Cerebral organoid
A cerebral organoid, or brain organoid, describes artificially grown, in vitro, miniature organs resembling the brain. Cerebral organoids are created by culturing human pluripotent stem cells in a three-dimensional rotational bioreactor and develop over a course of months.[1] The human brain is an extremely complex system of heterogeneous tissues and consists of an extremely diverse array of neurons. This complexity has made studying the brain and understanding how it works a difficult task in neuroscience, especially when it comes to neurodegenerative diseases. The purpose of creating an in vitro neurological model is to study these diseases in a more simple and variable space; free of in vivo limitations, especially when working with humans. The varying physiology between human and other mammalian models limits the scope of study in neurological disorders. Cerebral organoids are synthesized tissues that contain several types of nerve cells and have anatomical features that resemble mammalian brains. Cerebral organoids are most similar to layers of neurons called the cortex and choroid plexus. In some cases, structures similar to the retina, meninges and hippocampus can form.[1][2] Stem cells have the potential to grow into many different types of tissues and their fate is dependent on many factors. Below is an image showing some of the chemical factors that can lead stem cells to differentiate into various neural tissues with a more in-depth table of generating specific organoid identity being published since.[3] Similar techniques are used on stem cells used to grow cerebral organoids.[4]
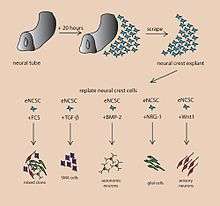
Model development
Using human pluripotent stem cells to create in vitro cerebral organoids allows researchers to summarize current developmental mechanisms for human neural tissue as well as study the roots of human neurological diseases. Cerebral organoids are an investigative tool, used to understand how disease pathology works. These organoids can be used in experiments that current in vitro methods are too simple for, while also being more human applicable than rodent or other mammalian models. Historically, major breakthroughs in how the brain works have resulted from injury or disorder in human brain function, leading to an understanding of how regions of the brain work. An in vitro human brain model would allow for the next wave in an understanding of the human brain.[1][4]
Applications
In addition to being used as tools to study disease pathology and treatments, future application of cerebral organoids include direct implantation into a human host. The organoid can fuse with host tissue in areas of neurodegeneration, being incorporated with the host vasculature and be immunologically silent.[5] A list of potential applications for cerebral organoids is highlighted below.
Potential applications include:[6]
- Tissue morphogenesis with respect to cerebral organoids covers how neural organs form in vertebrates. Cerebral organoids can serve as in vitro tools to study the formation, modulate it, and further understand the mechanisms controlling it.[6]
- Migration assays
- Cerebral organoids can help to study cell migration. Neural glial cells cover a wide variety of neural cells, some of which move around the neurons. The factors that govern their movements can be studied using cerebral organoids.[4]
- Clonal lineage tracing
- Clonal lineage tracing is part of fate mapping, where the lineage of differentiated tissues is traced to the pluripotent progenitors. The local stimuli released and mechanism of differentiation can be studied using cerebral organoids as a model.[6]
- Transplantation
- Cerebral organoids can be used to grow specific brain regions and transplant them into regions of neurodegeneration as a therapeutic treatment.[7][8]
- Cell fate potential
- Cross species developmental timing
- Cerebral organoids provide a unique insight into the timing of development of neural tissues and can be used as a tool to study the differences across species.[6]
- Cerebral organoids can be used as simple models of complex brain tissues to study the effects of drugs and to screen them for initial safety and efficacy.
