC 3253963. PMID 21963353.
- Jacobs GE, Kamerling IM, de Kam ML, Derijk RH, van Pelt J, Zitman FG, van Gerven JM (January 2010). "Enhanced tolerability of the 5-hydroxytryptophane challenge test combined with granisetron". Journal of Psychopharmacology. 24 (1): 65–72. doi:10.1177/0269881108094299. PMID 18719048.
- "Carbidopa/Levodopa". Truestarhealth.com. Archived from the original on 8 January 2014. Retrieved 2014-01-09.
- Joy T, Walsh G, Tokmakejian S, Van Uum SH (January 2008). "Increase of urinary 5-hydroxyindoleacetic acid excretion but not serum chromogranin A following over-the-counter 5-hydroxytryptophan intake". Canadian Journal of Gastroenterology. 22 (1): 49–53. doi:10.1155/2008/472159. PMC 2659120. PMID 18209781.
- Hallin ML, Mahmoud K, Viswanath A, Gama R (January 2013). "'Sweet Dreams', 'Happy Days' and elevated 24-h urine 5-hydroxyindoleacetic acid excretion". Annals of Clinical Biochemistry. 50 (Pt 1): 80–2. doi:10.1258/acb.2012.012041. PMID 23086978.
- Gustafsson BI, Tømmerås K, Nordrum I, Loennechen JP, Brunsvik A, Solligård E, Fossmark R, Bakke I, Syversen U, Waldum H (March 2005). "Long-term serotonin administration induces heart valve disease in rats". Circulation. 111 (12): 1517–22. doi:10.1161/01.CIR.0000159356.42064.48. PMID 15781732.
- Xu J, Jian B, Chu R, Lu Z, Li Q, Dunlop J, Rosenzweig-Lipson S, McGonigle P, Levy RJ, Liang B (December 2002). "Serotonin mechanisms in heart valve disease II: the 5-HT2 receptor and its signaling pathway in aortic valve interstitial cells". The American Journal of Pathology. 161 (6): 2209–18. doi:10.1016/S0002-9440(10)64497-5. PMC 1850896. PMID 12466135.
- Turner EH, Loftis JM, Blackwell AD (March 2006). "Serotonin a la carte: supplementation with the serotonin precursor 5-hydroxytryptophan". Pharmacol Ther. 109 (3): 325–38. doi:10.1016/j.pharmthera.2005.06.004. PMID 16023217.
- Michelson D, Page SW, Casey R, Trucksess MW, Love LA, Milstien S, Wilson C, Massaquoi SG, Crofford LJ, Hallett M (December 1994). "An eosinophilia-myalgia syndrome related disorder associated with exposure to L-5-hydroxytryptophan". The Journal of Rheumatology. 21 (12): 2261–5. PMID 7699627.
- Turner EH, Loftis JM, Blackwell AD (March 2006). "Serotonin a la carte: supplementation with the serotonin precursor 5-hydroxytryptophan". Pharmacology & Therapeutics. 109 (3): 325–38. doi:10.1016/j.pharmthera.2005.06.004. PMID 16023217.
- Rondanelli M, Opizzi A, Faliva M, Bucci M, Perna S (March 2012). "Relationship between the absorption of 5-hydroxytryptophan from an integrated diet, by means of Griffonia simplicifolia extract, and the effect on satiety in overweight females after oral spray administration". Eat Weight Disord. 17 (1): e22-8. doi:10.3275/8165. PMID 22142813.
- Magnussen I, Nielsen-Kudsk F (April 1980). "Bioavailability and related pharmacokinetics in man of orally administered L-5-hydroxytryptophan in steady state". Acta Pharmacologica et Toxicologica. 46 (4): 257–62. doi:10.1111/j.1600-0773.1980.tb02451.x. PMID 6966118.
- Magnussen I, Engbaek F (July 1978). "The effects of aromatic amino acid decarboxylase inhibitors on plasma concentrations of 5-hydroxytryptophan in man". Acta Pharmacologica et Toxicologica. 43 (1): 36–42. doi:10.1111/j.1600-0773.1978.tb02229.x. PMID 309271.
- Westenberg HG, Gerritsen TW, Meijer BA, van Praag HM (December 1982). "Kinetics of l-5-hydroxytryptophan in healthy subjects". Psychiatry Research. 7 (3): 373–85. doi:10.1016/0165-1781(82)90074-9. PMID 6187038.
