Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.
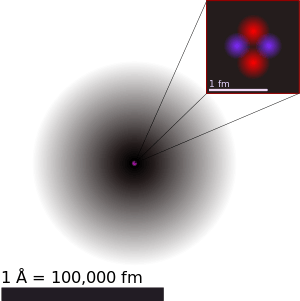
The word atom comes from the Ancient Greek adjective atomos, meaning "indivisible".[1] 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. Around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called "uncuttable atom" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all.
Since atoms were found to be divisible, physicists later invented the term "elementary particles" to describe the "uncuttable", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.
History
Philosophical atomism
The idea that matter is made up of discrete units is a very old idea, appearing in many ancient cultures such as Greece and India. The word "atom" (Greek: ἄτομος; atomos), meaning "uncuttable", was coined by the Pre-Socratic Greek philosophers Leucippus and his pupil Democritus (c. 460 – c. 370 BC).[2][3][4][5] Democritus taught that atoms were infinite in number, uncreated, and eternal, and that the qualities of an object result from the kind of atoms that compose it.[3][4][5] Democritus's atomism was refined and elaborated by the later Greek philosopher Epicurus (341 – 270 BC), and by the Roman Epicurean poet Lucretius (c. 99 – c. 55 BC).[4][5] During the Early Middle Ages, atomism was mostly forgotten in western Europe.[4] During the 12th century, atomism became known again in western Europe through references to it in the newly-rediscovered writings of Aristotle.[4]
In the 14th century, the rediscovery of major works describing atomist teachings, including Lucretius's De rerum natura and Diogenes Laërtius's Lives and Opinions of Eminent Philosophers, led to increased scholarly attention on the subject.[4] Nonetheless, because atomism was associated with the philosophy of Epicureanism, which contradicted orthodox Christian teachings, belief in atoms was not considered acceptable by most European philosophers.[4] The French Catholic priest Pierre Gassendi (1592 – 1655) revived Epicurean atomism with modifications, arguing that atoms were created by God and, though extremely numerous, are not infinite.[4][5] Gassendi's modified theory of atoms was popularized in France by the physician François Bernier (1620 – 1688) and in England by the natural philosopher Walter Charleton (1619 – 1707).[4] The chemist Robert Boyle (1627 – 1691) and the physicist Isaac Newton (1642 – 1727) both defended atomism and, by the end of the 17th century, it had become accepted by portions of the scientific community.[4]
John Dalton
Near the end of the 18th century, two laws about chemical reactions emerged without referring to the notion of an atomic theory. The first was the law of conservation of mass, closely associated with the work of Antoine Lavoisier, which states that the total mass in a chemical reaction remains constant (that is, the reactants have the same mass as the products).[6] The second was the law of definite proportions. First established by the French chemist Joseph Louis Proust in 1799,[7] this law states that if a compound is broken down into its constituent chemical elements, then the masses of the constituents will always have the same proportions by weight, regardless of the quantity or source of the original substance.
John Dalton studied and expanded upon this previous work and defended a new idea, later known as the law of multiple proportions: if the same two elements can be combined to form a number of different compounds, then the ratios of the masses of the two elements in their various compounds will be represented by small whole numbers. This is a common pattern in chemical reactions that was observed by Dalton and other chemists at the time. Dalton himself discovered that oxygen will combine with a certain amount of nitrous gas to form nitric acid, or twice that amount to form nitrous acid—a ratio of 1:2.[8] Joseph Proust discovered that 100 parts of iron will combine with either 28 or 42 parts of oxygen[lower-alpha 1][9][10] (a ratio of 2:3); and that 119 parts of tin will combine with either 16 or 32 parts of oxygen[11] (a ratio of 1:2). Dalton found that an atomic theory of matter could elegantly explain this pattern, as well as Proust's law of definite proportions. In the case of Proust's tin oxides, one tin atom will combine with either one or two oxygen atoms to form either the first or the second oxide of tin.[12]
Dalton believed atomic theory could also explain why water absorbed different gases in different proportions—for example, he found that water absorbed carbon dioxide far better than it absorbed nitrogen.[13] Dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles. Indeed, carbon dioxide molecules (CO2) are heavier and larger than nitrogen molecules (N2).
