Disufenton sodium
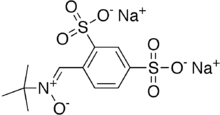
Disufenton sodium (NXY-059, Cerovive) is the disulfonyl derivative of the neuroprotective spin trap phenylbutylnitrone or "PBN". It was under development at the drug company AstraZeneca. A 2005 phase-3 clinical trial[1][2] called "SAINT-1" reported some efficacy in the acute treatment of ischemia injury due to stroke. However, a 2006 attempt to repeat this trial indicated no significant activity. After ruling out other causes, the authors tentatively attributed the positive results in the first trial to "chance".[1] AstraZeneca then terminated the development programme.[3] PBN and its derivatives hydrolyze and oxidize in vitro to form respectively MNP-OH (AKA, NtBHA) and its parent spin-trap MNP.
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C11H13NNa2O7S2 |
| Molar mass | 381.32 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
References
- Lees KR, Zivin JA, Ashwood T, Davalos A, Davis SM, Diener HC, et al. (February 2006). "NXY-059 for acute ischemic stroke". The New England Journal of Medicine. 354 (6): 588–600. doi:10.1056/NEJMoa052980. PMID 16467546.
- Lees KR, Davalos A, Davis SM, Diener HC, Grotta J, Lyden P, et al. (December 2006). "Additional outcomes and subgroup analyses of NXY-059 for acute ischemic stroke in the SAINT I trial". Stroke. 37 (12): 2970–8. doi:10.1161/01.STR.0000249410.91473.44. PMID 17068304.
- "Renovis: Press Release". Archived from the original on October 28, 2006.
External links
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.