Acryloyl chloride
Acryloyl chloride, also known as 2-propenoyl chloride or acrylic acid chloride, is the organic compound with the formula CH2=CHCO(Cl). It is a colorless liquid, although aged samples appear yellow. Although it belongs to the acid chlorides group of compounds, its synthesis and reactivity are somewhat different.[1]
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Prop-2-enoyl chloride | |
| Other names
2-Propenoyl chloride Acryloyl chloride | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.011.272 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C3H3ClO | |
| Molar mass | 90.51 g·mol−1 |
| Density | 1.119 g/cm3 |
| Melting point | N/A |
| Boiling point | 75.0 °C (167.0 °F; 348.1 K) |
| Hazards | |
| GHS pictograms |  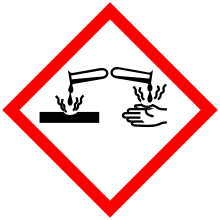  |
| GHS Signal word | Danger |
GHS hazard statements |
H225, H302, H314, H330 |
| P210, P280, P301+330+331, P303+361+353, P305+351+338, P310 | |
| Flash point | −4 °C (25 °F; 269 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation
Acryloyl chloride can be prepared by treating acrylic acid with acid chlorides, originally benzoyl chloride.[2] Conventional one-pot chlorinating agents, e.g. thionyl chloride, phosphorus trichloride, are ineffective. Flow conditions allows use of a broadened range of chlorinating agents including oxalyl chloride and thionyl chloride.[1] [3]
Reactions
This compound undergoes the reactions common for acid chlorides. For example, it reacts readily with water, producing acrylic acid. When treated with sodium salts of carboxylic acids, the anhydride is formed. Reactions with alcohols and amines gives esters and amides, respectively. Acryloyl chloride is most commonly employed for the introduction of acrylic groups into other compounds, e.g. the preparation of acrylate monomers and polymers.[4]
References
- PatentStorm LLC (2006). "Process for the manufacture of acryloyl chloride". PatentStorm LLC. Archived from the original on January 14, 2013. Retrieved December 21, 2007.
- Stempel, Guido H., Jr.; Cross, Robert P.; Mariella, Raymond P. (1950). "Preparation of acrylyl chloride". Journal of the American Chemical Society. 72: 2299–2300. doi:10.1021/ja01161a527.CS1 maint: multiple names: authors list (link)
- Movsisyan, Marine; Heugebaert, Thomas S. A.; Dams, Rudy; Stevens, Christian V. (9 August 2016). "Safe, Selective, and High-Yielding Synthesis of Acryloyl Chloride in a Continuous-Flow System". ChemSusChem. 9 (15): 1945–1952. doi:10.1002/cssc.201600348.
- Takashi Ohara, Takahisa Sato, Noboru Shimizu, Günter Prescher Helmut Schwind, Otto Weiberg, Klaus Marten, Helmut Greim (2003). "Acrylic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a01_161.pub2.CS1 maint: multiple names: authors list (link)
See also
- Methacryloyl chloride
- List of extremely hazardous substances