Benzocyclobutene
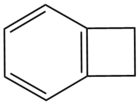
Benzocyclobutene (BCB) is a benzene ring fused to a cyclobutane ring. It has chemical formula C8H8.[1]
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Bicyclo[4.2.0]octa-1,3,5-triene | |
| Other names
Benzocyclobutane BCB Benzocyclobutene (not in accordance with IUPAC nomenclature) | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.161.355 |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H8 | |
| Molar mass | 104.152 g·mol−1 |
| Density | 0.957 g/cm3 |
| Boiling point | 150 °C (302 °F; 423 K) |
Refractive index (nD) |
1.541 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
BCB is frequently used to create photosensitive polymers. BCB-based polymer dielectrics may be spun on or applied to various substrates for use in Micro Electro-Mechanical Systems (MEMS) and microelectronics processing. Applications include wafer bonding, optical interconnects, low-κ dielectrics, or even intracortical neural implants.
Reactions
Benzocyclobutene is a strained system which, upon heating to approximately 180 °C, causes the cyclobutene to undergo a conrotatory ring-opening reaction, forming o-xylylene. Since this process destroys the aromaticity of the benzene ring, the reverse reaction is highly favored.

o-Xylylenes generated in this way have been used prolifically in cycloaddition reactions, which restore the aromaticity to the benzene ring, while forming a new annulated species.[2]
Uses
The benzocyclobutene moiety has also appeared in a number of chemical compounds with pharmacological properties such as ivabradine and S33005. Additionally, the benzocyclobutene analog of 2C-B has been prepared[3] and a benzocyclobutene-derived amphetamine has been patented.[4]
See also
References
- 164410 Benzocyclobutene 98%
- Mehta, G.; Kotha, S. (2001). "Recent chemistry of benzocyclobutenes" (PDF). Tetrahedron Lett. 57 (4): 625–659. doi:10.1016/s0040-4020(00)00958-3.
- "The Binding Database".
- US 3149159, "Substituted 7-aminoalkylbicyclo-[4. 2. 0]octa-1,3,5-trienes"