Dalton proposed that each chemical element is composed of atoms of a single, unique type, and though they cannot be altered or destroyed by chemical means, they can combine to form more complex structures (chemical compounds). This marked the first truly scientific theory of the atom, since Dalton reached his conclusions by experimentation and examination of the results in an empirical fashion.

In 1803 Dalton orally presented his first list of relative atomic weights for a number of substances. This paper was published in 1805, but he did not discuss there exactly how he obtained these figures.[13] The method was first revealed in 1807 by his acquaintance Thomas Thomson, in the third edition of Thomson's textbook, A System of Chemistry. Finally, Dalton published a full account in his own textbook, A New System of Chemical Philosophy, 1808 and 1810.
Dalton estimated the atomic weights according to the mass ratios in which they combined, with the hydrogen atom taken as unity. However, Dalton did not conceive that with some elements atoms exist in molecules—e.g. pure oxygen exists as O2. He also mistakenly believed that the simplest compound between any two elements is always one atom of each (so he thought water was HO, not H2O).[14] This, in addition to the crudity of his equipment, flawed his results. For instance, in 1803 he believed that oxygen atoms were 5.5 times heavier than hydrogen atoms, because in water he measured 5.5 grams of oxygen for every 1 gram of hydrogen and believed the formula for water was HO. Adopting better data, in 1806 he concluded that the atomic weight of oxygen must actually be 7 rather than 5.5, and he retained this weight for the rest of his life. Others at this time had already concluded that the oxygen atom must weigh 8 relative to hydrogen equals 1, if one assumes Dalton's formula for the water molecule (HO), or 16 if one assumes the modern water formula (H2O).[15]
Avogadro
The flaw in Dalton's theory was corrected in principle in 1811 by Amedeo Avogadro. Avogadro had proposed that equal volumes of any two gases, at equal temperature and pressure, contain equal numbers of molecules (in other words, the mass of a gas's particles does not affect the volume that it occupies).[16] Avogadro's law allowed him to deduce the diatomic nature of numerous gases by studying the volumes at which they reacted. For instance: since two liters of hydrogen will react with just one liter of oxygen to produce two liters of water vapor (at constant pressure and temperature), it meant a single oxygen molecule splits in two in order to form two particles of water. Thus, Avogadro was able to offer more accurate estimates of the atomic mass of oxygen and various other elements, and made a clear distinction between molecules and atoms.
Brownian Motion
In 1827, the British botanist Robert Brown observed that dust particles inside pollen grains floating in water constantly jiggled about for no apparent reason. In 1905, Albert Einstein theorized that this Brownian motion was caused by the water molecules continuously knocking the grains about, and developed a hypothetical mathematical model to describe it.[17] This model was validated experimentally in 1908 by French physicist Jean Perrin, thus providing additional validation for particle theory (and by extension atomic theory).
Discovery of subatomic particles
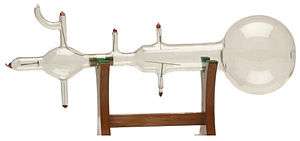
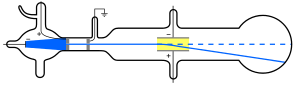
Atoms were thought to be the smallest possible division of matter until 1897 when J.J. Thomson discovered the electron through his work on cathode rays.[18]
A Crookes tube is a sealed glass container in which two electrodes are separated by a vacuum. When a voltage is applied across the electrodes, cathode rays are generated, creating a glowing patch where they strike the glass at the opposite end of the tube. Through experimentation, Thomson discovered that the rays could be deflected by an electric field (in addition to magnetic fields, which was already known). He concluded that these rays, rather than being a form of light, were composed of very light negatively charged particles he called "corpuscles" (they would later be renamed electrons by other scientists). He measured the mass-to-charge ratio and discovered it was 1800 times smaller than that of hydrogen, the smallest atom. These corpuscles were a particle unlike any other previously known.
Thomson suggested that atoms were divisible, and that the corpuscles were their building blocks.[19] To explain the overall neutral charge of the atom, he proposed that the corpuscles were distributed in a uniform sea of positive charge; this was the plum pudding model[20] as the electrons were embedded in the positive charge like raisins in a plum pudding (although in Thomson's model they were not stationary).
