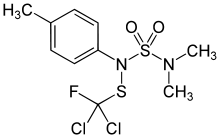
Tolylfluanid
Tolylfluanid is an organic chemical compound that is used as an active ingredient in fungicides and wood preservatives.
 | |
| Names | |
|---|---|
| IUPAC name
N-[dichloro(fluoro)methyl]sulfanyl-N-(dimethylsulfamoyl)-4-methylaniline | |
| Identifiers | |
3D model (JSmol) |
|
| ECHA InfoCard | 100.010.898 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H13Cl2FN2O2S2 | |
| Molar mass | 347.244 g/mol |
| Appearance | colourless, odourless crystals |
| Density | 1.52 g/cm3 |
| Melting point | 93°C |
| Boiling point | Decomposes on distillation |
| water, 0.9 mg/L at 20°C. Miscible in all proportions with acetone, ethanol, ethyl acetate, methylene chloride | |
| Vapor pressure | <1.3 mPa (20°C) |
| Related compounds | |
Related compounds |
Dichlofluanid |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
|
LC50 (median concentration) |
0.02-0.3 mg/L |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Synthesis
The synthesis of tolylfluanid begins with the reaction of dimethylamine and sulfuryl chloride. The product further reacts with p-toluidine and dichlorofluoromethanesulfenyl chloride to yield the final product.[1]
Use
Tolylfluanid is used on fruit and ornamental plants against gray mold (Botrytis), against late blight on tomatoes and against powdery mildew on cucumbers.
Environmental behavior
Tolylfluanid hydrolyzes slowly in acidic conditions. The half-life is shorter when the pH is high; at pH = 7, it is at least 2 days. In aerobic media (pH = 7.7-8.0), tolylfluanid hydrolytically and microbially decomposes to N,N-dimethyl-N-(4-methylphenyl) sulfamide (DMST) and dimethylsulfamide. After 14 days, tolylfluanid is generally considered to have degraded. The half-life of DMST is 50-70 days. [2]
Absorption, metabolism and excretion
Tolylfluanid is rapidly and almost completely absorbed in the gastrointestinal tract. The highest concentrations are found in the blood, lungs, liver, kidneys, spleen and thyroid gland. 99% is excreted in the urine within two days, although there is some accumulation in the thyroid gland. [2]
References
- Thomas A. Unger (1996). Pesticide Synthesis Handbook. William Andrew. p. 985. ISBN 0-8155-1853-6.
- Svensk Chemicals Inspection : Tolylfluanid (PDF). Archived from the original (PDF) on 2007-09-27.