- Cell replacement therapy
- Cerebral organoids can be used as a simple model to show how cell replacement therapy would work on brain tissues.[6]
- Cell-type specific genome assays
Disease
Organoids can be used to study the crucial early stages of brain development, test drugs and, because they can be made from living cells, study individual patients.[2]
Microcephaly
In one case, a cerebral organoid grown from a patient with microcephaly demonstrated related symptoms and revealed that apparently, the cause is overly rapid development, followed by slower brain growth. Microencephaly is a developmental condition in which the brain remains undersized, producing an undersized head and debilitation. Microcephaly is not suitable for mouse models, which do not replicate the condition.[2] The primary form of the disease is thought to be caused by a homozygous mutation in the microcephalin gene. The disease is difficult to reproduce in mouse models because mice lack the developmental stages for an enlarged cerebral cortex that humans have. Naturally, a disease which affects this development would be impossible to show in a model which does not have it to begin with.[9] To use cerebral organoids to model a human's microcephaly, one group of researchers has taken patient skin fibroblasts and reprogrammed them using four well known reprogramming factors. These include OCT4, SOX2, MYC and KLF4. The reprogrammed sample was able to be cloned into induced pluripotent stem cells. The cells were cultured into a cerebral organoid following a process described in the cerebral organoid creation section below. The organoid that resulted had decreased numbers of neural progenitor cells and smaller tissues. Additionally, the patient-derived tissues displayed fewer and less frequent neuroepithelial tissues made of progenitors, decreased radial glial stem cells, and increased neurons. These results suggest that the underlying mechanism of microcephaly is caused by cells prematurely differentiating into neurons leaving a deficit of radial glial cells.[1]
Alzheimer's disease
Alzheimer's disease pathology has also been modelled with cerebral organoids.[10] Affected individual’s pluripotent stem cells were used to generate brain organoids and then compared to control models, synthesised from healthy individuals. It was found that in the affected models, structures similar to that of plaques caused by amyloid beta proteins and neurofibrillary tangles, that cause the disease's symptoms were observed.[11] Previous attempts to model this so accurately have been unsuccessful with drugs being developed on the basis off efficacy in pre-clinical models such as mice not having any effect in human trials.[12]
Autism spectrum diseases
Similarly, autism has also been studied through the comparison of healthy verses affected synthesised brain organoids.[13] Observation of the two models showed the overexpression of a transcription factor FOXG1 that produced a larger amount of GABAergic inhibitory neurons in the affected models. The significance of this use of brain organoids is that it has added great support for the excitatory/inhibitory imbalance hypothesis [14] which if proven true could help identify targets for drugs so that the disease could be treated. A pending study at the University of California, San Diego will use cerebral organoids derived from autistic adolescent boys to measure how cannabidiol (CBD) could affect brain connectivity and neuroinflammation as it relates to autism spectrum disorder. The "mini brains" will allow researchers to measure the effects of many different concentrations of CBD without testing directly on the study's participants.
Preterm Hypoxia/Ischemia
Preterm hypoxic injury remain difficult to study because of limited availability of human fetal brain tissues and inadequate animal models to study human corticogenesis. Cerebral organoid can be used to model prenatal pathophysiology and to compare the susceptibility of the different neural cell types to hypoxia during corticogenesis. Intermediate progenitors seems to be particularly affected, due to the unfolded protein response pathway.[15] It has also been observed that hypoxia resulted in apoptosis in cerebral organoids, with outer radial glia and neuroblasts/immature neurons being particularly affected.[16]
Construction
To make an organoid, an embryoid (tissue that has some embryonic features) grown from natural stem cells is used. Embryos have three layers: endoderm, mesoderm and ectoderm. Each turns into various body parts. The nervous system grows from the ectoderm (which also contributes dental enamel and the epidermis).[4] Ectodermal cells were placed into gel droplets and floated in a nutrient broth in a rotating bioreactor, which supported cell growth without forming by the container. After ten days the organoid developed neurons. After 30 days it displayed regions similar to parts of brains. Lacking a blood supply, cerebral organoids reach about 4 mm across and can last a year or more.[2] The general procedure can be broken down into 5 steps. First human pluripotent stem cells are cultured. They are then allowed to cultivate into an embryoid body. Next the cell culture is induced to form a neuroectoderm. The neuroectoderm is then grown in a matrigel droplet. The matrigel provides nutrients and the neuroectoderm starts to proliferate and grow. However, the lack of vasculature limits the size the organoid can grow. This has been the major limitation in organoid development, however new methods using a spinning bioreactor have allowed an increase in the availability of nutrients to cells inside the organoid. This last step has been the key breakthrough in organoid development.[17] Spinning bioreactors have been used increasingly in cell culture and tissue growth applications. The reactor is able to deliver faster cell doubling times, increased cell expansion and increased extra-cellular matrix components when compared to statically cultured cells.[18]
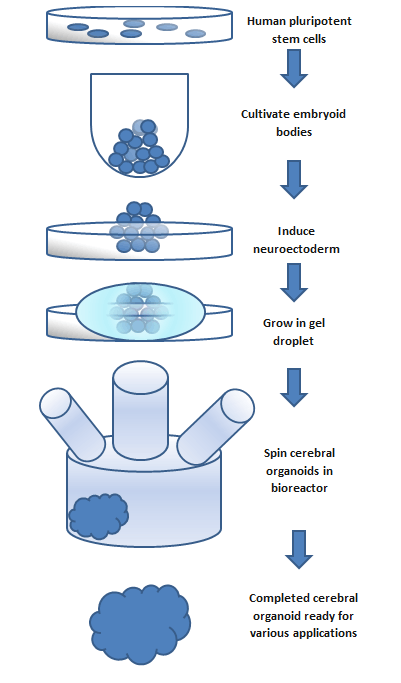
Figure: This flow chart outlines the basic steps to create a cerebral organoid. The process takes a span of months and the size of the organoid is limited to the availability of nutrients.
