Milk fat globule membrane
Milk fat globule membrane (MFGM) is a complex and unique structure composed primarily of lipids and proteins that surrounds milk fat globule secreted from the milk producing cells of humans and other mammals. It is a source of multiple bioactive compounds, including phospholipids, glycolipids, glycoproteins, and carbohydrates that have important functional roles within the brain and gut.
Preclinical studies have demonstrated effects of MFGM-derived bioactive components on brain structure and function, intestinal development, and immune defense. Similarly, pediatric clinical trials have reported beneficial effects on cognitive and immune outcomes. In populations ranging from premature infants to preschool-age children, dietary supplementation with MFGM or its components has been associated with improvements in cognition and behavior, gut and oral bacterial composition, fever incidence, and infectious outcomes including diarrhea and otitis media.
MFGM may also play a role in supporting cardiovascular health by modulating cholesterol and fat uptake. Clinical trials in adult populations have shown that MFGM could positively affect markers associated with cardiovascular disease including lowering serum cholesterol and triacylglycerol levels as well as blood pressure.
Origin
MFGM secretion process in milk
Milk lipids are secreted in a unique manner by lactocytes, which are specialized epithelial cells within the alveoli of the lactating mammary gland.
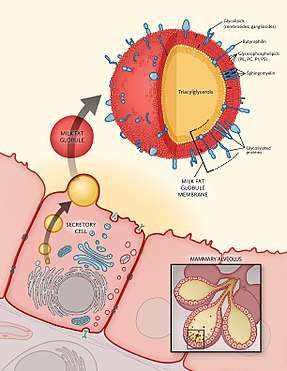
The process takes place in multiple stages. First, fat synthesized within the endoplasmic reticulum accumulates in droplets between the inner and outer phospholipid monolayers of the endoplasmic reticulum membrane. As these droplets increase in size, the two monolayers separate further and eventually pinch off. This leads to the surrounding of the droplet in a phospholipid monolayer that allows it to disperse within the aqueous cytoplasm. In the next stage, lipid droplets then migrate to the apical surface of the cell, where plasma membrane subsequently envelops the droplet and extrudes together with it. It fully encases the fat droplet in an additional bilayer of phospholipids. The milk fat globule thus released into the glandular lumen, measuring 3-6 μm in average diameter, is surrounded by a phospholipid trilayer containing associated proteins, carbohydrates, and lipids derived primarily from the membrane of the secreting lactocyte. This trilayer is collectively known as MFGM.[1][2]
This secretion process occurs in all types of mammalian milk, including human and bovine. However, it is distinct from the lipid secretion mechanism used by all other non-mammary cells. That makes MFGM unique to milk and it is not present in non-dairy food products.[2]
Sources of MFGM
MFGM is a structurally complex bioactive milk component, found in human milk as well as the milk of other mammalian species. The MFGM in human milk contains many bioactive components with diverse functions and has been linked to cognitive and health benefits to infants. Some compositional differences are reported to exist between species, but bovine MFGM, the best-studied non-human source, generally contains a lipid and protein composition, which is similar to that of human MFGM.[3][4]
MFGM makes up an estimated 2-6% of the total fat globules.[5] As raw milk has an average total fat content around 4%,[6][7] it therefore contains around 0.08-0.24% of MFGM. In other words, 417–1250 kg of raw milk is needed to supply 1 kg of MFGM. The content of MFGM in dairy products varies depending on the processing involved. During dairy processing such as churning or decreaming, the MFGM is disrupted and preferentially distributed into aqueous phases such as buttermilk, butter serum, or certain type of whey.[8] Thus they can be a good source of MFGM for addition into food products.
For example, infant formulas traditionally were lacking the MFGM because this fraction is lost during regular dairy processing.[9] However, more recent advances in technology have facilitated the separation of MFGM from the fat globule, allowing bovine MFGM to be added in concentrated form.[8] The MFGM fraction is now commercially available and can be added to infant formula or other nutritional products.
Structure and components
General structure
The milk fat globule is surrounded by a phospholipid trilayer containing associated proteins, carbohydrates, and lipids derived primarily from the membrane of the secreting mammary epithelial cell (lactocyte). This trilayer is collectively known as MFGM. While MFGM makes up only an estimated 2% to 6% of the total milk fat globule,[1] it is an especially rich phospholipid source, accounting for the majority of total milk phospholipids.[10][11] In contrast, the inner core of the milk fat globule is composed predominantly of triacylglycerols.
The MFGM structure is complex and comprises a variety of phospholipids, glycolipids, proteins, and glycoproteins, along with cholesterol and other lipids. Specific lipids and proteins are localized to different layers of the membrane, with carbohydrate chains of glycoproteins and glycolipids directed toward the outer surface of the milk fat globule; the lipid-to-protein weight ratio in MFGM is approximately 1:1.[12]
However, the nutritional significance of these components is defined not only by their structure or macronutrient category, but also by the physiological role that each nutrient serves. As a quantitatively minor presence within milk, MFGM likely contributes little to energy production, but its constituents may confer structural and functional benefits.[8] Many of these nutrients are known to play important functional roles within the gut, brain, and elsewhere in the body; the functions of other components are still being elucidated.