Discovery of the nucleus
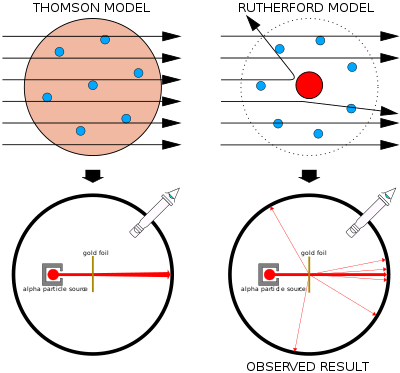
Left: Expected results: alpha particles passing through the plum pudding model of the atom with negligible deflection.
Right: Observed results: a small portion of the particles were deflected by the concentrated positive charge of the nucleus.
Thomson's plum pudding model was disproved in 1909 by one of his former students, Ernest Rutherford, who discovered that most of the mass and positive charge of an atom is concentrated in a very small fraction of its volume, which he assumed to be at the very center.
In the Geiger–Marsden experiment, Hans Geiger and Ernest Marsden (colleagues of Rutherford working at his behest) shot alpha particles at thin sheets of metal and measured their deflection through the use of a fluorescent screen.[21] Given the very small mass of the electrons, the high momentum of the alpha particles, and the low concentration of the positive charge of the plum pudding model, the experimenters expected all the alpha particles to pass through the metal foil without significant deflection. To their astonishment, a small fraction of the alpha particles experienced heavy deflection. Rutherford concluded that the positive charge of the atom must be concentrated in a very tiny volume to produce an electric field sufficiently intense to deflect the alpha particles so strongly.
This led Rutherford to propose a planetary model in which a cloud of electrons surrounded a small, compact nucleus of positive charge. Only such a concentration of charge could produce the electric field strong enough to cause the heavy deflection.[22]
First steps toward a quantum physical model of the atom
The planetary model of the atom had two significant shortcomings. The first is that, unlike planets orbiting a sun, electrons are charged particles. An accelerating electric charge is known to emit electromagnetic waves according to the Larmor formula in classical electromagnetism. An orbiting charge should steadily lose energy and spiral toward the nucleus, colliding with it in a small fraction of a second. The second problem was that the planetary model could not explain the highly peaked emission and absorption spectra of atoms that were observed.
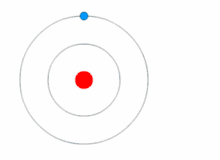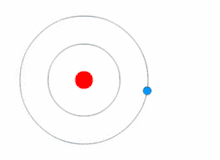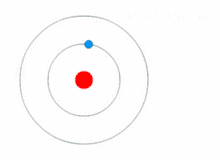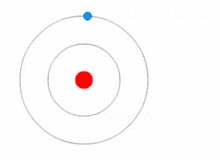
Quantum theory revolutionized physics at the beginning of the 20th century, when Max Planck and Albert Einstein postulated that light energy is emitted or absorbed in discrete amounts known as quanta (singular, quantum). In 1913, Niels Bohr incorporated this idea into his Bohr model of the atom, in which an electron could only orbit the nucleus in particular circular orbits with fixed angular momentum and energy, its distance from the nucleus (i.e., their radii) being proportional to its energy.[23] Under this model an electron could not spiral into the nucleus because it could not lose energy in a continuous manner; instead, it could only make instantaneous "quantum leaps" between the fixed energy levels.[23] When this occurred, light was emitted or absorbed at a frequency proportional to the change in energy (hence the absorption and emission of light in discrete spectra).[23]
Bohr's model was not perfect. It could only predict the spectral lines of hydrogen; it couldn't predict those of multielectron atoms. Worse still, as spectrographic technology improved, additional spectral lines in hydrogen were observed which Bohr's model couldn't explain. In 1916, Arnold Sommerfeld added elliptical orbits to the Bohr model to explain the extra emission lines, but this made the model very difficult to use, and it still couldn't explain more complex atoms.