This was the original method outlined by Madeline Lancaster [1] and has since been developed and refined. Newer methods allow development of cerebrovascular organoids,[19] and micro pumps to provide circulation through them are being developed, as explained in this video by Dr George M. Church.
Testing
Differentiation
It has been shown that cerebral organoids grown using the spinning bioreactor 3D culture method differentiate into various neural tissue types, such as the optic cup, hippocampus, ventral parts of the teleencephelon and dorsal cortex.[20] The neural stem/progenitor cells are unique because they are able to self-renew and are multipotent. This means they can generate neurons and glial cells which are the two main components of neural systems. The fate of these cells is controlled by several factors that affect the differentiation process. The spatial location and temporal attributes of neural progenitor cells can influence if the cells form neurons or glial cells. Further differentiation is then controlled by extracellular conditions and cell signaling.[21] The exact conditions and stimuli necessary to differentiate neural progenitor cells into specific neural tissues such as hippocampal tissue, optic nerve, cerebral cortex, etc. are unknown. It is believed that cerebral organoids can be used to study the developmental mechanisms of these processes.[17]
Gene expression
To test if the neural progenitor cells and stem cells are differentiating into specific neural tissues, several gene markers can be tested. Two markers that are present during pluripotent stages are OCT4 and NANOG. These two markers are diminished during the course of development for the organoid. Neural identity markers that note successful neural induction, SOX1 and PAX6, are upregulated during organoid development. These changes in expression support the case for self-guided differentiation of cerebral organoids.[1] Markers for forebrain and hindbrain can also be tested. Forebrain markers FOXG1 and SIX3 are highly expressed throughout organoid development. However, hindbrain markers EGR2 and ISL1 show early presence but a decrease in the later stages. This imbalance towards forebrain development is similar to the developmental expansion of forebrain tissue in human brain development.[1] To test if organoids develop even further into regional specification, gene markers for cerebral cortex and occipital lobe have been tested. Many regions that have forebrain marker FOXG1, labeling them as regions with cerebral cortical morphology, were also positive for marker EMX1 which indicates dorsal cortical identity. These specific regions can be even further specified by markers AUTS2, TSHZ2, and LMO4 with the first representing cerebral cortex and the two after representing the occipital lobe.[1] Genetic markers for the hippocampus, ventral forebrain, and choroid plexus are also present in cerebral organoids, however, the overall structures of these regions have not yet been formed.
Localization
Functional
Cerebral organoids also possess functional cerebral cortical neurons. These neurons must form on the radially organized cortical plate. The marker TBR1 is present in the preplate, the precursor to the cortical plate, and is present, along with MAP2, a neuronal marker, in 30-day-old cerebral organoids. These markers are indicative of a basal neural layer similar to a preplate. These cells are also apically adjacent to a neutral zone and are reelin+ positive, which indicates the presence of Cajal-Retzius cells. The Cajal-Retzius cells are important to the generation of cortical plate architecture.[17] The cortical plate is usually generated inside-out such that later-born neurons migrate to the top superficial layers. This organization is also present in cerebral organoids based on genetic marker testing. Neurons that are early born have marker CTIP2 and are located adjacent to the TBR1 exhibiting preplate cells. Late-born neurons with markers SATB2 and BRN2 are located in a superficial layer, further away from the preplate than the early born neurons suggesting cortical plate layer formation. Additionally, after 75 days of formation, cerebral organoids show a rudimentary marginal zone, a cell-poor region. The formation of layered cortical plate is very basic in cerebral organoids and suggests the organoid lacks the cues and factors to induce formation of layer II-VI organization.[1] The cerebral organoid neurons can, however, form axons as shown by GFP staining. GFP labeled axons have been shown to have complex branching and growth cone formation. Additionally, calcium dye imaging has shown cerebral organoids to have Ca2+ oscillations and spontaneous calcium surges in individual cells. The calcium signaling can be enhanced through glutamate and inhibited through tetrodotoxin.[1]
Physical
It is not fully understood how individual localized tissues formed by stem cells are able to coordinate with surrounding tissues to develop into a whole organ.[22] It has been shown however that most tissue differentiation requires interactions with surrounding tissues and depends on diffusible induction factors to either inhibit or encourage various differentiation and physical localization.[22] Cerebral organoid differentiation is somewhat localized. The previously mentioned markers for forebrain and hindbrain are physically localized, appearing in clusters. This suggests that local stimuli are released once one or more cells differentiate into a specific type as opposed to a random pathway throughout the tissue. The markers for subspecification of cortical lobes, prefrontal cortex and occipital lobe, are also physically localized. However, the hippocampus and ventral forebrain cells are not physically localized and are randomly located through the cerebral organoid.[1] Cerebral organoids lack blood vessels and are limited in size by nutrient uptake in the innermost cells. Spinning bioreactors and advanced 3D scaffolding techniques are able to increase organoid size, though the integration of in vitro nutrient delivery systems is likely to spark the next major leap in cerebral organoid development.[6]
Ethics
Ethical concerns have been raised with using cerebral organoids as a model from disease due to the potential of them experiencing sensations such as pain or having the ability to develop a consciousness.[23] Currently it is unlikely given the simplicity of synthesised models compared to the complexity of a human brain, however models have been shown to respond to light-based stimulation,[24] so present models do have some scope of responding to some stimuli at present. If such sensations could be proven to be present in any of the models, then the ethics of their use would be questionable.
