Potassium carbonate
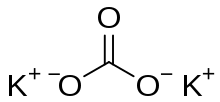
Potassium carbonate is the inorganic compound with the formula K2CO3. It is a white salt, which is soluble in water. It is deliquescent, often appearing as a damp or wet solid. Potassium carbonate is mainly used in the production of soap and glass.[2]
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Potassium carbonate | |
| Other names
Carbonate of potash, dipotassium carbonate, sub-carbonate of potash, pearl ash, potash, salt of tartar, salt of wormwood. | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.008.665 |
| E number | E501(i) (acidity regulators, ...) |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| K 2CO 3 | |
| Molar mass | 138.205 g/mol |
| Appearance | White, hygroscopic solid |
| Density | 2.43 g/cm3 |
| Melting point | 891 °C (1,636 °F; 1,164 K) |
| Boiling point | Decomposes |
| 112 g/100 mL (20 °C) 156 g/100 mL (100 °C) | |
| Solubility | |
| −59.0·10−6 cm3/mol | |
| Hazards | |
| Safety data sheet | ICSC 1588 |
| GHS pictograms |  |
| GHS Signal word | Warning |
GHS hazard statements |
H302, H315, H319, H335 |
| P261, P305+351+338 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
1870 mg/kg (oral, rat)[1] |
| Related compounds | |
Other anions |
Potassium bicarbonate |
Other cations |
Lithium carbonate Sodium carbonate Rubidium carbonate Caesium carbonate |
Related compounds |
Ammonium carbonate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
History
Potassium carbonate is the primary component of potash and the more refined pearl ash or salts of tartar. Historically, pearl ash was created by baking potash in a kiln to remove impurities. The fine, white powder remaining was the pearl ash. The first patent issued by the US Patent Office was awarded to Samuel Hopkins in 1790 for an improved method of making potash and pearl ash.
In late 18th century North America, before the development of baking powder, pearl ash was used as a leavening agent for quick breads.[3][4]
Production
Potassium carbonate is prepared commercially by the reaction potassium hydroxide with carbon dioxide:[2]
- 2 KOH + CO2 → K2CO3 + H2O
From the solution crystallizes the sesquihydrate K2CO3·3⁄2H2O ("potash hydrate"). Heating this solid above 200 °C gives the anhydrous salt. Alternative method, potassium chloride is treated with carbon dioxide in the presence of an organic amine to give potassium bicarbonate, which is then calcined:
- 2 KHCO3 → K2CO3 + H2O + CO2
Applications
- (historically) for soap, glass, and china production
- as a mild drying agent where other drying agents, such as calcium chloride and magnesium sulfate, may be incompatible. It is not suitable for acidic compounds, but can be useful for drying an organic phase if one has a small amount of acidic impurity. It may also be used to dry some ketones, alcohols, and amines prior to distillation.[5]
- in cuisine, where it has many traditional uses. It is an ingredient in the production of grass jelly, a food consumed in Chinese and Southeast Asian cuisines, as well as Chinese hand-pulled noodles and moon cake. It is also used to tenderize tripe. German gingerbread recipes often use potassium carbonate as a baking agent, although in combination with hartshorn. Use of potassium carbonate must be limited to a certain amount to prevent harm, and should not be used without guidance.
- in the alkalization of cocoa powder to produce Dutch process chocolate by balancing the pH (i.e., reduce the acidity) of natural cocoa beans; it also enhances aroma. The process of adding potassium carbonate to cocoa powder is usually called "Dutching" (and the products referred to as Dutch-processed cocoa powder), as the process was first developed in 1828 by Dutchman Coenraad Johannes van Houten.
- as a buffering agent in the production of mead or wine.
- in antique documents, it is reported to have been used to soften hard water.[6]
- as a fire suppressant in extinguishing deep-fat fryers and various other B class-related fires.
- in condensed aerosol fire suppression, although as the byproduct of potassium nitrate.
- as an ingredient in welding fluxes, and in the flux coating on arc-welding rods.
- as an animal feed ingredient to satisfy the potassium requirements of farmed animals such as broiler breeders.
- as an acidity regulator in Swedish snus
References
- Chambers, Michael. "ChemIDplus - 584-08-7 - BWHMMNNQKKPAPP-UHFFFAOYSA-L - Potassium carbonate [USP] - Similar structures search, synonyms, formulas, resource links, and other chemical information". chem.sis.nlm.nih.gov. Archived from the original on 2014-08-12.
- H. Schultz, G. Bauer, E. Schachl, F. Hagedorn, P. Schmittinger (2005). "Potassium Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a22_039.CS1 maint: uses authors parameter (link)
- See references to "pearl ash" in "American Cookery" by Amelia Simmons, printed by Hudson & Goodwin, Hartford, 1796.
- Civitello, Linda (2017). Baking powder wars : the cutthroat food fight that revolutionized cooking. Urbana, Illinois: University of Illinois Press. pp. 18–22. ISBN 9780252041082.
- Leonard, J.; Lygo, B.; Procter, G. "Advanced Practical Organic Chemistry" 1998, Stanley Thomas Publishers Ltd
- Child, Lydia M. "The American Frugal Housewife" 1832
Bibliography
- A Dictionary of Science, Oxford University Press, New York, 2004
- Yu. Platonov, Andrew; Evdokimov, Andrey; Kurzin, Alexander; D. Maiyorova, Helen (29 June 2002). "Solubility of Potassium Carbonate and Potassium Hydrocarbonate in Methanol". Journal of Chemical & Engineering Data. 47 (5): 1175–1176. doi:10.1021/je020012v.