Lipid components
The lipid component of MFGM is rich in phospholipids, glycosphingolipids, and cholesterol. Phospholipids make up approximately 30% of the total lipid weight of MFGM, the three most prominent being sphingomyelin (SM), phosphatidylcholine (PC), and phosphatidylethanolamine (PE), which together represent up to 85% of total phospholipids.[1][12] Phospholipids and sphingolipids play central roles in cerebral neurogenesis and migration during fetal development, as well as promoting neuronal growth, differentiation, and synaptogenesis during the first year of life.[13][14] Other important polar lipids present in the membrane include the glycerophospholipids phosphatidylserine (PS) and phosphatidylinositol (PI), as well as gangliosides (GG), which are sphingolipids containing sialic acid and an oligosaccharide side chain. Each of these lipid classes is known to play functional roles within the body, including the support of gut, immune, and central nervous system development.[9][15]
Protein components
Besides the polar lipids, the outer layer of MFGM contains a number of glycosylated and non-glycosylated proteins. Proteomic analysis has revealed at least 191 different known proteins in human MFGM, and comparable numbers in bovine milk protein concentrates.[1] While quantitatively these only represent 1% to 2% of total milk protein content,[16] MFGM proteins are of significant interest because many are known to have bioactive and potentially beneficial properties; almost half of identified proteins have membrane/protein trafficking or cell signaling functions.[17] The glycosylated proteins, including mucins (MUC-1, MUC-4, MUC-15), butyrophilin, lactadherin, and CD36, have been suggested to enhance triacylglyceride digestion efficiency.[1] Furthermore, lactadherin and MUC-1, in addition to the non-glycosylated protein xanthine oxidase, have been shown or suggested in preclinical studies to possess antimicrobial properties.[18][19][20][21][22]
Health Benefits of MFGM
Research has indicated that MFGM, or components thereof, may potentially play roles in brain development and cognitive function, immunity and gut health, and cardiovascular health.
Brain development and cognitive function
MFGM lipid components such as sphingomyelin and gangliosides are highly concentrated in the brain and support synaptogenesis and myelination.[23][24] In the central nervous system, sphingomyelin is a key component of the myelin sheath, which insulates axons and supports efficient transmission of nerve impulses.[25][26] During myelination, nerve axons are wrapped with multiple layers of cell membrane by oligodendrocyte glial cells, a process that accounts for a large portion of brain growth during late gestation and the first two years of life,[27] but which can also continue up to 5–10 years of age.[28] Meanwhile, gangliosides are concentrated within the brain’s gray matter and constitute approximately 6% to 10% of the total human brain lipid mass.[29][30][31][32][33][34] Additionally, gangliosides are enriched at the synaptic membrane of neurons, and are functionally involved in neurotransmission and synapse formation.[35][24] Brain ganglioside accretion occurs at an accelerated rate in the early years of life, coinciding with the most active period of myelination, axonal outgrowth, and synaptogenesis.[36][37] Alongside the growth of brain size, total brain ganglioside concentration also increases 3-fold from early fetal development to 5 years of age.[36]
Preclinical data
A number of preclinical studies have been conducted using MFGM and combinations of MFGM-derived components. Liu et al. (2014) studied brain development and spatial learning and memory in neonatal piglets.[38] Piglets that were fed with a formula containing milk phospholipids and gangliosides to mimic levels in human milk made choices more rapidly and with fewer errors in a spatial T-maze cognitive test compared to controls, implying improved spatial learning. Similarly, Vickers et al. (2009) demonstrated that administration of complex milk lipids to rats from postnatal day 10 through adulthood (day 80) led to significant improvements in learning and memory tasks compared to control animals.[39] Conversely, a study of complex milk lipid supplementation to pregnant mice did not have an effect on cognitive tasks in their offspring.[40]
Clinical data
Several studies of diets supplemented with MFGM and its components, including gangliosides and sphingomyelin, have aimed to address measures of cognitive development in pediatric populations. In some of the studies, MFGM supplementation to infant formula was shown to narrow the gap in cognitive development between breastfed and formula-fed infants.
Tanaka et al. (2013) studied the neurobehavioral effects of feeding formula supplemented with sphingomyelin-enriched phospholipid in 24 very low birth weight preterm infants (birth weight <1500 g).[23] In this double-blind RCT, the preterm infants were fed either control formula containing phospholipids derived from egg yolk lecithin with sphingomyelin at 13% of total phospholipid or a supplemented formula with milk-derived phospholipids containing 20% sphingomyelin. Infants fed the supplemented formula had significantly higher percentages of sphingomyelin in total plasma phospholipids after 4, 6, and 8 weeks of feeding compared to those fed the control formula. The infants fed the supplemented formula also showed improvements across multiple developmental measures at 18 months, with significantly better scores on the Behavior Rating Scale of the Bayley Scales of Infant Development II (BSID-II), the Fagan test (novelty preference rate), the latency of visual evoked potentials (VEP), and sustained attention test than in the control group.
Gurnida et al. (2012) assessed the cognitive effects of formula supplemented with a ganglioside-enriched, MFGM-derived complex milk lipid in term infants.[41] In this double-blind RCT, healthy infants (2–8 weeks of age) were fed until 6 months of age, either control infant formula (n=30), or a supplemented infant formula (n=29) with added complex milk lipids to increase ganglioside concentration to approximately 11-12 μg/mL to be within the human milk range. A breastfed reference group (n=32) was also included. Results showed that serum ganglioside levels in the supplemented group were significantly higher compared to the control group at 6 months, but did not significantly differ from levels in the breastfed group. The cognitive outcomes measured using the Griffiths Mental Development Scale showed that the supplemented group had significantly increased scores for Hand and Eye Coordination, Performance, and Total Score (General Quotient) at 6 months compared to the control group, and there were no significant differences in cognitive performance compared to the breastfed reference group.
Timby et al. (2014) also assessed the potential impact of MFGM supplementation on cognitive development in term infants.[42] In this double-blind RCT, term infants (<2 months old) were fed until 6 months of age either a control formula (n=64) or an MFGM-supplemented formula (n=71). A breastfed reference group (n=70) was also included. Cognitive assessment done using the BSID-III at 12 months of age showed that the MFGM-fed infants exhibited significantly higher mean cognitive scores than the control group (105.8 vs 101.8; P<0.008), and not significantly different from the breastfed reference group. In contrast, there were no significant differences in motor domain scores between the three groups, and both experimental and control formula groups scored lower than the reference group in the verbal domain.