Steps are being taken towards resolving the grey area such as a 2018 symposium at Oxford University where experts in the field, philosophers and lawyers met to try to clear up the ethical concerns with the new technology.[25] Similarly, projects such as Brainstorm from Case Western University aim to observe the progress of the field by monitoring labs working with brain organoids to try to begin the ‘building of a philosophical framework’ that future guidelines and legislation could be built upon.[26]
See also
References
- Lancaster MA, Renner M, Martin CA, Wenzel D, Bicknell LS, Hurles ME, Homfray T, Penninger JM, Jackson AP, Knoblich JA (September 2013). "Cerebral organoids model human brain development and microcephaly". Nature. 501 (7467): 373–9. Bibcode:2013Natur.501..373L. doi:10.1038/nature12517. PMC 3817409. PMID 23995685.
- "Growing model brains: An embryonic idea". The Economist. 2013-08-31. Retrieved 2013-09-07.
- Di Lullo E, Kriegstein AR (October 2017). "The use of brain organoids to investigate neural development and disease". Nature Reviews. Neuroscience. 18 (10): 573–584. doi:10.1038/nrn.2017.107. PMC 5667942. PMID 28878372.
Table 1: Protocols for brain organoid generation
- Purves D, Augustine GJ, Fitzpatrick D, Hall WC, LaMantia AS, White LE, eds. (2007). Neuroscience (4th ed.). New York: W. H. Freeman. ISBN 978-0-87893-697-7.
- Lelkes PI, Unsworth BR (2002). "Neuroectodermal Cell Culture: Endocrine Cells". In Atala A, Lanza R (eds.). Methods of tissue engineering (1st ed.). San Diego, CA: Academic Press. p. 381. ISBN 978-0-12-436636-7.
- Chambers SM, Tchieu J, Studer L (October 2013). "Build-a-brain". Cell Stem Cell. 13 (4): 377–8. doi:10.1016/j.stem.2013.09.010. PMID 24094317.
- Mansour, Abed AlFatah; Gonçalves, J Tiago; Bloyd, Cooper W; Li, Hao; Fernandes, Sarah; Quang, Daphne; Johnston, Stephen; Parylak, Sarah L; Jin, Xin (May 2018). "An in vivo model of functional and vascularized human brain organoids". Nature Biotechnology. 36 (5): 432–441. doi:10.1038/nbt.4127. ISSN 1087-0156. PMC 6331203. PMID 29658944.
- Daviaud, Nicolas; Friedel, Roland H.; Zou, Hongyan (November 2018). "Vascularization and Engraftment of Transplanted Human Cerebral Organoids in Mouse Cortex". eNeuro. 5 (6): ENEURO.0219–18.2018. doi:10.1523/ENEURO.0219-18.2018. ISSN 2373-2822. PMC 6243198. PMID 30460331.
- Opitz JM, Holt MC (1990). "Microcephaly: general considerations and aids to nosology". Journal of Craniofacial Genetics and Developmental Biology. 10 (2): 175–204. PMID 2211965.
- Gonzalez C, Armijo E, Bravo-Alegria J, Becerra-Calixto A, Mays CE, Soto C (August 2018). "Modeling amyloid beta and tau pathology in human cerebral organoids". Molecular Psychiatry. 23 (12): 2363–2374. doi:10.1038/s41380-018-0229-8. PMC 6594704. PMID 30171212.
