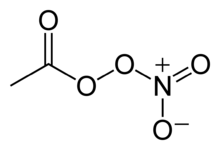
Peroxyacetyl nitrate
 | |
 | |
| Names | |
|---|---|
| IUPAC name
nitroethaneperoxoate | |
| Systematic IUPAC name
ethanoic nitric peroxyanhydride | |
| Other names
PAN peroxyacetyl nitrate α-oxoethylperoxylnitrate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.017.187 |
| EC Number | 218-905-6 |
PubChem CID |
|
| |
| |
| Properties | |
| C2H3NO5 | |
| Molar mass | 121.05 g mol−1 |
| 1.46 × 10 5 mg l−1 at 298 K | |
| log P | −0.19 |
| Vapor pressure | 29.2 mmHg at 298 K |
Henry's law constant (kH) |
0.000278 m3 atm mol−1 at 298 K |
Atmospheric OH rate constant |
10−13 cm3 molecule−1 s−1 at 298 K |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Peroxyacetyl nitrate is a peroxyacyl nitrate. It is a secondary pollutant present in photochemical smog. It is thermally unstable and decomposes into peroxyethanoyl radicals and nitrogen dioxide gas. It is a lachrymatory substance.
Peroxyacetyl nitrate, or PAN, is an oxidant that is more stable than ozone. Hence, it is more capable of long-range transport than ozone. It serves as a carrier for oxides of nitrogen (NOx) into rural regions and causes ozone formation in the global troposphere.
External links
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.