Veereman-Wauters et al. (2012) assessed the potential behavioral benefits of MFGM supplementation in young children.[43] In this double-blind RCT, healthy preschool children (2.5 to 6 years of age) consumed for 4 months, either a control formula (n=97) providing 60 mg/day of endogenous phospholipid, or an MFGM-supplemented formula (n=85) providing a total of 500 mg/day of dairy-derived phospholipids. At the end of the trial, parents and teachers completed the Achenbach System of Empirically Based Assessment (ASEBA), a validated questionnaire considered to be a gold standard for assessing emotion and behavior in preschool children.[44] Significant differences in internal, external, and total behavioral problem scores were observed in favor of the supplemented formula group, as reported by parents (but not by teachers).
Immunity and gut health
MFGM bioactive protein components, including the glycoproteins lactadherin, MUC-1, and butyrophilin, have been shown in preclinical studies to affect immune response.[45] These components influence the immune system by several mechanisms, including interference with microbe adhesion to intestinal epithelia, bacteriocidal action, support of beneficial microbiota, and modulation of other parts of the immune system.[9]
MFGM phospholipid components such as phosphatidylcholine are a key constituent of the intestinal mucus barrier, and therefore may contribute to intestinal defense against invasive pathogens.[46] Sphingolipids, including sphingomyelin, are present in the apical membrane of the gut epithelia, and are also important for maintaining membrane structure, modulating growth factor receptors, and serving competitive binding inhibitors for microorganisms, microbial toxins, and viruses.[47] Gangliosides are also present in intestinal mucosa and may possibly contribute to improved gut microflora and antibacterial defense.[48]
Preclinical data
MFGM may be capable of modulating immune function in the gut through distinct but potentially complementary mechanisms. Glycosylated proteins (MUC-1, MUC-15, butyrophilin, and lactadherin) and glycosylated sphingolipids from MFGM may promote the development of healthy gut microbiota by favoring beneficial Bifidobacterium species.[49] Another key to the immunomodulatory function of MFGM may be that its structure is similar to that of the intestinal cell membrane, allowing human milk glycans (including those on glycoproteins and glycolipids) to competitively inhibit the binding of pathogens (bacteria, viruses, even toxins) to host cells.[50]
A number of preclinical studies have demonstrated inhibitory effects of MFGM against several pathogens. Both whole bovine MFGM and its extracted lipid components were found to exhibit dose-dependent inhibition of rotavirus infectivity in vitro.[51] Antibacterial effects of MFGM have included decreased gastric colonization and inflammation after H. pylori infection in mice;[52] inhibition of shiga toxin gene expression by E. coli O157:H7;[53] and decreased colonization and translocation of L. monocytogenes.[54] Mice that were fed prophylactically with bovine whey glycoprotein fraction, including MFGM proteins, did not develop diarrhea after exposure to rotavirus.[55]
Clinical data
The previously described study by Timby et al. (2015) also assessed the effects of MFGM supplementation in term infants on the risk of infectious diseases and other disease symptoms.[56] In particular, the cumulative incidence of acute otitis media was analyzed between the two randomized feeding groups (control formula or MFGM- supplemented formula to 6 months of age), and compared to a breastfed reference group. The MFGM-supplemented group experienced a significant reduction in episodes of acute otitis media up to 6 months of age compared with infants fed control formula (1% vs 9%; P=0.034); with no significant difference in otitis media incidence compared to the breastfed group (0%). In addition, a significantly lower incidence and longitudinal prevalence of antipyretic drug use was seen in the MFGM-supplemented group (25%) compared with the control formula group (43%). Timby et al. (2017) further showed that the MFGM supplementation influenced the infants’ oral microbiota; the authors noted that, Moraxella catarrhalis, a common bacterial cause of acute otitis media, was less prevalent in infants fed the MFGM-supplemented formula than in those fed control formula.[57]
Zavaleta et al. (2011) evaluated the effects of an MFGM-enriched complementary food on health outcomes in term infants 6 to 11 months of age in Peru.[58] In this double-blind RCT, 499 primarily breast-fed infants were fed for 6 months with a daily milk-based complementary food that included either whey protein concentrate enriched in MFGM, or an equal amount of additional protein from skim milk (control). Results showed that the group with the MFGM-supplemented diet had a significantly lower prevalence of diarrhea during the study compared to the control group (3.84% vs 4.37%; P<0.05), as well as a significant reduction (46%) in episodes of bloody diarrhea compared to the control group (P=0.025).
Later through analyzing the metabolome and immune markers of those infants, Lee et al. (2018) reported that supplementation with MFGM may improve micronutrient status, amino acid, and energy metabolism along with a reduced proinflammatory response (e.g. interleukin-2).[59]
The previously described study by Veereman-Wauters et al. (2012) in preschool-age children (2.5 to 6 years old) also reported the effect of MFGM-supplemented formula consumption on health outcomes. Children receiving the MFGM-supplemented formula reported a significant reduction in the number of days with fever, and particularly the number of short febrile episodes (<3 days), compared to the control group.[43]
Cardiovascular health
Dietary guidelines generally recommend limiting full-fat dairy products. This recommendation has been based on traditional hypothesis that dietary saturated fatty acids, such as those derived from milk fat, have serum LDL cholesterol raising effects. Subsequently, although not demonstrated in randomized controlled trials, serum LDL cholesterol has been associated with cardiovascular disease (CVD) risk based on observational evidence.[60][61] A review of observational studies has suggested that the association between milk fat intake and serum cholesterol measures could vary depending on the type of dairy products.[61] Differential effects of various dairy foods on plasma lipids might be partly dependent on the presence of MFGM.[62] MFGM lipid components may play a role in supporting cardiovascular health by modulating cholesterol and fat uptake.
