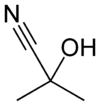
Acetone cyanohydrin
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
2-Hydroxy-2-methylpropanenitrile[1] | |||
| Other names | |||
| Identifiers | |||
3D model (JSmol) |
|||
| 3DMet | B00479 | ||
| 605391 | |||
| ChEBI | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.000.828 | ||
| EC Number | 200-909-4 | ||
| KEGG | |||
| MeSH | acetone+cyanohydrin | ||
PubChem CID |
|||
| RTECS number | OD9275000 | ||
| UN number | 1541 | ||
| |||
| |||
| Properties | |||
| C4H7NO | |||
| Molar mass | 85.11 g·mol−1 | ||
| Appearance | Colourless liquid | ||
| Density | 932 mg mL−1 | ||
| Melting point | −21.2 °C; −6.3 °F; 251.9 K | ||
| Boiling point | 95 °C (203 °F; 368 K) | ||
| Vapor pressure | 2 kPa (at 20 °C) | ||
Refractive index (nD) |
1.399 | ||
| Thermochemistry | |||
Std enthalpy of formation (ΔfH |
−121.7–−120.1 kJ mol−1 | ||
Std enthalpy of combustion (ΔcH |
−2.4514–−2.4498 MJ mol−1 | ||
| Hazards | |||
| Safety data sheet | fishersci.com | ||
| GHS pictograms |   | ||
| GHS signal word | DANGER | ||
| H300, H310, H330, H410 | |||
| P260, P273, P280, P284, P301+310 | |||
| NFPA 704 | |||
| Flash point | 75 °C (167 °F; 348 K) | ||
| Explosive limits | 2.25–11% | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) |
| ||
| US health exposure limits (NIOSH): | |||
PEL (Permissible) |
none[2] | ||
REL (Recommended) |
C 1 ppm (4 mg/m3) [15-minute][2] | ||
IDLH (Immediate danger) |
N.D.[2] | ||
| Related compounds | |||
Related alkanenitriles |
|||
Related compounds |
DBNPA | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Acetone cyanohydrin (ACH) is an organic compound used in the production of methyl methacrylate, the monomer of the transparent plastic polymethyl methacrylate (PMMA), also known as acrylic. It is extremely toxic.
Preparation and use
In the laboratory, this compound may be prepared by treating sodium cyanide with acetone, followed by acidification:[3]
Considering the high toxicity of acetone cyanohydrin, a lab scale production has been developed using microreactor technology.[4] Alternatively, a simplified procedure involves the action of sodium or potassium cyanide on the sodium bisulfite adduct of acetone prepared in situ. This gives a less pure product that is suitable for most syntheses.[5]
Reactions
It is used as a surrogate in place of HCN, as illustrated by this synthesis of lithium cyanide: [6]
- (CH3)2C(OH)CN + LiH → (CH3)2CO + LiCN + H2
In transhydrocyanation, an equivalent of HCN is transferred from acetone cyanohydrin to another acceptor. The transfer is an equilibrium process, initiated by base. The reaction can be driven by trapping reactions or by the use of a superior HCN acceptor, such as an aldehyde.[7]
Acetone cyanohydrin is an intermediate en route to methyl methacrylate. Treated with sulfuric acid give the sulfate ester of the methacrylamide, methanolysis of which gives ammonium bisulfate and MMA.[8]
Natural occurrence
Cassava tubers contain linamarin, a glucoside of acetohydrin, and the enzyme linamarinase for hydrolysing the glucoside. Crushing the tubers releases these compounds and produces acetone cyanohydrin.
Safety
Acetone cyanohydrin is classified as an extremely hazardous substance in the US Emergency Planning and Community Right-to-Know Act. The principal hazards of acetone cyanohydrin arise from its ready decomposition on contact with water, which releases highly toxic hydrogen cyanide.
References
- ↑ "acetone cyanohydrin - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 16 September 2004. Identification. Retrieved 8 June 2012.
- 1 2 3 4 5 6 7 "NIOSH Pocket Guide to Chemical Hazards #0005". National Institute for Occupational Safety and Health (NIOSH).
- ↑ Cox, R. F. B.; Stormont, R. T. "Acetone Cyanohydrin". Organic Syntheses. ; Collective Volume, 2, p. 7
- ↑ Heugebaert, Thomas S. A.; Roman, Bart I.; De Blieck, Ann; Stevens, Christian V. (2010-08-11). "A safe production method for acetone cyanohydrin". Tetrahedron Letters. 51 (32): 4189–4191. doi:10.1016/j.tetlet.2010.06.004.
- ↑ Wagner, E. C.; Baizer, Manuel. "5,5-Dimethylhydantoin". Organic Syntheses. ; Collective Volume, 3, p. 323
- ↑ Tom Livinghouse (1981). "Trimethylsilyl Cyanide: Cyanosilylation of p-Benzoquinone". Org. Synth. 60: 126. doi:10.15227/orgsyn.060.0126.
- ↑ Serkos A. Haroutounian (2001). "Acetone Cyanohydrin". eEROS. doi:10.1002/047084289X.ra014.
- ↑ Bauer, Jr., William (2002). "Methacrylic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a16_441. ISBN 3-527-30673-0. .