Discovery of isotopes
While experimenting with the products of radioactive decay, in 1913 radiochemist Frederick Soddy discovered that there appeared to be more than one element at each position on the periodic table.[24] The term isotope was coined by Margaret Todd as a suitable name for these elements.
That same year, J.J. Thomson conducted an experiment in which he channeled a stream of neon ions through magnetic and electric fields, striking a photographic plate at the other end. He observed two glowing patches on the plate, which suggested two different deflection trajectories. Thomson concluded this was because some of the neon ions had a different mass.[25] The nature of this differing mass would later be explained by the discovery of neutrons in 1932.
Discovery of nuclear particles
In 1917 Rutherford bombarded nitrogen gas with alpha particles and observed hydrogen nuclei being emitted from the gas (Rutherford recognized these, because he had previously obtained them bombarding hydrogen with alpha particles, and observing hydrogen nuclei in the products). Rutherford concluded that the hydrogen nuclei emerged from the nuclei of the nitrogen atoms themselves (in effect, he had split a nitrogen).[26]
From his own work and the work of his students Bohr and Henry Moseley, Rutherford knew that the positive charge of any atom could always be equated to that of an integer number of hydrogen nuclei. This, coupled with the atomic mass of many elements being roughly equivalent to an integer number of hydrogen atoms - then assumed to be the lightest particles - led him to conclude that hydrogen nuclei were singular particles and a basic constituent of all atomic nuclei. He named such particles protons. Further experimentation by Rutherford found that the nuclear mass of most atoms exceeded that of the protons it possessed; he speculated that this surplus mass was composed of previously-unknown neutrally charged particles, which were tentatively dubbed "neutrons".
In 1928, Walter Bothe observed that beryllium emitted a highly penetrating, electrically neutral radiation when bombarded with alpha particles. It was later discovered that this radiation could knock hydrogen atoms out of paraffin wax. Initially it was thought to be high-energy gamma radiation, since gamma radiation had a similar effect on electrons in metals, but James Chadwick found that the ionization effect was too strong for it to be due to electromagnetic radiation, so long as energy and momentum were conserved in the interaction. In 1932, Chadwick exposed various elements, such as hydrogen and nitrogen, to the mysterious "beryllium radiation", and by measuring the energies of the recoiling charged particles, he deduced that the radiation was actually composed of electrically neutral particles which could not be massless like the gamma ray, but instead were required to have a mass similar to that of a proton. Chadwick now claimed these particles as Rutherford's neutrons.[27] For his discovery of the neutron, Chadwick received the Nobel Prize in 1935.
Quantum physical models of the atom
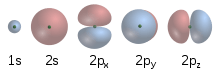
In 1924, Louis de Broglie proposed that all moving particles—particularly subatomic particles such as electrons—exhibit a degree of wave-like behavior. Erwin Schrödinger, fascinated by this idea, explored whether or not the movement of an electron in an atom could be better explained as a wave rather than as a particle. Schrödinger's equation, published in 1926,[28] describes an electron as a wavefunction instead of as a point particle. This approach elegantly predicted many of the spectral phenomena that Bohr's model failed to explain. Although this concept was mathematically convenient, it was difficult to visualize, and faced opposition.[29] One of its critics, Max Born, proposed instead that Schrödinger's wavefunction described not the electron but rather all its possible states, and thus could be used to calculate the probability of finding an electron at any given location around the nucleus.[30] This reconciled the two opposing theories of particle versus wave electrons and the idea of wave–particle duality was introduced. This theory stated that the electron may exhibit the properties of both a wave and a particle. For example, it can be refracted like a wave, and has mass like a particle.[31]
A consequence of describing electrons as waveforms is that it is mathematically impossible to simultaneously derive the position and momentum of an electron. This became known as the Heisenberg uncertainty principle after the theoretical physicist Werner Heisenberg, who first described it and published it in 1927.[32] This invalidated Bohr's model, with its neat, clearly defined circular orbits. The modern model of the atom describes the positions of electrons in an atom in terms of probabilities. An electron can potentially be found at any distance from the nucleus, but, depending on its energy level, exists more frequently in certain regions around the nucleus than others; this pattern is referred to as its atomic orbital. The orbitals come in a variety of shapes-sphere, dumbbell, torus, etc.-with the nucleus in the middle.[33]
See also
- Spectroscopy
- History of the molecule
- Discoveries of the chemical elements
- Introduction to quantum mechanics
- Kinetic theory of gases
- Atomism
- The Physical Principles of the Quantum Theory
Footnotes
- These are Proust's measurements. Given atomic weights known today, more accurate figures would be 28.6 and 42.9 parts of oxygen for 100 parts of iron.