Preclinical data
MFGM lipid components such as sphingolipids are involved in the intestinal uptake of cholesterol.[8] Studies in adult rodents have shown that milk sphingomyelin could lower the intestinal absorption of cholesterol in a dose-dependent manner.[63][64] Intestinal cholesterol absorption in adult rodents consuming a high fat diet was limited by sphingomyelin supplementation.[65] Milk sphingomyelin and other phospholipids with high affinity for cholesterol could limit the micellar solubility of intestinal cholesterol, thereby limiting the cholesterol uptake by the enterocyte.[63] Dietary sphingolipids have been shown to dose-dependently lower plasma cholesterol and triacylglycerol in adult rodents fed with Western-type diet and protect the liver from fat- and cholesterol-induced steatosis.[66] Dietary sphingolipids also lowers hepatic cholesterol and triglyceride levels in adult rodents partly by modulating hepatic gene expression.[65]
Clinical data
Several clinical studies have shown that MFGM could positively affect circulating lipids. A single-blind RCT in overweight adults has shown that the effects of milk fat on plasma lipids were modulated by the MFGM content; compared to butter oil (control diet), consumption of whipping cream (MFGM diet) for 8 weeks did not impair the lipoprotein profile.[62] Another double-blind RCT in overweight and obese adults has also shown that MFGM attenuated the negative effects of a high-saturated fats meal by reducing postprandial cholesterol, inflammatory markers and insulin response.[67] A double-blind RCT in normal healthy adults has indicated that one month consumption of buttermilk rich in MFGM led to reduction in serum cholesterol and triacylglycerol levels as well as blood pressure.[68][69]
MFGM supplementation in infancy is hypothesized to have programming effects that may influence circulating lipid levels later in life. Breastfed infants are known to have a higher total serum cholesterol and LDL cholesterol than formula-fed infants in infancy, but lower levels in adulthood. A clinical study in infants has suggested that MFGM supplementation could narrow the gap between breastfed and formula-fed infants with regard to serum lipid status.[70] Specifically, as compared with a control formula, infants receiving MFGM-supplemented formula had higher total serum cholesterol until 6 months of age, similar to breastfed infants. The LDL:HDL ratio did not differ between the formula-fed groups and was significantly higher in the breastfed reference group as compared with both formula-fed groups.
References
- Lopez, Christelle; Ménard, Olivia (1 March 2011). "Human milk fat globules: polar lipid composition and in situ structural investigations revealing the heterogeneous distribution of proteins and the lateral segregation of sphingomyelin in the biological membrane". Colloids and Surfaces. B, Biointerfaces. 83 (1): 29–41. doi:10.1016/j.colsurfb.2010.10.039. PMID 21126862.
- Heid, Hans W.; Keenan, Thomas W. (1 March 2005). "Intracellular origin and secretion of milk fat globules". European Journal of Cell Biology. 84 (2–3): 245–258. doi:10.1016/j.ejcb.2004.12.002. PMID 15819405.
- Timby, Niklas; Hernell, Olle; Vaarala, Outi; Melin, Merit; Lönnerdal, Bo; Domellöf, Magnus (1 March 2015). "Infections in infants fed formula supplemented with bovine milk fat globule membranes". Journal of Pediatric Gastroenterology and Nutrition. 60 (3): 384–389. doi:10.1097/MPG.0000000000000624. PMID 25714582.
- Billeaud, Claude; Puccio, Giuseppe; Saliba, Elie; Guillois, Bernard; Vaysse, Carole; Pecquet, Sophie; Steenhout, Philippe (22 September 2017). "Safety and tolerance evaluation of milk fat globule membrane-enriched infant formulas: a randomized controlled multicenter non-inferiority trial in healthy term infants". Clinical Medicine Insights. Pediatrics. 8: 51–60. doi:10.4137/CMPed.S16962. PMC 4219856. PMID 25452707.
- Singh, Harjinder (2006). "The milk fat globule membrane—A biophysical system for food applications". Current Opinion in Colloid & Interface Science. 11 (2–3): 154–163. doi:10.1016/j.cocis.2005.11.002.
- Walstra, P.; Walstra, Pieter; Wouters, Jan T. M.; Geurts, Tom J. (2006). Dairy Science and Technology, Second Edition. Florida: CRC Press. p. 433. ISBN 9781420028010.
- Le, Thien Trung; Phan, Thi Thanh Que; Van Camp, John; Dewettinck, Koen (2015). "Milk and Dairy Polar Lipids: Occurrence, Purification, and Nutritional and Technological Properties". Polar Lipids. pp. 91–143. doi:10.1016/b978-1-63067-044-3.50009-1. ISBN 9781630670443.
- Dewettinck, Koen; Rombaut, Roeland; Thienpont, Natacha; Le, Thien Trung; Messens, Kathy; Van Camp, John (2008). "Nutritional and technological aspects of milk fat globule membrane material". International Dairy Journal. 18 (5): 436–457. doi:10.1016/j.idairyj.2007.10.014.
- Lönnerdal, Bo (1 March 2014). "Infant formula and infant nutrition: bioactive proteins of human milk and implications for composition of infant formulas". The American Journal of Clinical Nutrition. 99 (3): 712S–7S. doi:10.3945/ajcn.113.071993. PMID 24452231.
