Bodroux–Chichibabin aldehyde synthesis
The Bodroux–Chichibabin aldehyde synthesis is a chemical reaction whereby a Grignard reagent is converted to an aldehyde one carbon longer.
Reaction of a Grignard reagent with triethyl orthoformate gives an acetal, which can be hydrolyzed to an aldehyde. For example, the synthesis of n-hexanal:[1]
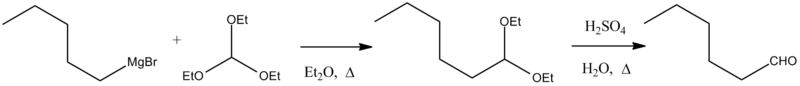 Bodroux-Chichibabin hexanal.[1]
Bodroux-Chichibabin hexanal.[1]
See also
References
- 1 2 G. Bryant Bachman (1943). "n-Hexaldehyde". Organic Syntheses. ; Collective Volume, 2, p. 323
- Bodroux, F. (1904). "Synthése d'aldehydes aromatiques" [Synthesis of aromatic aldehydes]. Compt. Rend. (in French). 138: 92.
- Tschitschibabin, A. E. (1904). "Eine neue allgemeine Darstellungsmethode der Aldehyde". Ber. (in German). 37: 186. doi:10.1002/cber.19040370133.
- Tschitschibabin, A. E. (1904). "Ueber den Hexahydro-m-toluylaldehyd". Ber. (in German). 37: 850. doi:10.1002/cber.190403701140.
- Smith, L. I.; Bayliss, M. (1941). "The Bodroux-Tschitschibabin, and the Bouveault Aldehyde Syntheses". J. Org. Chem. 6 (3): 437. doi:10.1021/jo01203a009.
- Smith, L. I.; Nichols, J. (1941). "The Synthesis of Aldehydes from Grignard Reagents. II. Polymethylbenzaldehydes". J. Org. Chem. 6 (4): 489. doi:10.1021/jo01204a003.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.