- Berryman, Sylvia, "Ancient Atomism", Stanford Encyclopedia of Philosophy (Fall 2008 Edition), Edward N. Zalta (ed.)
- Pullman, Bernard (1998). The Atom in the History of Human Thought. Oxford, England: Oxford University Press. pp. 31–33. ISBN 978-0-19-515040-7.CS1 maint: ref=harv (link)
- Kenny, Anthony (2004). Ancient Philosophy. A New History of Western Philosophy. 1. Oxford, England: Oxford University Press. pp. 26–28. ISBN 0-19-875273-3.CS1 maint: ref=harv (link)
- Pyle, Andrew (2010). "Atoms and Atomism". In Grafton, Anthony; Most, Glenn W.; Settis, Salvatore (eds.). The Classical Tradition. Cambridge, Massachusetts and London, England: The Belknap Press of Harvard University Press. pp. 103–104. ISBN 978-0-674-03572-0.CS1 maint: ref=harv (link)
- Cohen, Henri; Lefebvre, Claire, eds. (2017). Handbook of Categorization in Cognitive Science (Second ed.). Amsterdam, The Netherlands: Elsevier. p. 427. ISBN 978-0-08-101107-2.CS1 maint: ref=harv (link)
- Weisstein, Eric W. "Lavoisier, Antoine (1743-1794)". scienceworld.wolfram.com. Retrieved 2009-08-01.
- Proust, Joseph Louis. "Researches on Copper", excerpted from Ann. chim. 32, 26-54 (1799) [as translated and reproduced in Henry M. Leicester and Herbert S. Klickstein, A Source Book in Chemistry, 1400–1900 (Cambridge, Massachusetts: Harvard, 1952)]. Retrieved on August 29, 2007.
- Millington (1906). John Dalton, pp. 76-77
- Millington (1906). John Dalton, p. 113
- Joseph Louis Proust (1794). "Recherches sur le Bleu de Prusse" [Research on Prussian Blue]. Journal de Physique, de Chimie, et d'Histoire Naturelle (in French). 2: 334–341.
- Chemical News and Journal of Industrial Science, Volume 64 (1891), pp. 33-34
- Melsen (1952). From Atomos to Atom, p. 137
- Dalton, John. "On the Absorption of Gases by Water and Other Liquids", in Memoirs of the Literary and Philosophical Society of Manchester. 1803. Retrieved on August 29, 2007.
- Johnson, Chris. "Avogadro - his contribution to chemistry". Archived from the original on 2002-07-10. Retrieved 2009-08-01.
- Alan J. Rocke (1984). Chemical Atomism in the Nineteenth Century. Columbus: Ohio State University Press.
- Avogadro, Amedeo (1811). "Essay on a Manner of Determining the Relative Masses of the Elementary Molecules of Bodies, and the Proportions in Which They Enter into These Compounds". Journal de Physique. 73: 58–76.
- Einstein, A. (1905). "Über die von der molekularkinetischen Theorie der Wärme geforderte Bewegung von in ruhenden Flüssigkeiten suspendierten Teilchen" (PDF). Annalen der Physik. 322 (8): 549–560. Bibcode:1905AnP...322..549E. doi:10.1002/andp.19053220806. hdl:10915/2785.
- Thomson, J.J. (1897). "Cathode rays" ([facsimile from Stephen Wright, Classical Scientific Papers, Physics (Mills and Boon, 1964)]). Philosophical Magazine. 44 (269): 293. doi:10.1080/14786449708621070.