- Gallier, Sophie; Gragson, Derek; Jiménez-Flores, Rafael; Everett, David (14 April 2010). "Using confocal laser scanning microscopy to probe the milk fat globule membrane and associated proteins". Journal of Agricultural and Food Chemistry. 58 (7): 4250–4257. doi:10.1021/jf9032409. PMC 2853928. PMID 20218614.
- Keenan, T. W. (1 July 2001). "Milk lipid globules and their surrounding membrane: a brief history and perspectives for future research". Journal of Mammary Gland Biology and Neoplasia. 6 (3): 365–371. doi:10.1023/A:1011383826719. PMID 11547904.
- Kanno, C. (1990). "Secretory membranes of the lactating mammary gland". Protoplasma. 159 (2–3): 184–208. doi:10.1007/BF01322601.
- Vance, J. E.; Campenot, R. B.; Vance, D. E. (26 June 2000). "The synthesis and transport of lipids for axonal growth and nerve regeneration". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids. 1486 (1): 84–96. doi:10.1016/S1388-1981(00)00050-0. PMID 10856715.
- Hirabayashi, Yoshio; Furuya, Shigeki (1 May 2008). "Roles of l-serine and sphingolipid synthesis in brain development and neuronal survival". Progress in Lipid Research. 47 (3): 188–203. doi:10.1016/j.plipres.2008.01.003. PMID 18319065.
- Küllenberg, Daniela; Taylor, Lenka A.; Schneider, Michael; Massing, Ulrich (5 January 2012). "Health effects of dietary phospholipids". Lipids in Health and Disease. 11: 3. doi:10.1186/1476-511X-11-3. PMC 3316137. PMID 22221489.
- Riccio, Paolo (2004). "The proteins of the milk fat globule membrane in the balance". Trends in Food Science & Technology. 15 (9): 458–461. doi:10.1016/j.tifs.2003.12.005.
- Reinhardt, Timothy A.; Lippolis, John D. (2006). "Bovine Milk Fat Globule Membrane Proteome". Journal of Dairy Research. 73 (4): 406–16. doi:10.1017/S0022029906001889. PMID 16834814.
- Bojsen, A.; Buesa, J.; Montava, R.; Kvistgaard, A. S.; Kongsbak, M. B.; Petersen, T. E.; Heegaard, C. W.; Rasmussen, J. T. (1 January 2007). "Inhibitory activities of bovine macromolecular whey proteins on rotavirus infections in vitro and in vivo". Journal of Dairy Science. 90 (1): 66–74. doi:10.3168/jds.S0022-0302(07)72609-7. PMID 17183076.
- Kvistgaard, A. S.; Pallesen, L. T.; Arias, C. F.; López, S.; Petersen, T. E.; Heegaard, C. W.; Rasmussen, J. T. (1 December 2004). "Inhibitory effects of human and bovine milk constituents on rotavirus infections". Journal of Dairy Science. 87 (12): 4088–4096. doi:10.3168/jds.S0022-0302(04)73551-1. PMID 15545370.
- Spitsberg, V. L. (1 July 2005). "Invited review: Bovine milk fat globule membrane as a potential nutraceutical". Journal of Dairy Science. 88 (7): 2289–2294. doi:10.3168/jds.S0022-0302(05)72906-4. PMID 15956291.
- Clare, Debra A.; Zheng, Zuoxing; Hassan, Hosni M.; Swaisgood, Harold E.; Catignani, George L. (1 January 2008). "Antimicrobial properties of milkfat globule membrane fractions". Journal of Food Protection. 71 (1): 126–133. doi:10.4315/0362-028X-71.1.126. PMID 18236672.
- Cavaletto, Maria; Giuffrida, Maria Gabriella; Conti, Amedeo (22 September 2017). Milk fat globule membrane components--a proteomic approach. Advances in Experimental Medicine and Biology. 606. pp. 129–141. doi:10.1007/978-0-387-74087-4_4. ISBN 978-0-387-74086-7. PMID 18183927.
- Tanaka, K.; Hosozawa, M.; Kudo, N.; Yoshikawa, N.; Hisata, K.; Shoji, H.; Shinohara, K.; Shimizu, T. (1 January 2013). "The pilot study: sphingomyelin-fortified milk has a positive association with the neurobehavioural development of very low birth weight infants during infancy, randomized control trial". Brain & Development. 35 (1): 45–52. doi:10.1016/j.braindev.2012.03.004. PMID 22633446.
- McJarrow, Paul; Schnell, Nicholas; Jumpsen, Jacqueline; Clandinin, Tom (1 August 2009). "Influence of dietary gangliosides on neonatal brain development". Nutrition Reviews. 67 (8): 451–463. doi:10.1111/j.1753-4887.2009.00211.x. PMID 19674342.
- Jana, Arundhati; Pahan, Kalipada (1 December 2010). "Sphingolipids in multiple sclerosis". Neuromolecular Medicine. 12 (4): 351–361. doi:10.1007/s12017-010-8128-4. PMC 2987401. PMID 20607622.
- Oshida, Kyoichi; Shimizu, Takashi; Takase, Mitsunori; Tamura, Yoshitaka; Shimizu, Toshiaki; Yamashiro, Yuichiro (1 April 2003). "Effects of dietary sphingomyelin on central nervous system myelination in developing rats". Pediatric Research. 53 (4): 589–593. doi:10.1203/01.PDR.0000054654.73826.AC. PMID 12612207.
- Kinney, H. C.; Brody, B. A.; Kloman, A. S.; Gilles, F. H. (1 May 1988). "Sequence of central nervous system myelination in human infancy. II. Patterns of myelination in autopsied infants". Journal of Neuropathology and Experimental Neurology. 47 (3): 217–234. doi:10.1097/00005072-198805000-00003. PMID 3367155. S2CID 13986261.