- Rahman MK, Nagatsu T, Sakurai T, Hori S, Abe M, Matsuda M (October 1982). "Effect of pyridoxal phosphate deficiency on aromatic L-amino acid decarboxylase activity with L-DOPA and L-5-hydroxytryptophan as substrates in rats". Japanese Journal of Pharmacology. 32 (5): 803–11. doi:10.1254/jjp.32.803. PMID 6983619.
- Bouchard S, Bousquet C, Roberge AG (September 1981). "Characteristics of dihydroxyphenylalanine/5-hydroxytryptophan decarboxylase activity in brain and liver of cat". Journal of Neurochemistry. 37 (3): 781–7. doi:10.1111/j.1471-4159.1982.tb12555.x. PMID 6974228.
- Nakatani Y, Sato-Suzuki I, Tsujino N, Nakasato A, Seki Y, Fumoto M, Arita H (May 2008). "Augmented brain 5-HT crosses the blood-brain barrier through the 5-HT transporter in rat". The European Journal of Neuroscience. 27 (9): 2466–72. doi:10.1111/j.1460-9568.2008.06201.x. PMID 18445233.
- Bouchard S, Roberge AG (July 1979). "Biochemical properties and kinetic parameters of dihydroxyphenylalanine--5-hydroxytryptophan decarboxylase in brain, liver, and adrenals of cat". Canadian Journal of Biochemistry. 57 (7): 1014–8. doi:10.1139/o79-126. PMID 39668.
- Amamoto T, Sarai K (September 1976). "On the tryptophan-serotonin metabolism in manic-depressive disorders. Changes in plasma 5-HT and 5-HIAA levels and urinary 5-HIAA excretion following oral loading of L-5HTP in patients with depression". Hiroshima Journal of Medical Sciences. 25 (2–3): 135–40. PMID 1088369.
- "5-HTP: Uses, Side Effects, Interactions and Warnings - WebMD". Archived from the original on 16 November 2009. Retrieved 5 October 2009.
- Magnussen I, Jensen TS, Rand JH, Van Woert MH (September 1981). "Plasma accumulation of metabolism of orally administered single dose L-5-hydroxytryptophan in man". Acta Pharmacologica et Toxicologica. 49 (3): 184–9. doi:10.1111/j.1600-0773.1981.tb00890.x. PMID 6175178.
- Sternberg EM, Van Woert MH, Young SN, Magnussen I, Baker H, Gauthier S, Osterland CK (October 1980). "Development of a scleroderma-like illness during therapy with L-5-hydroxytryptophan and carbidopa". The New England Journal of Medicine. 303 (14): 782–7. doi:10.1056/NEJM198010023031403. PMID 6997735.
- "Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations".
- "5-HTP: MedlinePlus Supplements".
- Jacobsen JP, Rudder ML, Roberts W, Royer EL, Robinson TJ, Oh A, Spasojevic I, Sachs BD, Caron MG (August 2016). "SSRI Augmentation by 5-Hydroxytryptophan Slow Release: Mouse Pharmacodynamic Proof of Concept". Neuropsychopharmacology. 41 (9): 2324–34. doi:10.1038/npp.2016.35. PMC 4946063. PMID 26932820.
- Thombre AG (2005). "Assessment of the feasibility of oral controlled release in an exploratory development setting". Drug Discov Today. 10 (17): 1159–66. doi:10.1016/S1359-6446(05)03551-8. PMID 16182208.
- "5-Hydroxytryptophan". University of Maryland Medical Center. Archived from the original on 6 January 2010. Retrieved 21 January 2010.
- "5-Hydroxytryptophan (5-HTP)". A.D.A.M., Inc. University of Maryland Medical Center. Animated Dissection of Anatomy for Medicine, Inc. (A.D.A.M., Inc.) provided health and benefits information and technology to healthcare organizations, employers, consumers, and educational institutions
- Emanuele E, Bertona M, Minoretti P, Geroldi D (2010). "An open-label trial of L-5-hydroxytryptophan in subjects with romantic stress". Neuro Endocrinology Letters. 31 (5): 663–6. PMID 21178946.
- "5-hydroxy-L-tryptophan", National Center for Biotechnology Information, PubChem Compound Database, September 2004CID=439280
- Rondanelli M, Opizzi A, Faliva M, Bucci M, Perna S (March 2012). "Relationship between the absorption of 5-hydroxytryptophan from an integrated diet, by means of Griffonia simplicifolia extract, and the effect on satiety in overweight females after oral spray administration". Eating and Weight Disorders. 17 (1): e22–8. doi:10.3275/8165. PMID 22142813.