Cyclooctadiene iridium chloride dimer
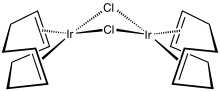
Cyclooctadiene iridium chloride dimer is an organoiridium compound with the formula Ir2Cl2(C8H12)2, where C8H12 is the diene 1,5-cyclooctadiene. It is an orange solid that is soluble in organic solvents. The complex is used as a precursor to other iridium complexes, some of which are used in homogeneous catalysis.[1] The solid is air-stable but its solutions degrade in air.
 | |
2.jpg) | |
| Names | |
|---|---|
| Other names
Bis(1,5-cyclooctadiene)diiridium(I) dichloride | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.031.961 |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| C16H24Cl2Ir2 | |
| Molar mass | 671.70 |
| Appearance | red-orange solid |
| Density | 2.65 g/cm3 (red polymorph) |
| Hazards | |
| GHS pictograms |  |
| GHS Signal word | Warning |
GHS hazard statements |
H302, H312, H315, H319, H335 |
| P261, P264, P270, P271, P280, P301+312, P302+352, P304+340, P305+351+338, P312, P321, P322, P330, P332+313, P337+313, P362, P363, P403+233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation, structure, reactions
The compound is prepared by heating hydrated iridium trichloride and cyclooctadiene in alcohol solvent. In the process, Ir(III) is reduced to Ir(I).[2]
In terms of its molecular structure, the iridium centers are square planar as is typical for a d8 complex. The Ir2Cl2 core is folded with a dihedral angle of 86°. The molecule crystallizes in yellow-orange and red-orange polymorphs; the latter one is more common.[3][4]
The complex is widely used precursor to other iridium complexes. A notable derivative is Crabtree's catalyst.[5] The chloride ligands can also be replaced with methoxide to give cyclooctadiene iridium methoxide dimer, Ir2(OCH3)2(C8H12)2.[6] The cyclooctadiene ligand is prone to isomerize in cationic complexes of the type (C8H8)IrL2]+.[7]
References
- J. Hartwig, "Organotransition Metal Chemistry: From Bonding to Catalysis" University Science Books, 2009. ISBN 978-1891389535.
- Herdé, J. L.; Lambert, J. C.; Senoff, C. V. (1974). "Cyclooctene and 1,5-Cyclooctadiene Complexes of Iridium(I)". Inorganic Syntheses. 15: 18–20. doi:10.1002/9780470132463.ch5.
- F. Albert Cotton, Pascual Lahuerta, Mercedes Sanau, Willi Schwotzer "Air oxidation of Ir2(Cl)2(COD)2 revisited. The structures of [Ir(μ2-Cl)(COD)]2 (ruby form) and its oxidation product, Ir2Cl2(COD)2(μ2-OH)2(μ2-O)" Inorganica Chimica Acta, 1986 vol. 120, Pages 153–157. doi:10.1016/S0020-1693(00)86102-2
- Tabrizi, D., Manoli, J. M., Dereigne, A., "Etude radiocristallographique de μ-dichloro-bis (π cyclooctadiène-1,5) diiridium: [(COD-1,5)IrCl]2, variété jaune-orange", Journal of the Less Common Metals 1970, vol. 21, pp. 337. doi:10.1016/0022-5088(70)90155-4
- Crabtree, Robert H.; Morehouse, Sheila M. (1986). "[η4-1,5-Cyclooctadiene)(Pyridine)(Tricyclohexylphosphine)Iridium(I)Hexafluorophosphate". Inorganic Syntheses. 24: 173–176. doi:10.1002/9780470132555.ch50.
- Uson, R.; Oro, L. A.; Cabeza, J. A. (1985). "Dinuclear Methoxy, Cyclooctadiene, and Barrelene Complexes of Rhodium(I) and Iridium(I)". Inorganic Syntheses. 23: 126–130. doi:10.1002/9780470132548.ch25. ISBN 9780470132548.
- Martín, Marta; Sola, Eduardo; Torres, Olga; Plou, Pablo; Oro, Luis A. (2003). "Versatility of Cyclooctadiene Ligands in Iridium Chemistry and Catalysis". Organometallics. 22 (26): 5406–5417. doi:10.1021/om034218g.