- Grantham-McGregor, Sally; Cheung, Yin Bun; Cueto, Santiago; Glewwe, Paul; Richter, Linda; Strupp, Barbara (2007-01-06). "Developmental potential in the first 5 years for children in developing countries". Lancet. 369 (9555): 60–70. doi:10.1016/S0140-6736(07)60032-4. ISSN 0140-6736. PMC 2270351. PMID 17208643.
- Ledeen, RW; Wu, G; Cannella, MS; Oderfeld-Nowak, B; Cuello, AC (1990). "Gangliosides as neurotrophic agents: studies on the mechanism of action" (PDF). Acta Neurobiologiae Experimentalis. 50 (4–5): 439–49. PMID 2130660. Retrieved 22 September 2017.
- Ledeen, R. W.; Yu, R. K. (22 September 1982). Gangliosides: structure, isolation, and analysis. Methods in Enzymology. 83. pp. 139–191. doi:10.1016/0076-6879(82)83012-7. ISBN 9780121819835. PMID 7047999.
- Kracun, I.; Rosner, H.; Drnovsek, V.; Heffer-Lauc, M.; Cosović, C.; Lauc, G. (1 September 1991). "Human brain gangliosides in development, aging and disease". The International Journal of Developmental Biology. 35 (3): 289–295. PMID 1814411.
- Yu, Robert K.; Nakatani, Yoshihiko; Yanagisawa, Makoto (1 April 2009). "The role of glycosphingolipid metabolism in the developing brain". Journal of Lipid Research. 50 Suppl: S440–445. doi:10.1194/jlr.R800028-JLR200. PMC 2674698. PMID 18845618.
- Kolter, Thomas (22 September 2017). "Ganglioside biochemistry". ISRN Biochemistry. 2012: 506160. doi:10.5402/2012/506160. PMC 4393008. PMID 25969757.
- Wang, Bing (22 September 2017). "Sialic acid is an essential nutrient for brain development and cognition". Annual Review of Nutrition. 29: 177–222. doi:10.1146/annurev.nutr.28.061807.155515. PMID 19575597.
- Palmano, Kate; Rowan, Angela; Guillermo, Rozey; Guan, Jian; McJarrow, Paul (22 May 2015). "The role of gangliosides in neurodevelopment". Nutrients. 7 (5): 3891–3913. doi:10.3390/nu7053891. PMC 4446785. PMID 26007338.
- Svennerholm, L.; Boström, K.; Fredman, P.; Månsson, J. E.; Rosengren, B.; Rynmark, B. M. (25 September 1989). "Human brain gangliosides: developmental changes from early fetal stage to advanced age". Biochimica et Biophysica Acta (BBA) - Lipids and Lipid Metabolism. 1005 (2): 109–117. doi:10.1016/0005-2760(89)90175-6. PMID 2775765.
- Kinney, Hannah C. (15 February 2005). "Human myelination and perinatal white matter disorders". Journal of the Neurological Sciences. 228 (2): 190–192. doi:10.1016/j.jns.2004.10.006. PMID 15694202.
- Liu, Hongnan; Radlowski, Emily C.; Conrad, Matthew S.; Li, Yao; Dilger, Ryan N.; Johnson, Rodney W. (1 December 2014). "Early supplementation of phospholipids and gangliosides affects brain and cognitive development in neonatal piglets". The Journal of Nutrition. 144 (12): 1903–1909. doi:10.3945/jn.114.199828. PMC 4230208. PMID 25411030.
- Vickers, Mark H.; Guan, Jian; Gustavsson, Malin; Krägeloh, Christian U.; Breier, Bernhard H.; Davison, Michael; Fong, Bertram; Norris, Carmen; McJarrow, Paul; Hodgkinson, Steve C. (1 June 2009). "Supplementation with a mixture of complex lipids derived from milk to growing rats results in improvements in parameters related to growth and cognition". Nutrition Research (New York, N.Y.). 29 (6): 426–435. doi:10.1016/j.nutres.2009.06.001. PMID 19628110.
- Gustavsson, Malin; Hodgkinson, Steve C.; Fong, Bertram; Norris, Carmen; Guan, Jian; Krageloh, Christian U.; Breier, Bernhard H.; Davison, Michael; McJarrow, Paul; Vickers, Mark H. (1 April 2010). "Maternal supplementation with a complex milk lipid mixture during pregnancy and lactation alters neonatal brain lipid composition but lacks effect on cognitive function in rats". Nutrition Research (New York, N.Y.). 30 (4): 279–289. doi:10.1016/j.nutres.2010.04.005. PMID 20534331.
- Gurnida, Dida A.; Rowan, Angela M.; Idjradinata, Ponpon; Muchtadi, Deddy; Sekarwana, Nanan (2012). "Association of complex lipids containing gangliosides with cognitive development of 6-month-old infants". Early Human Development. 88 (8): 595–601. doi:10.1016/j.earlhumdev.2012.01.003. PMID 22289412.
- Timby, Niklas; Domellöf, Erik; Hernell, Olle; Lönnerdal, Bo; Domellöf, Magnus (1 April 2014). "Neurodevelopment, nutrition, and growth until 12 mo of age in infants fed a low-energy, low-protein formula supplemented with bovine milk fat globule membranes: a randomized controlled trial". The American Journal of Clinical Nutrition. 99 (4): 860–868. doi:10.3945/ajcn.113.064295. PMID 24500150.