- Whittaker, E. T. (1951), A history of the theories of aether and electricity. Vol 1, Nelson, London
- Thomson, J.J. (1904). "On the Structure of the Atom: an Investigation of the Stability and Periods of Oscillation of a number of Corpuscles arranged at equal intervals around the Circumference of a Circle; with Application of the Results to the Theory of Atomic Structure". Philosophical Magazine. 7 (39): 237. doi:10.1080/14786440409463107.
- Geiger, H (1910). "The Scattering of the α-Particles by Matter". Proceedings of the Royal Society. A 83: 492–504.
- Rutherford, Ernest (1911). "The Scattering of α and β Particles by Matter and the Structure of the Atom" (PDF). Philosophical Magazine. 21 (4): 669. Bibcode:2012PMag...92..379R. doi:10.1080/14786435.2011.617037.
- Bohr, Niels (1913). "On the constitution of atoms and molecules" (PDF). Philosophical Magazine. 26 (153): 476–502. Bibcode:1913PMag...26..476B. doi:10.1080/14786441308634993.
- "Frederick Soddy, The Nobel Prize in Chemistry 1921". Nobel Foundation. Retrieved 2008-01-18.
- Thomson, J.J. (1913). "Rays of positive electricity". Proceedings of the Royal Society. A 89 (607): 1–20. Bibcode:1913RSPSA..89....1T. doi:10.1098/rspa.1913.0057. [as excerpted in Henry A. Boorse & Lloyd Motz, The World of the Atom, Vol. 1 (New York: Basic Books, 1966)]. Retrieved on August 29, 2007.
- Rutherford, Ernest (1919). "Collisions of alpha Particles with Light Atoms. IV. An Anomalous Effect in Nitrogen". Philosophical Magazine. 37 (222): 581. doi:10.1080/14786440608635919.
- Chadwick, James (1932). "Possible Existence of a Neutron" (PDF). Nature. 129 (3252): 312. Bibcode:1932Natur.129Q.312C. doi:10.1038/129312a0.
- Schrödinger, Erwin (1926). "Quantisation as an Eigenvalue Problem". Annalen der Physik. 81 (18): 109–139. Bibcode:1926AnP...386..109S. doi:10.1002/andp.19263861802.
- Mahanti, Subodh. "Erwin Schrödinger: The Founder of Quantum Wave Mechanics". Archived from the original on 2009-04-17. Retrieved 2009-08-01.
- Mahanti, Subodh. "Max Born: Founder of Lattice Dynamics". Archived from the original on 2009-01-22. Retrieved 2009-08-01.
- Greiner, Walter. "Quantum Mechanics: An Introduction". Retrieved 2010-06-14.
- Heisenberg, W. (1927). "Über den anschaulichen Inhalt der quantentheoretischen Kinematik und Mechanik". Zeitschrift für Physik (in German). 43 (3–4): 172–198. Bibcode:1927ZPhy...43..172H. doi:10.1007/BF01397280.
- Milton Orchin; Roger Macomber; Allan Pinhas; R. Wilson. "The Vocabulary and Concepts of Organic Chemistry, Second Edition" (PDF). Retrieved 2010-06-14.
Bibliography
- Andrew G. van Melsen (1960) [First published 1952]. From Atomos to Atom: The History of the Concept Atom. Translated by Henry J. Koren. Dover Publications. ISBN 0-486-49584-1.
- J. P. Millington (1906). John Dalton. J. M. Dent & Co. (London); E. P. Dutton & Co. (New York).
Further reading
- Bernard Pullman (1998) The Atom in the History of Human Thought, trans. by Axel Reisinger. Oxford Univ. Press.
- Eric Scerri (2007) The Periodic Table, Its Story and Its Significance, Oxford University Press, New York.
- Charles Adolphe Wurtz (1881) The Atomic Theory, D. Appleton and Company, New York.
- Alan J. Rocke (1984) Chemical Atomism in the Nineteenth Century: From Dalton to Cannizzaro, Ohio State University Press, Columbus (open access full text at http://digital.case.edu/islandora/object/ksl%3Ax633gj985).
External links
| Wikiquote has quotations related to: Atomic theory |
- Atomism by S. Mark Cohen.
- Atomic Theory - detailed information on atomic theory with respect to electrons and electricity.