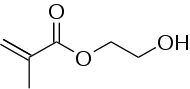
(Hydroxyethyl)methacrylate
 | |
 | |
| Names | |
|---|---|
| IUPAC name
2-Hydroxyethyl 2-methylprop-2-enoate | |
| Other names
HEMA; hydroxyethylmethacrylate; glycol methacrylate; glycol monomethacrylate; hydroxyethyl methacrylate; ethylene glycol methacrylate; 2-(methacryloyloxy)ethanol | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.011.621 |
| KEGG | |
| UNII | |
| |
| |
| Properties | |
| C6H10O3 | |
| Molar mass | 130.14 g·mol−1 |
| Appearance | Colourless liquid |
| Density | 1.07 g/cm3 |
| Melting point | −99 °C (−146 °F; 174 K)[1] |
| Boiling point | 213 °C (415 °F; 486 K)[1] |
| log P | 0.50[2] |
| Vapor pressure | 0.08 hPa |
| Hazards | |
| Main hazards | Eye irritation |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Hydroxyethylmethacrylate or HEMA is the monomer that is used to make the polymer polyhydroxyethylmethacrylate. The polymer is hydrophobic; however, when the polymer is subjected to water it will swell due to the molecule's hydrophilic pendant group. Depending on the physical and chemical structure of the polymer, it is capable of absorbing from 10 to 600% water relative to the dry weight. Because of this property, it was one of the first materials to be successfully used in the manufacture of soft contact lenses[3]
Use in 3D printed glass
HEMA is also used as the monomeric matrix in which 40 nm silica particles are suspended for 3D glass printing.[4]
References
- 1 2 "GPS Safety Summary 2-Hydroxyethyl methacrylate (HEMA)". July 2013.
- ↑ "2-hydroxyethyl methacrylate_msds".
- ↑ Blasco, Joe; Kehoe, Vincent J-R; The professional make-up artist : motion pictures, television, print, theatre; ISBN 0-9771580-0-4; LCC# PN2068.B53 2005
- ↑ Kotz (20 April 2017). "Three-dimensional printing of transparent fused silica glass". Nature. Retrieved 27 April 2017.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.