- Veereman-Wauters, Gigi; Staelens, Sofie; Rombaut, Roeland; Dewettinck, Koen; Deboutte, Dirk; Brummer, Robert-Jan; Boone, Marc; Le Ruyet, Pasale (1 July 2012). "Milk fat globule membrane (INPULSE) enriched formula milk decreases febrile episodes and may improve behavioral regulation in young children". Nutrition (Burbank, Los Angeles County, Calif.). 28 (7–8): 749–752. doi:10.1016/j.nut.2011.10.011. PMID 22305534.
- Bérubé, R.L. and Achenbach, T.M., 2007. Bibliography of published studies using the ASEBA. Burlington, VT: University of Vermont, Research Center for Children, Youth, and Families.
- Peterson, J. A.; Patton, S.; Hamosh, M. (22 September 1998). "Glycoproteins of the human milk fat globule in the protection of the breast-fed infant against infections". Biology of the Neonate. 74 (2): 143–162. doi:10.1159/000014020. PMID 9691156.
- Olson, Alicia; Diebel, Lawrence N.; Liberati, David M. (1 October 2014). "Exogenous phosphatidylcholine supplementation improves intestinal barrier defense against Clostridium difficile toxin". The Journal of Trauma and Acute Care Surgery. 77 (4): 570–575, discussion 576. doi:10.1097/TA.0000000000000378. PMID 25250596.
- Vesper, H.; Schmelz, E. M.; Nikolova-Karakashian, M. N.; Dillehay, D. L.; Lynch, D. V.; Merrill, A. H. (1 July 1999). "Sphingolipids in food and the emerging importance of sphingolipids to nutrition". The Journal of Nutrition. 129 (7): 1239–1250. doi:10.1093/jn/129.7.1239. PMID 10395583.
- Rueda, Ricardo (2007). "The role of dietary gangliosides on immunity and the prevention of infection". British Journal of Nutrition. 98: S68–73. doi:10.1017/S0007114507832946. PMID 17922964.
- Bourlieu, Claire; Michalski, Marie-Caroline (1 March 2015). "Structure-function relationship of the milk fat globule". Current Opinion in Clinical Nutrition and Metabolic Care. 18 (2): 118–127. doi:10.1097/MCO.0000000000000138. PMID 25581036.
- Newburg, D. S. (1 April 2009). "Neonatal protection by an innate immune system of human milk consisting of oligosaccharides and glycans". Journal of Animal Science. 87 (13 Suppl): 26–34. doi:10.2527/jas.2008-1347. PMID 19028867.
- Fuller, K. L.; Kuhlenschmidt, T. B.; Kuhlenschmidt, M. S.; Jiménez-Flores, R.; Donovan, S. M. (1 June 2013). "Milk fat globule membrane isolated from buttermilk or whey cream and their lipid components inhibit infectivity of rotavirus in vitro". Journal of Dairy Science. 96 (6): 3488–3497. doi:10.3168/jds.2012-6122. PMID 23548280.
- Wang, B.; Brand-Miller, J.; McVeagh, P.; Petocz, P. (1 October 2001). "Concentration and distribution of sialic acid in human milk and infant formulas". The American Journal of Clinical Nutrition. 74 (4): 510–515. doi:10.1093/ajcn/74.4.510. PMID 11566650.
- Tellez, A.; Corredig, M.; Guri, A.; Zanabria, R.; Griffiths, M. W.; Delcenserie, V. (2012). "Bovine milk fat globule membrane affects virulence expression in Escherichia coli O157:H7". Journal of Dairy Science. 95 (11): 6313–6319. doi:10.3168/jds.2012-5560. ISSN 1525-3198. PMID 22981580.
- Sprong, R. Corinne; Hulstein, Marco F. E.; Lambers, Tim T.; van der Meer, Roelof (14 December 2012). "Sweet buttermilk intake reduces colonisation and translocation of Listeria monocytogenes in rats by inhibiting mucosal pathogen adherence". The British Journal of Nutrition. 108 (11): 2026–2033. doi:10.1017/S0007114512000165. PMID 22370235.
- Inagaki, Mizuho; Nagai, Sayaka; Yabe, Tomio; Nagaoka, Satoshi; Minamoto, Nobuyuki; Takahashi, Takeshi; Matsuda, Tsukasa; Nakagomi, Osamu; Nakagomi, Toyoko; Ebina, Takusaburo; Kanamaru, Yoshihiro (22 September 2017). "The bovine lactophorin C-terminal fragment and PAS6/7 were both potent in the inhibition of human rotavirus replication in cultured epithelial cells and the prevention of experimental gastroenteritis". Bioscience, Biotechnology, and Biochemistry. 74 (7): 1386–1390. doi:10.1271/bbb.100060. PMID 20622446.
- Timby, Niklas; Hernell, Olle; Vaarala, Outi; Melin, Merit; Lönnerdal, Bo; Domellöf, Magnus (March 2015). "Infections in infants fed formula supplemented with bovine milk fat globule membranes". Journal of Pediatric Gastroenterology and Nutrition. 60 (3): 384–389. doi:10.1097/MPG.0000000000000624. ISSN 1536-4801. PMID 25714582.
- Timby, Niklas; Domellöf, Magnus; Holgerson, Pernilla Lif; West, Christina E.; Lönnerdal, Bo; Hernell, Olle; Johansson, Ingegerd (2017). "Oral Microbiota in Infants Fed a Formula Supplemented with Bovine Milk Fat Globule Membranes - A Randomized Controlled Trial". PLOS ONE. 12 (1): e0169831. Bibcode:2017PLoSO..1269831T. doi:10.1371/journal.pone.0169831. PMC 5242539. PMID 28099499.
