Homoaconitic acid
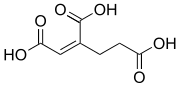 cis-Homoaconitic acid | |
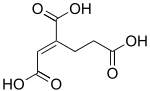 trans-Homoaconitic acid | |
| Names | |
|---|---|
| IUPAC names
(1Z)-1-Butene-1,2,4-tricarboxylic acid (1E)-1-Butene-1,2,4-tricarboxylic acid | |
| Other names
Homo-cis-aconitate; Homo-trans-aconitate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider |
|
PubChem CID |
|
| |
| Properties | |
| C7H8O6 | |
| Molar mass | 188.14 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Homoaconitatic acid (homoaconitate) is related to aconitic acid but with one extra carbon. It is part of the α-aminoadipate pathway for lysine biosynthesis, where it is made from homocitrate by homoaconitase. It is converted to homoisocitrate by homoisocitrate dehydrogenase.
See also
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.