- Swerdlow RH (September 2007). "Pathogenesis of Alzheimer's disease". Clinical Interventions in Aging. 2 (3): 347–59. PMC 2685260. PMID 18044185.
- Laurijssens B, Aujard F, Rahman A (September 2013). "Animal models of Alzheimer's disease and drug development". Drug Discovery Today: Technologies. 10 (3): e319-27. doi:10.1016/j.ddtec.2012.04.001. PMID 24050129.
- Wang H (2018-06-08). "Modeling Neurological Diseases With Human Brain Organoids". Frontiers in Synaptic Neuroscience. 10: 15. doi:10.3389/fnsyn.2018.00015. PMC 6002496. PMID 29937727.
- Rubenstein JL (April 2010). "Three hypotheses for developmental defects that may underlie some forms of autism spectrum disorder". Current Opinion in Neurology. 23 (2): 118–23. doi:10.1097/WCO.0b013e328336eb13. PMID 20087182.
- Pașca, Anca M.; Park, Jin-Young; Shin, Hyun-Woo; Qi, Qihao; Revah, Omer; Krasnoff, Rebecca; O’Hara, Ruth; Willsey, A. Jeremy; Palmer, Theo D. (2019-05-06). "Human 3D cellular model of hypoxic brain injury of prematurity". Nature Medicine. 25 (5): 784–791. doi:10.1038/s41591-019-0436-0. ISSN 1078-8956. PMC 7020938. PMID 31061540.
- Zou, Hongyan; Friedel, Roland H.; Chevalier, Clément; Daviaud, Nicolas (2019). "Distinct Vulnerability and Resilience of Human Neuroprogenitor Subtypes in Cerebral Organoid Model of Prenatal Hypoxic Injury". Frontiers in Cellular Neuroscience. 13: 336. doi:10.3389/fncel.2019.00336. ISSN 1662-5102. PMC 6682705. PMID 31417360.
- Vogel G (August 2013). "Neurodevelopment. Lab dishes up mini-brains". Science. 341 (6149): 946–7. doi:10.1126/science.341.6149.946. PMID 23990534.
- Reichardt A, Polchow B, Shakibaei M, Henrich W, Hetzer R, Lueders C (14 June 2013). "Large scale expansion of human umbilical cord cells in a rotating bed system bioreactor for cardiovascular tissue engineering applications". The Open Biomedical Engineering Journal. 7 (1): 50–61. doi:10.2174/1874120701307010050. PMC 3706833. PMID 23847691.
- Church G. "The future of genetic codes and BRAIN codes". YouTube. NIHvcast. Retrieved 10 February 2017.
- Bershteyn M, Kriegstein AR (September 2013). "Cerebral organoids in a dish: progress and prospects". Cell. 155 (1): 19–20. doi:10.1016/j.cell.2013.09.010. PMC 5127703. PMID 24074857.
- Sakayori N, Kikkawa T, Osumi N (October 2012). "Reduced proliferation and excess astrogenesis of Pax6 heterozygous neural stem/progenitor cells". Neuroscience Research. 74 (2): 116–21. doi:10.1016/j.neures.2012.08.004. PMID 22944581.
- Eiraku M, Takata N, Ishibashi H, Kawada M, Sakakura E, Okuda S, Sekiguchi K, Adachi T, Sasai Y (April 2011). "Self-organizing optic-cup morphogenesis in three-dimensional culture". Nature. 472 (7341): 51–6. Bibcode:2011Natur.472...51E. doi:10.1038/nature09941. PMID 21475194.
- Lavazza A, Massimini M (September 2018). "Cerebral organoids: ethical issues and consciousness assessment". Journal of Medical Ethics. 44 (9): 606–610. doi:10.1136/medethics-2017-104555. PMID 29491041.
- Quadrato G, Nguyen T, Macosko EZ, Sherwood JL, Min Yang S, Berger DR, Maria N, Scholvin J, Goldman M, Kinney JP, Boyden ES, Lichtman JW, Williams ZM, McCarroll SA, Arlotta P (May 2017). "Cell diversity and network dynamics in photosensitive human brain organoids". Nature. 545 (7652): 48–53. Bibcode:2017Natur.545...48Q. doi:10.1038/nature22047. PMC 5659341. PMID 28445462.
- "Human Brain Organoids: the Science, the Ethics". International Neuroethics Society. June 2018.
- Gogol A (October 2018). "A human brain model in a petri dish?". EurekAlert!.