- Zavaleta, Nelly; Kvistgaard, Anne Staudt; Graverholt, Gitte; Respicio, Graciela; Guija, Henry; Valencia, Norma; Lönnerdal, Bo (November 2011). "Efficacy of an MFGM-enriched complementary food in diarrhea, anemia, and micronutrient status in infants". Journal of Pediatric Gastroenterology and Nutrition. 53 (5): 561–568. doi:10.1097/MPG.0b013e318225cdaf. ISSN 1536-4801. PMID 21637131.
- Lee, Hanna; Zavaleta, Nelly; Chen, Shin-Yu; Lönnerdal, Bo; Slupsky, Carolyn (2018). "Effect of bovine milk fat globule membranes as a complementary food on the serum metabolome and immune markers of 6-11-month-old Peruvian infants". NPJ Science of Food. 2: 6. doi:10.1038/s41538-018-0014-8. PMC 6550191. PMID 31304256.
- Ramsden, Christopher E.; Zamora, Daisy; Majchrzak-Hong, Sharon; Faurot, Keturah R.; Broste, Steven K.; Frantz, Robert P.; Davis, John M.; Ringel, Amit; Suchindran, Chirayath M.; Hibbeln, Joseph R. (2016). "Re-evaluation of the traditional diet-heart hypothesis: Analysis of recovered data from Minnesota Coronary Experiment (1968-73)". BMJ. 353: i1246. doi:10.1136/bmj.i1246. PMC 4836695. PMID 27071971.
- Huth, Peter J.; Park, Keigan M. (1 May 2012). "Influence of dairy product and milk fat consumption on cardiovascular disease risk: a review of the evidence". Advances in Nutrition (Bethesda, Md.). 3 (3): 266–285. doi:10.3945/an.112.002030. PMC 3649459. PMID 22585901.
- Rosqvist, Fredrik; Smedman, Annika; Lindmark-Månsson, Helena; Paulsson, Marie; Petrus, Paul; Straniero, Sara; Rudling, Mats; Dahlman, Ingrid; Risérus, Ulf (1 July 2015). "Potential role of milk fat globule membrane in modulating plasma lipoproteins, gene expression, and cholesterol metabolism in humans: a randomized study". The American Journal of Clinical Nutrition. 102 (1): 20–30. doi:10.3945/ajcn.115.107045. PMID 26016870.
- Eckhardt, Erik R. M.; Wang, David Q.-H.; Donovan, Joanne M.; Carey, Martin C. (1 April 2002). "Dietary sphingomyelin suppresses intestinal cholesterol absorption by decreasing thermodynamic activity of cholesterol monomers". Gastroenterology. 122 (4): 948–956. doi:10.1053/gast.2002.32539. PMID 11910347.
- Noh, Sang K.; Koo, Sung I. (1 October 2004). "Milk sphingomyelin is more effective than egg sphingomyelin in inhibiting intestinal absorption of cholesterol and fat in rats". The Journal of Nutrition. 134 (10): 2611–2616. doi:10.1093/jn/134.10.2611. PMID 15465755.
- Chung, Rosanna W. S.; Kamili, Alvin; Tandy, Sally; Weir, Jacquelyn M.; Gaire, Raj; Wong, Gerard; Meikle, Peter J.; Cohn, Jeffrey S.; Rye, Kerry-Anne (22 September 2017). "Dietary sphingomyelin lowers hepatic lipid levels and inhibits intestinal cholesterol absorption in high-fat-fed mice". PLOS ONE. 8 (2): e55949. Bibcode:2013PLoSO...855949C. doi:10.1371/journal.pone.0055949. PMC 3567029. PMID 23409094.
- Duivenvoorden, Ilse; Voshol, Peter J.; Rensen, Patrick CN; Van Duyvenvoorde, Wim; Romijn, Johannes A.; Emeis, Jef J.; Havekes, Louis M.; Nieuwenhuizen, Willem F. (2006). "Dietary sphingolipids lower plasma cholesterol and triacylglycerol and prevent liver steatosis in APOE*3Leiden mice1–3". The American Journal of Clinical Nutrition. 84 (2): 312–321. doi:10.1093/ajcn/84.1.312. PMID 16895877.
- Demmer, Elieke; Van Loan, Marta D.; Rivera, Nancy; Rogers, Tara S.; Gertz, Erik R.; German, J. Bruce; Smilowitz, Jennifer T.; Zivkovic, Angela M. (2016). "Addition of a dairy fraction rich in milk fat globule membrane to a high-saturated fat meal reduces the postprandial insulinaemic and inflammatory response in overweight and obese adults". Journal of Nutritional Science. 5: e14. doi:10.1017/jns.2015.42. ISSN 2048-6790. PMC 4791522. PMID 27313850.
- Conway, V.; Couture, P.; Richard, C.; Gauthier, S. F.; Pouliot, Y.; Lamarche, B. (1 December 2013). "Impact of buttermilk consumption on plasma lipids and surrogate markers of cholesterol homeostasis in men and women". Nutrition, Metabolism, and Cardiovascular Diseases: NMCD. 23 (12): 1255–1262. doi:10.1016/j.numecd.2013.03.003. PMID 23786821.
- Conway, Valérie; Couture, Patrick; Gauthier, Sylvie; Pouliot, Yves; Lamarche, Benoît (1 January 2014). "Effect of buttermilk consumption on blood pressure in moderately hypercholesterolemic men and women". Nutrition (Burbank, Los Angeles County, Calif.). 30 (1): 116–119. doi:10.1016/j.nut.2013.07.021. PMID 24206823.
- Timby, Niklas; Lönnerdal, Bo; Hernell, Olle; Domellöf, Magnus (October 2014). "Cardiovascular risk markers until 12 mo of age in infants fed a formula supplemented with bovine milk fat globule membranes". Pediatric Research. 76 (4): 394–400. doi:10.1038/pr.2014.110. ISSN 1530-0447. PMID 25116230.