Sodium orthosilicate
Sodium orthosilicate is the chemical compound Na
4SiO
4. It is one of the sodium silicates, specifically an orthosilicate, formally a salt of the unstable orthosilicic acid H
4SiO
4.[2][3][4]
| Names | |
|---|---|
| IUPAC name
Tetrasodium silicate | |
| Other names
Sodium orthosilicate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.033.387 |
| EC Number |
|
PubChem CID |
|
| UNII | |
| UN number | 1759 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Na4O4Si | |
| Molar mass | 184.040 g·mol−1 |
| Appearance | White powder |
| Melting point | 1,018 °C (1,864 °F; 1,291 K) |
| Hazards | |
| GHS pictograms | 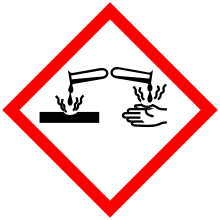  |
| GHS Signal word | Danger[1] |
GHS hazard statements |
H302, H314, H318, H335[1] |
| P260, P303+361+353, P305+351+338, P301+330+331, P405, P501[1] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Uses
Sodium orthosilicate has been considered as an interfacial tension reducing additive in the waterflooding of oil fields for enhanced oil extraction. In laboratory settings, it was found to be more effective than sodium hydroxide for some types of oil.[5]
Sodium orthosilicate has been found to stabilize ferrate films as an anticorrosion treatment of iron and steel surfaces.[6]
Natural occurrence
Sodium orthosilicate has not been found in nature. However, the mineral chesnokovite, chemically the related salt disodium dihydrogen orthosilicate [Na+
]2[SiO
2(OH)2−
2] · 8H
2O, was recently identified in the Kola Peninsula.[7]
See also
- Sodium metasilicate, Na
2SiO
3
References
- "Sodium Orthosilicate". American Elements. Retrieved September 18, 2018.
- Werner H. Baur, Erich Halwax, and Horst Völlenkle (1986): "Comparison of the crystal structures of sodium orthosilicate, {[chem|Na|4|SiO|4, and sodium orthogermanate, Na
4GeO
4". Monatshefte für Chemie, volume 117, issue 6–7, pages 793–797. doi:10.1007/BF00810070 - M,. G. Barker, P. G.Gadd (1981): "The preparation and crystal structure of sodium orthosilicate, Na
4SiO
4." Journal of Chemical Research, London] Chemical Society, volume 9, pages S:274 (synopse), M:3446-3466 (main). - J. F. Schairer and N. L. Bowen (1956): "The system Na
2O—Al
2O
3—SiO
2". American Journal of Science, volume 254, issue 3, pages 129-195 doi:10.2475/ajs.254.3.129 - Thomas C. Campbell (11977): "A Comparison Of Sodium Orthosilicate And Sodium Hydroxide For Alkaline Waterflooding." Society of Petroleum engineers, SPE California Regional Meeting 13–15 April 1977, Bakersfield; document IDSPE-6514-MS doi:10.2118/6514-MS
- John R. Harrison (1954): "Process for treating metals with ferrate solution". US Patent US2850415A, assigned to E. I. du Pont de Nemours
- I. V. Pekov, N. V. Chukanov, A. E. Zadov, N. V. Zubkova, and D. Yu. Pushcharovsky (2007): "Chesnokovite, Na
2[SiO
2(OH)
2] · 8H
2O, the first natural sodium orthosilicate from the Lovozero alkaline pluton, Kola Peninsula: Description and crystal structure of a new mineral species", Geology of Ore Deposits, volume 49, issue 8, pages 727–738. doi:10.1134/S1075701507080077