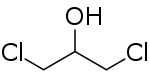
1,3-Dichloropropan-2-ol
 | |
| Names | |
|---|---|
| IUPAC name
1,3-Dichloropropan-2-ol | |
| Other names
1,3-Dichloropropanol; 1,3-Dichloro-2-propanol; 1,3-Dichloroisopropanol; Glycerol-α,γ-dichlorohydrin; α-Dichlorohydrin; 1,3-DCP; DC2P | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.266 |
| EC Number | 202-491-9 |
| KEGG | |
PubChem CID |
|
| RTECS number | UB1400000 |
| UNII | |
| UN number | 2750 |
| |
| |
| Properties | |
| C3H6Cl2O | |
| Molar mass | 128.98 g·mol−1 |
| Appearance | Colorless liquid[1] |
| Odor | Phenol-like[1] |
| Density | 1.39 g/cm3 (20 °C)[1] |
| Melting point | −4 °C (25 °F; 269 K)[1] |
| Boiling point | 175 °C (347 °F; 448 K)[1] |
| 110 g/L (20 °C)[1] | |
| Hazards | |
| GHS pictograms |    |
| GHS signal word | Danger |
| H301, H301, H311, H312, H336, H350, H370, H373 | |
| P201, P202, P260, P261, P264, P270, P271, P280, P281, P301+310, P302+352, P304+340, P307+311, P308+313, P312, P314, P321, P322, P330, P361, P363, P403+233, P405, P501 | |
| Flash point | 74 °C (165 °F; 347 K)[1] |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
110 mg/kg (oral, rat)[1] 1080 mg/kg (dermal, rabbit)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
1,3-Dichloropropan-2-ol (1,3-DCP) is a chemical compound in the class of chloropropanols. It is used as a solvent and in the manufacture of some paints, lacquers, and photographic materials[2] as well as being an intermediate in the production of epichlorohydrin.[3]
1,3-DCP is a believed to be a carcinogen and mutagen.[2][4] The International Agency for Research on Cancer classifies it as a Group 2B carcinogen ("possibly carcinogenic to humans").[5]
Along with 3-monochloropropane-1,2-diol (3-MCPD), 1,3-DCP is found in some Asian style sauces such as soy sauce and oyster sauce.[6][7][8]
References
- 1 2 3 4 5 6 7 8 9 Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- 1 2 "Hazardous Substance Fact Sheet: 1,3-Dichloropropanol" (PDF). New Jersey Department of Health and Senior Services.
- ↑ "1,3-Dichloro-2-propanol". Some Chemicals Present in Industrial and Consumer Products, Food and Drinking-Water.
- ↑ "Evidence on the Carcinogenicity of 1,3-Dichloro-2-Propanol (1,3-DCP; α,γ-Dichlorohydrin)" (PDF). Reproductive and Cancer Hazard Assessment Branch Office of Environmental Health Hazard Assessment, California Environmental Protection Agency. June 2010.
- ↑ "List of Classifications". International Agency for Research on Cancer.
- ↑ Genualdi, Susan; Nyman, Patricia; Dejager, Lowri (2017). "Simultaneous Analysis of 3-MCPD and 1,3-DCP in Asian Style Sauces Using QuEChERS Extraction and Gas Chromatography–Triple Quadrupole Mass Spectrometry". Journal of Agricultural and Food Chemistry. 65 (4): 981. doi:10.1021/acs.jafc.6b05051. PMID 28064506.
- ↑ Crews, C; Lebrun, G; Brereton, P. A (2002). "Determination of 1,3-dichloropropanol in soy sauces by automated headspace gas chromatography-mass spectrometry". Food Additives and Contaminants. 19 (4): 343. doi:10.1080/02652030110098580. PMID 11962691.
- ↑ Lee, Bai Qin; Khor, Sook Mei (2015). "3-Chloropropane-1,2-diol (3-MCPD) in Soy Sauce: A Review on the Formation, Reduction, and Detection of This Potential Carcinogen". Comprehensive Reviews in Food Science and Food Safety. 14: 48. doi:10.1111/1541-4337.12120.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.