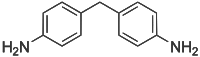
4,4'-Methylenedianiline
 | |
| Names | |
|---|---|
| IUPAC name
Bis(4-aminophenyl)methane | |
| Other names
4,4'-Diaminodiphenylmethane; 4,4'-Methylenebisbenzenamine; MDA; para, para'-Diaminodiphenyl-methane; Dianilinomethane; 4,4'-Diphenylmethanediamine | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.705 |
| EC Number | 202-974-4 |
| KEGG | |
PubChem CID |
|
| RTECS number | BY5425000 |
| UNII | |
| UN number | 2651 |
| |
| |
| Properties | |
| C13H14N2 | |
| Molar mass | 198.27 g·mol−1 |
| Appearance | Pale brown, crystalline solid |
| Odor | faint, amine-like[1] |
| Density | 1.05 g/cm3 (100°C) |
| Melting point | 89 °C (192 °F; 362 K) |
| Boiling point | 398 to 399 °C (748 to 750 °F; 671 to 672 K) |
| 0.125 g/100 ml (20 °C) | |
| Vapor pressure | 0.0000002 mmHg (20°C)[1] |
| Hazards | |
| Main hazards | potential carcinogen[1] |
| Flash point | 190 °C; 374 °F; 463 K [1] |
| US health exposure limits (NIOSH): | |
PEL (Permissible) |
TWA 0.010 ppm ST 0.100 ppm[1] |
REL (Recommended) |
Ca[1] |
IDLH (Immediate danger) |
Ca [N.D.][1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
4,4'-Methylenedianiline (MDA) is an organic compound with the formula CH2(C6H4NH2)2. It is a colorless or white solid. It is produced on industrial scale as a precursor to polyurethanes.
Synthesis and applications
In the industrial production, MDA is produced by reaction of formaldehyde and aniline in the presence of hydrochloric acid.[2] This reaction consumes the majority of aniline produced worldwide.[3]
MDA is consumed mainly as a precursor to 4,4 ́-methylene diphenyl diisocyanate (MDI). MDA is treated with phosgene to give MDI. MDI is a precursor to many polyurethane foams.[4][5] Lower quantities are used as hardeners in epoxy resins and adhesives, as well as in the production of high-performance polymers.[2] MDA is hydrogenated to give 4,4-diaminodicyclohexylmethane, which is also used in polymer chemistry.
Safety
MDA is considered a potential occupational carcinogen by the US National Institute for Occupational Safety and Health. The Occupational Safety and Health Administration has set a permissible exposure limit at 0.01 ppm over an eight-hour time-weighted average, and a short-term exposure limit at 0.10 ppm.[6]
It is suspected carcinogen.[4] It is included in the "substances of very high concern" list of the European Chemicals Agency (ECHA).[5] The compound was blamed in an mass poisoning in the vicinity of Epping, Essex, United Kingdom during 1965 during which 84 individuals were poisoned through accidental contamination of flour used to make bread.[7]
References
- 1 2 3 4 5 6 7 "NIOSH Pocket Guide to Chemical Hazards #0415". National Institute for Occupational Safety and Health (NIOSH).
- 1 2 Data on manufacture, import, export, uses and release of 4-4’ diaminodiphenylmethane as well as ... Archived 2011-10-01 at the Wayback Machine.
- ↑ Christian Six, Frank Richter "Isocyanates, Organic" in Ulmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a14_611
- 1 2 ToxFAQs for 4,4'-Methylenedianiline, Agency for Toxic Substances and Disease Registry
- 1 2 Background document for 4,4’-Diaminodiphenylmethane (MDA)
- ↑ NIOSH Pocket Guide on Chemical Hazards
- ↑ Kopelman, H; Robertson, MH; Sanders, PG; Ash, I. "The Epping jaundice". Br Med J. 1: 514–6. doi:10.1136/bmj.1.5486.514. PMC 1843808. PMID 5902696.
External links
- International Labour Organization icsc1111
- Centers for Disease Control and Prevention NIOSH Pocket Guide to Chemical Hazards
- European Union Risk Assessment Report
- J.H. Petersen, S.K. Mortensen, G.A. Pedersen, Memorandum for the Danish Veterinary and Food Administration on An acute case of primary aromatic amines migrating from cooking utensils, Danish Institute for Food and Veterinary Research, 12 October 2004