Acetylenedicarboxylic acid
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
But-2-ynedioic acid | |
| Other names
2-Butynedioic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.005.033 |
| KEGG | |
PubChem CID |
|
| |
| |
| Properties | |
| C4H2O4 | |
| Molar mass | 114.06 g·mol−1 |
| Appearance | Crystalline solid |
| Melting point | 175 to 176 °C (347 to 349 °F; 448 to 449 K) (decomposes)[2] 180–187 °C (decomposes)[1] |
| Conjugate base | Hydrogenacetylenedicarboxylate (chemical formula HC4O4−) |
| Hazards | |
| R-phrases (outdated) | R25 R36/37/38 |
| S-phrases (outdated) | S26 S45 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
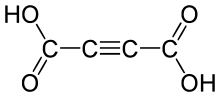
Acetylenedicarboxylic acid or butynedioic acid is an organic compound (a dicarboxylic acid) with the formula C4H2O4 or HO2C-C≡C-CO2H. It is a crystalline solid that is soluble in diethyl ether.
The removal of two protons yields the acetylenedicarboxylate dianion C4O42−, which consists only of carbon and oxygen, making it an oxocarbon anion. Partial ionization yields the monovalent hydrogenacetylenedicarboxylate anion HC4O4−.
The acid was first described in 1877 by Polish chemist Ernest Bandrowski.[2][3][4] It can be obtained by treating α,β-dibromosuccinic acid with potassium hydroxide KOH in methanol or ethanol. The reaction yields potassium bromide and potassium acetylenedicarboxylate. The salts are separated and the latter is treated with sulfuric acid.[2]
Acetylenedicarboxylic acid is used in the synthesis of dimethyl acetylenedicarboxylate, an important laboratory reagent. The acid is commonly traded as a laboratory chemical. It can also be reacted with sulfur tetrafluoride to produce hexafluoro-2-butyne, a powerful dienophile for use in Diels-Alder reactions.
Anions and salts
Hydrogenacetylenedicarboxylate (often abbreviated as Hadc or HADC) is a monovalent anion of acetylenedicarboxylic acid with the formula C4HO4− or HO2C-C≡C-CO2−. The anion can be derived from acetylenedicarboxylic acid by removal of a single proton or from the acetylenedicarboxylate dianion by addition of a proton. The name is also used for any salt of this anion. Salts of this anion are of interest in crystallography because they contain unusually short and strong hydrogen bonds. In many crystalline salts (with the exception of the lithium one), the HADC units form linear chains connected by strong hydrogen bonds. Each carboxylate group is usually planar; but the two groups may lie in different planes due to rotation about the C-C bonds. They are coplanar in the hydrated salts NaHC4O4·2H20 and CsHC4O4·2H20, nearly coplanar in the guanidinium salt [C(NH2)3]+ · C4HO4−, but off by 60 degrees or more in other salts such as anhydrous potassium hydrogenacetylenedicarboxylate.[5]
Potassium hydrogenacetylenedicarboxylate is a potassium salt of HADC with chemical formula KC4HO4 or K+·HC4O4−, often abbreviated as KHadc. It is often called potassium hydrogen acetylenedicarboxylate or monopotassium acetylenedicarboxylate. The salt can be obtained from acetylenedicarboxylic acid and is a common laboratory starting material for the synthesis of other derivatives of that acid. In the crystalline form, the hydrogenacetylenedicarboxylate anions are joined into linear chains by uncommonly short hydrogen bonds.[6][7]
Acetylenedicarboxylate (often abbreviated as ADC or adc) is a divalent anion with formula C4O42− or [O2C–C≡C–CO2]2−; or any salt or ester thereof. The anion can be derived from acetylenedicarboxylic acid by the loss of two protons. It is one of several oxocarbon anions which, like carbonate CO32− and oxalate C2O42−, consist solely of carbon and oxygen. The ADC anion can at as a ligand in organometallic complexes, e.g. the blue polymeric complex with copper(II) and 2,2'-bipyridine, [Cu2+ · C4O42− · (C5H4N)2]n.[8][9] Thallium acetylenedicarboxylate (Tl2C4O4) decomposes at 195 °C (383 °F), leaving a residue of pyrophoric thallium powder.[10]

See also
References
- 1 2 "Acetylenedicarboxylic acid". Sigma-Aldrich.
- 1 2 3 Abbott, T. W.; Arnold, R. T.; Thompson, R. B. "Acetylenedicarboxylic acid". Organic Syntheses. ; Collective Volume, 2, p. 10
- ↑ Bandrowski, E. (1877). "Ueber Acetylendicarbonsäure". Berichte der deutschen chemischen Gesellschaft. 10: 838. doi:10.1002/cber.187701001231.
- ↑ E. Bandrowski (1879). "Weitere Beiträge zur Kenntniss der Acetylendicarbonsäure". Berichte der deutschen chemischen Gesellschaft. 12 (2): 2212–2216. doi:10.1002/cber.187901202261.
- ↑ Leban, I; Rupnik, A (1992). "Structure of guanidinium hydrogen acetylenedicarboxylate, CH6N3+.C4HO4−". Acta Crystallographica Section C. 48 (5): 821. doi:10.1107/S010827019101154X.
- ↑ Leban, Ivan; Golič, Ljubo; Speakman, J. Clare (1973). "Crystal structures of the acid salts of some dibasic acids. Part VII. An X-ray study of potassium hydrogen acetylenedicarboxylate: The α-form". J. Chem. Soc., Perkin Trans. 2 (6): 703. doi:10.1039/P29730000703.
- ↑ Keisuke Miyakubo (1994). Nuclear magnetic resonance studies of dynamicals structure of one-dimensional hyrogen-bonded system in the acid salts of some dicarboxylic acids. Ph.D. Thesis, Osaka University.
- ↑ Ming Xing LI, Min SHAO, Hui DAI, Bao Li AN, Wen Cong LU, Yu ZHU, Chen Xia DU (2005), "Synthesis and Crystal Structure of a Novel Copper (II) Complex with Acetylenedicarboxylate and 2,2'-Bipyridine". Chinese Chemical Letters, volume 16, issue 10, pp. 1405–1408.
- ↑ Min Shao, Ming-Xing Li, Hui Dai, Wen-Cong Lu and Bao-Li An (2007), "Polynuclear complexes incorporating Cu(II) and Mn(II) centers bridged by acetylenedicarboxylate: Structure, thermal stability and magnetism". Journal of Molecular Structure, volume 829, issues 1–3, pp. 155–160.
- ↑ Ruth Ahlers and Uwe Ruschewitz (2009), "Non-centrosymmetric coordination polymers based on thallium and acetylenedicarboxylate". Solid State Sciences, volume 11, issue 6, pp. 1058–1064.